Chemistry:Echinochrome A
| |
| Names | |
|---|---|
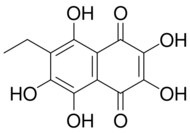
| Preferred IUPAC name
6-ethyl-2,3,5,7,8-pentahydroxynaphthalene-1,4-dione | |
| Other names
7-ethyl-2,3,5,6,8-pentahydroxy-1,4-nafphtoquinone; 6-Ethyl-2,7-trihydroxynaphthazarin; 6-ethyl-2,3,5,7,8-pentahydroxy-2-Ethyl-3,5,6,7,8-pentahydroxy-[1,4]naphthoquinone
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C12H10O7 | |
| Molar mass | 266.205 g·mol−1 |
| Appearance | Dark red crystalline powder |
| Melting point | −219 to 221.5 °C (−362.2 to 430.7 °F; 54.1 to 494.6 K) |
| Practically insoluble | |
| Solubility | Moderately soluble in ethanol; very slightly soluble in chloroform |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Echinochrome A (7-ethyl-2,3,5,6,8-pentahydroxy-1,4-naphthoquinone) is a polyhydroxylated 1,4-naphthoquinone,[1] a type of pigments commonly found in sea urchin shell ("test"), spine, gonads, coelomic fluid, and eggs, of sea urchin.[2] These types of pigments are commonly known as spinochromes and are natural marine phenolic compounds with potential pharmacological effects and modes of action.[3]
First extracted from the sea urchin Scaphechinus mirabilis, it is the active substance of histochrome[4] and Echino-A. Histochrome is used for ophthalmic diseases and ischemic heart disease. Echino-A has been used in nutraceutical form to diminish glucose levels, cholesterol and tryglicerides. The properties of echinochrome A as an antioxidant has made it the subject of scientific and clinical studies for more than 30 years.[5][6][7][8][9][10][11]
The several hydroxyl groups have the ability to diminish reactive oxygen species (ROS) in cells, preventing redox imbalance. Echinochrome A has been found to target ophthalmologic, cardiovascular, cerebrovascular, inflammatory and metabolic diseases through its biological functions by targeting specific molecular signals. The regulation effects produced by echinochrome A in the cells makes this molecule a candidate to improve health.[7][12] Sea urchins are known for their putative health properties for centuries, for example in the Materia medica of the Ming Dynasty authored by Li Zhongli in 1647.[13]
See also
- Ozopromide
References
- ↑ CID 135457951 from PubChem Retrieved April 13, 2022
- ↑ Anderson, H.A.; Mathieson, J.W.; Thomson, R.H. (January 1969). "Distribution of spinochrome pigments in echinoids". Comparative Biochemistry and Physiology 28 (1): 333–345. doi:10.1016/0010-406X(69)91347-4. PMID 5777380. Bibcode: 1969CmpBP..28..333A.
- ↑ Jeong, Seung; Kim, Hyoung; Song, In-Sung; Lee, Seon; Ko, Kyung; Rhee, Byoung; Kim, Nari; Mishchenko, Natalia et al. (13 May 2014). "Echinochrome A Protects Mitochondrial Function in Cardiomyocytes against Cardiotoxic Drugs". Marine Drugs 12 (5): 2922–2936. doi:10.3390/md12052922. PMID 24828295.
- ↑ Mishchenko, N. P.; Fedoreev, S. A.; Bagirova, V. L. (2003). "Histochrome: A New Original Domestic Drug". Pharmaceutical Chemistry Journal 37: 48–52. doi:10.1023/A:1023659331010.
- ↑ Hou, Yakun; Vasileva, Elena A.; Carne, Alan; McConnell, Michelle; El-Din A. Bekhit, Alaa; Mishchenko, Natalia P. (2018). "Naphthoquinones of the spinochrome class: occurrence, isolation, biosynthesis and biomedical applications". RSC Advances 8 (57): 32637–32650. doi:10.1039/C8RA04777D. PMID 35547692. Bibcode: 2018RSCAd...832637H.
- ↑ Rubilar, Tamara; Barbieri, Elena S.; Gazquez, Ayelén; Avaro, Marisa (May 2021). "Sea Urchin Pigments: Echinochrome A and Its Potential Implication in the Cytokine Storm Syndrome". Marine Drugs 19 (5): 267. doi:10.3390/md19050267. PMID 34064550.
- ↑ 7.0 7.1 Kim, Hyoung Kyu; Vasileva, Elena A.; Mishchenko, Natalia P.; Fedoreyev, Sergey A.; Han, Jin (August 2021). "Multifaceted Clinical Effects of Echinochrome". Marine Drugs 19 (8): 412. doi:10.3390/md19080412. PMID 34436251.
- ↑ Tang, Xiaokang; Nishimura, Akiyuki; Ariyoshi, Kohei; Nishiyama, Kazuhiro; Kato, Yuri; Vasileva, Elena; Mishchenko, Natalia; Fedoreyev, Sergey et al. (2023). "Echinochrome Prevents Sulfide Catabolism-Associated Chronic Heart Failure after Myocardial Infarction in Mice". Marine Drugs 21: 52. doi:10.3390/md21010052.
- ↑ doi:10.1016/j.biopha.2023.114589 PMID 37004327
- ↑ Khalaf, Marina Lotfy; Soliman, Amel Mahmoud; Fahmy, Sohair Ramadan; Mohamed, Ayman Saber (2025). "Anti-thrombotic Mechanisms of Echinochrome a on Arterial Thrombosis in Rats: In-silico, In-vitro and In-vivo Studies". Cardiovascular & Hematological Agents in Medicinal Chemistry 23 (2): 143–160. doi:10.2174/0118715257332064241104114546. PMID 39533475.
- ↑ doi:10.1016/j.ecoenv.2024.117489 PMID 39644572
- ↑ Artyukov, Aleksandr A.; Zelepuga, Elena A.; Bogdanovich, Larisa N.; Lupach, Natalia M.; Novikov, Vyacheslav L.; Rutckova, Tatyana A.; Kozlovskaya, Emma P. (May 2020). "Marine Polyhydroxynaphthoquinone, Echinochrome A: Prevention of Atherosclerotic Inflammation and Probable Molecular Targets". Journal of Clinical Medicine 9 (5): 1494. doi:10.3390/jcm9051494. PMID 32429179.
- ↑ Barbieri, Elena Susana; Rubilar, Tamara; Gázquez, Ayelén; Avaro, Marisa; Seiler, Erina Noé; Vera-Piombo, Mercedes; Gittardi, Agustín; Chaar, Florencia et al. (29 June 2020). Sea Urchin Pigments as Potential Therapeutic Agents Against the Spike Protein of SARS-CoV-2 Based on in Silico Analysis. doi:10.26434/chemrxiv.12568595.v1. This content is a preprint and has not been peer-reviewed
 |
