Chemistry:Lead picrate
From HandWiki
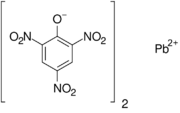
| |
| Names | |
|---|---|
| IUPAC name
Lead(II) 2,4,6-trinitrophenolate
| |
| Other names
Lead dipicrate
| |
| Identifiers | |
3D model (JSmol)
|
|
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C12H4N6O14Pb | |
| Molar mass | 663.4 g·mol−1 |
| Appearance | Orange powder |
| 7.06 g/L [1] | |
| Solubility | Soluble in ethanol, acetic acid[1] Very soluble in DMF,[1] dichloromethane[2] |
| Hazards | |
| GHS pictograms |    
|
| GHS Signal word | Danger |
| H201, H302, H332, H360, H373, H410 | |
| P260, P261, P264, P270, P271, P273, P280, P304+340, P330, P391, P405, P501 | |
| 270.75-327.15 °C (519.7-621.0 °F; 600.3-543.9 K)[2] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Lead picrate is an organic picrate salt.[3] It is a sensitive and highly explosive compound that is typically found as a hydrate.[2] Dry lead picrate is incredibly dangerous and cannot be handled without explosive decomposition occurring.
History
Lead picrate was first discovered in the early 1900s. It was investigated by numerous militaries during the First World War as a potential primary explosive, most notably Germany for using it in blasting caps.[2]
Preparation
Numerous lead precursors can be used to create lead picrate. Two of the simplest examples of lead picrate synthesis are the addition of lead(II) oxide or lead carbonate with picric acid.[2]
References
- ↑ 1.0 1.1 1.2 Yang, Li; Pei, Qin; Zhang, Tonglai; Zhang, Jianguo; Cao, Yunling (2007). "Solubilities and enthalpies of solution of picric acid and picrates at 298.15K in DMF, EtOH and acetic acid" (in en). Thermochimica Acta 463 (1–2): 13–14. doi:10.1016/j.tca.2007.04.013. https://linkinghub.elsevier.com/retrieve/pii/S0040603107001621.
- ↑ 2.0 2.1 2.2 2.3 2.4 Matsukakwa, Makoto; Matsunaga, Takehiro; Yoshida, Masatake; Fujiwara, Shuzo (2004). "Synthesis and properties of lead picrates". Science and Technology of Energetic Materials 65 (1): 7–13.
- ↑ Jai Prakash Agrawal (2015). High Energy Materials: Propellants, Explosives and Pyrotechnics. John Wiley & Sons.
 |
