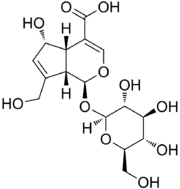
Chemistry:Deacetylasperulosidic acid
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
(1S,4aS,5S,7aS)-5-Hydroxy-7-(hydroxymethyl)-1-(((2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-1,4a,5,7a-tetrahydrocyclopenta[c]pyran-4-carboxylic acid
| |
| Other names
10-Deacetylasperulosidic acid; 10-Desacetylasperulosidic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C16H22O11 | |
| Molar mass | 390.341 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Deacetylasperulosidic acid is an iridoid compound found in a few medicinal plants, such as Morinda citrifolia.[1] Some in vitro and in vivo bioactivities of deacetylasperulosidic acid include anti-inflammatory, analgesic, anti-cancer, antioxidant, anti-arthritic, anti-mutagenic, anti-clastogenic, and hepatoprotection.[2][3][4][5][6]
References
- ↑ "Identification of TLC markers and quantification by HPLC-MS of various constituents in noni fruit powder and commercial noni-derived products". J. Agric. Food Chem. 55 (18): 7489–7494. 2007. doi:10.1021/jf071359a. PMID 17696360.
- ↑ Akihisa, T; Matsumoto, K; Tokuda, H; Yasukawa, K; Seino, K; Nakamoto, K; Kuninaga, H; Suzuki, T et al. (2007). "Anti-inflammatory and potential cancer chemopreventive constituents of the fruits of Morinda citrifolia (Noni)". Journal of Natural Products 70 (5): 754–7. doi:10.1021/np068065o. PMID 17480098.
- ↑ Liu, G; Bode, A; Ma, WY; Sang, S; Ho, CT; Dong, Z (2001). "Two novel glycosides from the fruits of Morinda citrifolia (noni) inhibit AP-1 transactivation and cell transformation in the mouse epidermal JB6 cell line.". Cancer Research 61 (15): 5749–56. PMID 11479211.
- ↑ Ling, SK; Tanaka, T; Kouno, I (2003). "Effects of iridoids on lipoxygenase and hyaluronidase activities and their activation by beta-glucosidase in the presence of amino acids.". Biological & Pharmaceutical Bulletin 26 (3): 352–6. doi:10.1248/bpb.26.352. PMID 12612446. http://naosite.lb.nagasaki-u.ac.jp/dspace/bitstream/10069/8383/1/BPBul26_352.pdf.
- ↑ "Iridoid glycosides isolated from Oldenlandia diffusa inhibit LDL-oxidation". Arch Pharm Res 28 (10): 1156–1160. 2005. doi:10.1007/BF02972979. PMID 16276972.
- ↑ Nakamura, T; Nakazawa, Y; Onizuka, S; Satoh, S; Chiba, A; Sekihashi, K; Miura, A; Yasugahira, N et al. (1997). "Antimutagenicity of Tochu tea (an aqueous extract of Eucommia ulmoides leaves): 1. The clastogen-suppressing effects of Tochu tea in CHO cells and mice". Mutation Research 388 (1): 7–20. doi:10.1016/s1383-5718(96)00096-4. PMID 9025787.
 |

