Chemistry:Lometraline
 | |
| Clinical data | |
|---|---|
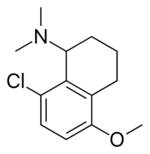
| Other names | N,N-dimethyl-8-chloro-5-methoxy-1-aminotetralin |
| ATC code |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C13H18ClNO |
| Molar mass | 239.74 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Lometraline (INN; developmental code name CP-14,368) is a drug and an aminotetralin derivative.[1] A structural modification of tricyclic neuroleptics, lometraline was originally patented by Pfizer as an antipsychotic, tranquilizer, and antiparkinsonian agent.[2][3] However, it was instead later studied as a potential antidepressant and/or anxiolytic agent, though clinical studies revealed no psychoactivity at the doses used and further investigation was suspended.[1][4][5] Further experimental modifications of the chemical structure of lometraline resulted in the discovery of tametraline, a potent inhibitor of the reuptake of dopamine and norepinephrine, which in turn led to the discovery of the now widely popular antidepressant sertraline, a selective serotonin reuptake inhibitor (SSRI).[6]
The precursor ketone can undergo a Mannich reaction with pyrrolidine to yield CID:12348935. This specific agent had analgesic activity in the same range as morphine and was not antagonized by naloxone.[7][8]
References
- ↑ 1.0 1.1 "Evaluation of an aminotetraline, CP 14.368, as an antidepressant". Current Therapeutic Research, Clinical and Experimental 14 (2): 65–70. February 1972. PMID 4401233.
- ↑ Saunders pharmaceutical word book, 1994. W.B. Saunders Co.. 27 January 1994. ISBN 978-0-7216-5254-2. https://books.google.com/books?id=Qk5rG8fVVGEC. Retrieved 27 April 2012.
- ↑ United States. Patent Office (1972). "Lometraline: tranquillizer, anti-parkinson agent". Official gazette of the United States Patent Office: Patents. The Office. https://books.google.com/books?id=u1VLybXT98IC. Retrieved 27 April 2012.
- ↑ Psychopharmacological agents. Futura Pub. Co.. 1974. ISBN 978-0-87993-052-3. https://books.google.com/books?id=vwdtAAAAMAAJ. Retrieved 27 April 2012.
- ↑ Bayerische Julius-Maximilians-Universität Würzburg. Arbeitskreis für Schlafforschung (1973). The Nature of sleep. Die Natur des Schlafes. La nature du sommeil: International symposium, Würzburg, 23-26.9.1971. G. Fischer. ISBN 978-3-437-10295-0. https://books.google.com/books?id=MuBqAAAAMAAJ. Retrieved 27 April 2012.
- ↑ "Discovery of sertraline (Zoloft®)". 2006. http://oasys2.confex.com/acs/231nm/techprogram/P936639.htm.
- ↑ "Analgesic and tranquilizing activity of 5,8-disubstituted 1-tetralone Mannich bases". Journal of Medicinal Chemistry 20 (5): 699–705. May 1977. doi:10.1021/jm00215a016. PMID 853506.
- ↑ "Analgesic and tranquilizing activity of 5,8-disubstituted 2-aminomethyl-3,4-dihydronaphthalenes". Journal of Medicinal Chemistry 21 (3): 257–63. March 1978. doi:10.1021/jm00201a004. PMID 628000.
 |
