Chemistry:Angustmycin A
From HandWiki
| |
| Names | |
|---|---|
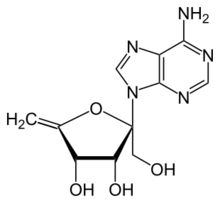
| IUPAC name
2-(6-aminopurin-9-yl)-2-(hydroxymethyl)-5-methylideneoxolane-3,4-diol[1]
| |
| Other names
Decoyinine
4′,5′-Didehydro-1′-C-(hydroxymethyl)adenosine | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C11H13N5O4 | |
| Molar mass | 279.256 g·mol−1 |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H315, H319, H335 | |
| P261, P264, P264+265Script error: No such module "Preview warning".Category:GHS errors, P271, P280, P302+352, P304+340, P305+351+338, P319Script error: No such module "Preview warning".Category:GHS errors, P321, P332+317Script error: No such module "Preview warning".Category:GHS errors, P337+317Script error: No such module "Preview warning".Category:GHS errors, P362+364Script error: No such module "Preview warning".Category:GHS errors, P403+233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Angustmycin A is a purine antibiotic and metabolite from Streptomyces bacteria with the molecular formula C11H13N5O4.[3][1] Angustmycin A is also a cytokinin.[4][5][6]
References
- ↑ 1.0 1.1 "Angustmycin A" (in en). Pubchem.ncbi.NLM.nih.gov. https://pubchem.ncbi.nlm.nih.gov/compound/2970#section=3D-Conformer.
- ↑ "Decoyinine" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/121578#section=Safety-and-Hazards.
- ↑ Scott, Thomas (20 January 2020) (in en). Concise encyclopedia of biochemistry. Walter de Gruyter GmbH & Co KG. p. 32. ISBN 978-3-11-171099-0.
- ↑ Yu, Le; Zhou, Wenting; She, Yixuan; Ma, Hongmin; Cai, You-Sheng; Jiang, Ming; Deng, Zixin; Price, Neil P. J. et al. (17 November 2021). "Efficient biosynthesis of nucleoside cytokinin angustmycin A containing an unusual sugar system" (in en). Nature Communications 12 (1): 6633. doi:10.1038/s41467-021-26928-y. ISSN 2041-1723. PMID 34789759. Bibcode: 2021NatCo..12.6633Y.
- ↑ Gottlieb, David; Shaw, Paul D. (6 December 2012) (in en). Mechanism of Action. Springer Science & Business Media. p. 465. ISBN 978-3-642-46051-7.
- ↑ Korzybski, Tadeusz; Kowszyk-Gindifer, Zuzanna; Kurylowicz, Wlodzimierz (3 September 2013) (in en). Antibiotics: Origin, Nature and Properties. Elsevier. ISBN 978-1-4832-2305-6.
Further reading
- Hoeksema, H.; Slomp, G.; van Tamelen, E. E. (1 January 1964). "Angustmycin A and decoyinine" (in en). Tetrahedron Letters 5 (27): 1787–1795. doi:10.1016/S0040-4039(01)89489-2. ISSN 0040-4039.
- McCarthy, James R.; Robins, Roland K.; Robins, Morris J. (August 1968). "Purine nucleosides. XXII. Synthesis of angustmycin A (decoyinine) and related unsaturated nucleosides". Journal of the American Chemical Society 90 (18): 4993–4999. doi:10.1021/ja01020a038. PMID 5665542.
 |
