Chemistry:Higher sulfur oxides
From HandWiki
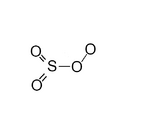
Higher sulfur oxides are a group of chemical compounds with the formula SO3+x where x lies between 0 and 1. They contain peroxo (O−O) groups, and the oxidation state of sulfur is +6 as in SO3.
Monomeric SO4 can be isolated at low temperatures (below 78 K) following the reaction of SO3 and atomic oxygen or photolysis of SO3–ozone mixtures. The favoured structure is:
Colourless polymeric condensates are formed in the reaction of gaseous SO3 or SO2 with O2 in a silent electric discharge. The structure of the polymers is based on β-SO3 (one of the three forms of solid SO3) with oxide bridges (−O−) replaced randomly by peroxide bridges (−O−O−). As such these compounds are non-stoichiometric.
References
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
 |