Chemistry:Formylhydrazine
From HandWiki
Short description: Chemical compound
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Formohydrazide[1] | |
| Other names
Formic hydrazide
| |
| Identifiers | |
3D model (JSmol)
|
|
| 635759 | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| CH4N2O | |
| Molar mass | 60.056 g·mol−1 |
| Melting point | 54 °C (129 °F; 327 K) |
| yes | |
| Solubility | ethanol |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H315, H319, H335, H336 | |
| P261, P264, P271, P280, P302+352, P304+340, P305+351+338, P312, P321, P332+313, P337+313, P362, P403+233, P405, P501 | |
| Related compounds | |
Related compounds
|
acetyl hydrazine, benzoyl hydrazine, diformyl hydrazine, N-methyl-N-formylhydrazine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
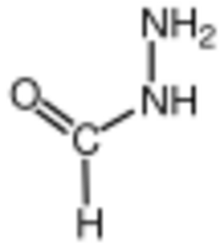
Formylhydrazine is a chemical compound with the molecular formula CH4N2O and it has a mass of 60 g/mol. It is also known as formic acid hydrazide, hydrazinecarboxaldehyde, formohydrazide, or formic hydrazide. It is one of the simplest compounds in the hydrazide class. Formylhydrazine can act as a bidentate ligand with cobalt, zinc,[2] or cadmium.[3]
Formation
Formylhydrazine can be produced by the acid hydrolysis of diazomethane. H2CN2 + H2O → HC(O)NHNH2.[4]
Properties
Formylhydrazine causes lung cancer in mice.[5]
References
- ↑ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. pp. 872. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ↑ Srinivasan, Krishnan; Govindarajan, Subbaiah; Harrison, William T.A. (July 2009). "Divalent metal complexes of formylhydrazine: Syntheses and crystal structures of M(CH4N2O)2(H2O)2·2NO3 (M=Zn, Co)". Inorganic Chemistry Communications 12 (7): 619–621. doi:10.1016/j.inoche.2009.05.002.
- ↑ Kharitonov, Yu Ya; Machkhoshvili, R. I.; Metreveli, D. P.; Pirtshalava, N. I. (1977). "Complex compounds of nitrates and thiocyanates of metals with formyl hydrazine" (in Russian). Koordinatsionnaya Khimiya 3 (6): 897–901. https://inis.iaea.org/search/search.aspx?orig_q=RN:9391026.
- ↑ Müller, Eugen (2013) (in de). Neuere Anschauungen der Organischen Chemie: Organische Chemie für Fortgeschrittene. Springer-Verlag. p. 453. ISBN 978-3-642-87591-5. https://books.google.com/books?id=a9_QBgAAQBAJ&pg=PA453.
- ↑ Arcos, Joseph C.; Woo, Yin-Tak; Argus, Mary F. (2013) (in en). Aliphatic Carcinogens: Structural Bases and Biological Mechanisms. Elsevier. p. 367. ISBN 978-1-4832-6372-4. https://books.google.com/books?id=nSQSBQAAQBAJ&pg=PA367.
 |
