Chemistry:Iron bis(diethyldithiocarbamate)
| |
| |
| Identifiers | |
|---|---|
| |
3D model (JSmol)
|
|
| ChemSpider |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C20H40Fe2N4S8 | |
| Molar mass | 704.74 g·mol−1 |
| Appearance | red solid |
| Density | 1.457 g/cm3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
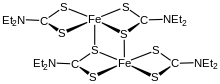
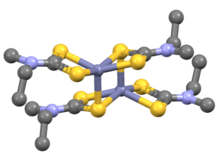
Iron bis(diethyldithiocarbamate) is a coordination complex with the formula [Fe(S
2CNEt
2)
2]
2 where Et = C
2H
5. A red solid, it is representative of several ferrous dithiocarbamates with diverse substituents in place of ethyl. In terms of structure, the species is dimeric, consisting of two pentacoordinate iron(II) centers. It is isostructural with [Zn(S
2CNEt
2)
2]
2, which in turn is similar to zinc bis(dimethyldithiocarbamate).[1]
Reactions
The complex reacts with a variety of reagents with concomitant formation of mono-iron derivatives. 9,10-Phenanthroline adds to iron bis(diethyldithiocarbamate) to give the blue-octahedral complex [Fe(S
2CNEt
2)
2](phen). 3,4-Bis(trifluoromethyl)-1,2-dithiete reacts to give the dithiolene complex [Fe(S
2CNEt
2)
2](S
2C
2(CF
3)
2).[2] Nitric oxide and carbon monoxide to give the nitrosyl complex Fe(S
2CNEt
2)
2NO and the carbonyl complex Fe(S
2CNEt
2)
2(CO)
2, respectively.
Related compounds
References
- ↑ Ileperuma, Oliver A.; Feltham, Robert D. (1975). "Crystal and Molecular Structure of Iron(II) Bis(diethyldithiocarbamate)". Inorganic Chemistry 14 (12): 3042–3045. doi:10.1021/ic50154a037.
- ↑ Coucouvanis, Dimitri (1979). "The Chemistry of the Dithioacid and 1,1-Dithiolate Complexes, 1968–1977". Prog. Inorg. Chem.. Progress in Inorganic Chemistry 26: 301–469. doi:10.1002/9780470166277.ch5. ISBN 9780470166277.
 |

