Chemistry:Diethyl sulfoxide
From HandWiki
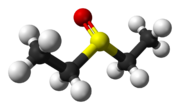
| |
| Names | |
|---|---|
| Preferred IUPAC name
(Ethanesulfinyl)ethane | |
| Other names
DESO, diethyl sulphoxide, ethyl sulfoxide, 1,1'-sulfinylbisethane
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C4H10OS | |
| Molar mass | 106.18 g·mol−1 |
| Appearance | colorless viscous liquid |
| Density | 1.066 g/cm3 |
| Melting point | 14 °C (57 °F; 287 K) |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
5650 mg/kg (oral, rat) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Diethyl sulfoxide is the organosulfur compound with the formula (C
2H
5)
2SO. It is closely related to the more common dimethyl sulfoxide.
Diethyl sulfoxide prepared by oxidation of diethyl sulfide.[2]
References
- ↑ MSDS for diethyl sulfoxide
- ↑ Rostami, Amin; Akradi, Jamal (2010). "A highly efficient, green, rapid, and chemoselective oxidation of sulfides using hydrogen peroxide and boric acid as the catalyst under solvent-free conditions". Tetrahedron Letters 51 (27): 3501–3503. doi:10.1016/j.tetlet.2010.04.103.
 |
