Chemistry:Cadmium permanganate
From HandWiki
| |
| Identifiers | |
|---|---|
| |
3D model (JSmol)
|
|
PubChem CID
|
|
| |
| |
| Properties | |
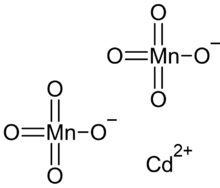
| Cd(MnO4)2 | |
| Molar mass | 350.28 |
| Appearance | dark purple crystals[1] |
| soluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Cadmium permanganate is an inorganic compound with the chemical formula Cd(MnO4)2. It can form the hexahydrate Cd(MnO4)2·6H2O.
Preparation
Cadmium permanganate can be obtained by reacting cadmium sulfate and barium permanganate. After removing the barium sulfate precipitate, the solution is crystallized in the dark:[1]
- Ba(MnO
4)
2 + CdSO
4 → Cd(MnO
4)
2 + BaSO
4↓
Cadmium permanganate can also be produced by reacting manganese heptoxide with cadmium oxide[2] or cadmium hydroxide.[3]
Properties
Cadmium permanganate hexahydrate loses water of crystallisation at 61~62 °C, and anhydrate can be obtained at a constant temperature of 90 °C. The anhydrous form begins to decompose at 108 °C:[3]
- Cd(MnO
4)
2 → CdMnO
3 + MnO
2 + 3/
2 O
2↑
References
- ↑ 1.0 1.1 Kótai, László; Banerji, Kalyan K. (2001-03-31). "An Improved Method for the Preparation of High-Purity Permanganate Salts" (in en). Synthesis and Reactivity in Inorganic and Metal-Organic Chemistry 31 (3): 491–495. doi:10.1081/SIM-100002234. ISSN 0094-5714. http://www.tandfonline.com/doi/abs/10.1081/SIM-100002234. Retrieved 2020-06-21.
- ↑ Kótai, László; Sajó, István E.; Gács, István; Sharma, Pradeep K.; Banerji, Kalyan K. (Jul 2007). "Convenient Routes for the Preparation of Barium Permanganate and other Permanganate Salts" (in de). Zeitschrift für anorganische und allgemeine Chemie 633 (8): 1257–1260. doi:10.1002/zaac.200700142. http://doi.wiley.com/10.1002/zaac.200700142. Retrieved 2020-06-21.
- ↑ 3.0 3.1 Zhang, Zhongsheng; Wu, Jigui; Zhang, Yanni (1991-07-02). "Research on Kinetic Parameters of Cadmium Permanganate by Thermogravimetry" (in zh). Journal of Lanzhou University (Natural Science) 27 (2). doi:10.13885/j.issn.0455-2059.1991.02.019. http://dx.doi.org/10.13885/j.issn.0455-2059.1991.02.019.
 |

