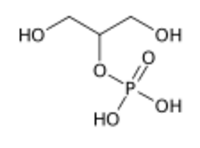
Chemistry:Glycerol 2-phosphate
| |
| Names | |
|---|---|
| Preferred IUPAC name
1,3-Dihydroxypropan-2-yl dihydrogen phosphate | |
| Other names
1,2,3-Propanetriol, 2-(dihydrogen phosphate)
Glycerol 2-phosphate β-Glycerophosphate β-Phosphoglycerol BGP | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| EC Number |
|
| KEGG | |
| MeSH | Beta-glycerophosphoric+acid |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C3H9O6P | |
| Molar mass | 172.073 g·mol−1 |
| Appearance | forms colorless salts |
| Related compounds | |
Related organophosphates
|
Glycerol 1-phosphate Glycerol 3-phosphate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Glycerol 2-phosphate is the conjugate base of phosphoric ester of glycerol. It is commonly known as β-glycerophosphate or BGP. Unlike glycerol 1-phosphate and glycerol 3-phosphate, this isomer is not chiral. It is also less common.
Applications
β-Glycerophosphate is an inhibitor of the enzyme serine/threonine phosphatase. It is often used in combination with other phosphatase/protease inhibitors for broad spectrum inhibition.[1][2]
Although previously presumed to be non-transportable and reliant on extracellular phosphatases such as alkaline phosphatase (PhoA) for utilization, recent research indicates that Escherichia coli can use β-Glycerophosphate as a sole phosphorus source in the absence of PhoA, relying on the Ugp transporter system encoded by the ugpBAECQ operon.[3]
β-Glycerophosphate is also used to drive osteogenic differentiation of bone marrow stem cells in vitro.[4]
β-Glycerophosphate is used to buffer M17 media for Lactococcus culture in recombinant protein expression.[5]
Formation
Glycerol 2-phosphate (G2P) can be formed through abiotic phosphorylation under prebiotic conditions, as demonstrated in laboratory simulations of early Earth environments.[6] In the presence of urea and heat, glycerol and phosphate undergo regioselective phosphorylation, favoring the 2-position to yield G2P.[6] This process suggests a plausible prebiotic pathway for the synthesis of key metabolic intermediates like G2P, which may have contributed to the origin of biological phospholipid synthesis prior to enzymatic catalysis.[6]
Notes
- ↑ "β-Glycerophosphate disodium salt hydrate". Sigma-Aldrich. https://www.sigmaaldrich.com/catalog/product/sigma/g6376. Retrieved 2018-05-30.
- ↑ "Protease and Phosphatase Inhibitors". Thermo Fisher Scientific. https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/protease-phosphatase-inhibitors.html. Retrieved 2018-05-30.
- ↑ Yang, Kechao; Wang, Mi; Metcalf, William W. (2009-07-15). "Uptake of Glycerol-2-Phosphate via the ugp-Encoded Transporter in Escherichia coli K-12". Journal of Bacteriology 191 (14): 4667–4670. doi:10.1128/jb.00235-09. PMID 19429609.
- ↑ Langenbach & Handschel (2013). "Effects of dexamethasone, ascorbic acid and β-glycerophosphate on the osteogenic differentiation of stem cells in vitro". Stem Cell Research & Therapy 4 (5): 117. doi:10.1186/scrt328. PMID 24073831.
- ↑ Terzaghi & Sandine (1975). "Improved medium for lactic streptococci and their bacteriophages". Applied Microbiology 29 (6): 807–813. doi:10.1128/AM.29.6.807-813.1975. PMID 16350018.
- ↑ 6.0 6.1 6.2 Zhu, Cheng; Turner, Andrew M.; Abplanalp, Matthew J.; Kaiser, Ralf I.; Webb, Bill; Siuzdak, Gary; Fortenberry, Ryan C. (2020-08-01). "An Interstellar Synthesis of Glycerol Phosphates". The Astrophysical Journal Letters 899 (1): L3. doi:10.3847/2041-8213/aba744. ISSN 2041-8205. Bibcode: 2020ApJ...899L...3Z.
 |
