Chemistry:Digitoxigenin
From HandWiki
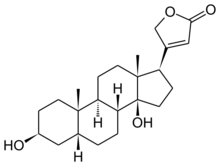
| |
| Names | |
|---|---|
| IUPAC name
3β,14-Dihydroxy-5β-card-20(22)-enolide
| |
| Systematic IUPAC name
4-[(1R,3aS,3bR,5aR,7S,9aS,9bS,11aR)-5a,7-Dihydroxy-9a,11a-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-1-yl]furan-2(5H)-one | |
| Other names
3-β,14-dihydroxy-5-β,14-β-card-20(22)-enolide
5β-Card-20(22)-enolide, 3β,14-dihydroxy- Card-20(22)-enolide, 3,14-dihydroxy-, (3β,5β)- NSC 407806 Δ20:22-3,14,21-Trihydroxynorcholenic acid lactone cerberigenin echujetin evonogenin thevetigenin | |
| Identifiers | |
3D model (JSmol)
|
|
| 95448 | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| C23H34O4 | |
| Molar mass | 374.51 |
| Appearance | solid[citation needed] |
| Melting point | 252 to 253 °C (486 to 487 °F; 525 to 526 K) |
| Hazards | |
| Main hazards | Toxic |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Digitoxigenin, a cardenolide, is the aglycone of digitoxin.
Digitoxigenin can be used to prepare actodigin.[2]
In Lednicer's book on steroids, it is made from deoxycholic acid.
References
- ↑ http://www.sigmaaldrich.com/catalog/ProductDetail.do?N4=D9404%7CSIGMA&N5=SEARCH_CONCAT_PNO%7CBRAND_KEY&F=SPEC [|permanent dead link|dead link}}]
- ↑ Ferland, J. M. (1974). "Synthetic Cardenolides and Related Products. III. Isocardenolides". Canadian Journal of Chemistry 52 (9): 1652–1661. doi:10.1139/v74-239. ISSN 0008-4042.
 |
