Chemistry:Melanotan II
| |
| Names | |
|---|---|
| Pronunciation | /mɛˈlænoʊtæn/ (Audio file "Pronunciation-melanotan.ogg" not found) |
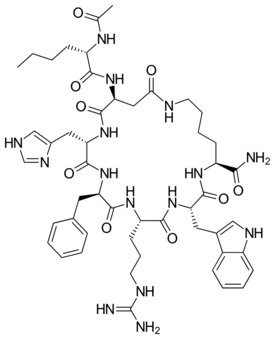
| Systematic IUPAC name
L-Lysinamide, N-acetyl-L-norleucyl-L-alpha-aspartyl-L-histidyl-D-phenylalanyl-L-arginyl-L-tryptophyl-, cyclic (2-7)-peptide | |
| Other names
List of other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| MeSH | melanotan-II |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C50H69N15O9 | |
| Molar mass | 1024.180 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |

Melanotan II is a synthetic analogue of the peptide hormone α-melanocyte-stimulating hormone (α-MSH) that stimulates melanogenesis to facilitate tanning. It may also increase sexual arousal.
It was developed as a successor to afamelanotide ("Melanotan I"), an FDA-approved drug operating through a similar pathway. Clinuvel Pharmaceuticals intended to offer it as a cosmetic, but abandoned this pursuit in the 2000s due to regulatory restrictions and concerns about the promotion of suntanning. Unlicensed Melanotan II is found on the internet, although health agencies advise against its use due to lack of testing and regulatory approval.
Melanotan-II may cause reversible darkening of moles and freckles. It is unclear if Melanotan II can increase (or reduce) the risk of developing melanoma, because reports of melanomas associated with its use have coincided with heavy UV exposure and sun bed use.[1][2] A 2013 scientific review found there was no conclusive evidence it causes melanoma,[1] and a 2021 review concluded "the increased risk of melanoma in Melanotan users, who use it for tanning and exhibit sun-seeking behaviour, can probably be explained by more UV exposure".[2]
Side effects may include facial flushing, nausea and erection in males.
Synthesis
In the synthesis of melanotan II, an ε-amino group of lysine and an γ-carboxy group of aspartic acid have their orthogonal protection removed before undergoing a carbodiimide mediated lactamization, leading to an intermediate. This intermediate, when attached to N-acetylnorleucine, forms melanotan II. The entire process can be accomplished in 12 steps with an overall yield of 2.6%, and the product is more than 90% pure without preparative chromatography.[3]
Mechanism of action
Melanotan II acts as a non-selective agonist of the melanocortin receptors MC1, MC3, MC4, and MC5.[4]
Melanotan II produces melanogenesis by activation of the MC1 receptor, whereas its clinically documented sexual effects are thought to be related to its ability to activate the MC4 receptor (though the MC3 is thought to also possibly be involved).[5][6]
Other effects of melanotan II, mostly regarded as adverse effects, include flushing, nausea, vomiting, stretching, yawning, and loss of appetite (the last via activation of MC4).[7][8]
History and research
Research in the early 1960s showed that in rats, administration of α-MSH caused sexual arousal, and work on this continued in many labs up through the 1980s, when scientists at the University of Arizona began attempting to develop α-MSH and analogs as potential sunless tanning agents, and synthesized and tested several analogs, including melanotan-I and melanotan II.[6][9]
Early in the research process one of the scientists, who was conducting experiments on himself with an early tool compound, melanotan II, injected himself with twice the dose he intended to and got an eight-hour erection, along with nausea and vomiting.[6][non-primary source needed]
As a tanning agent, melanotan I (now known as afamelanotide) was licensed by Competitive Technologies, a technology transfer company operating on behalf of the University of Arizona, to an Australian startup called Epitan,[10][11] which changed its name to Clinuvel in 2006.[12] Afamelanotide was approved by the FDA in 2019.[2]
As a sexual dysfunction agent, melanotan II was licensed by Competitive Technologies to Palatin Technologies.[9] Palatin ceased development of melanotan II in 2000 and synthesized, patented, and began to develop bremelanotide, a likely metabolite of melanotan II that differs in that it has a carboxy group where melanotan II has an amide.[6][13]
Clinuvel Pharmaceuticals intended to offer it as a cosmetic, but abandoned this pursuit in the 2000s.[14]
Competitive Technologies (Clinuvel) sued Palatin for breach of contract and tried to claim ownership of bremelanotide;[13] the parties settled in 2008 with Palatin retaining rights to bremelanotide, returning rights to melanotan II to Competitive Technologies, and paying US$800,000.[15]
Melanotan-II may cause reversible darkening of moles and freckles. It is unclear if Melanotan II can increase (or reduce) the risk of developing melanoma. Reports of melanomas associated with the use of Melanotan II have coincided with heavy sunbathing and sun bed use.[1][2] A 2013 scientific review found there was no conclusive evidence it causes melanoma,[1] and a 2021 review concluded "the increased risk of melanoma in Melanotan users, who use it for tanning and exhibit sun-seeking behaviour, can probably be explained by more UV exposure".[2] A 2020 in vivo study found that Melanotan II suppressed the progression of melanomas.[16]
A 2019 study showed Melanotan-II having improved sociability deficits in mice exhibiting autistic features.[17]
Society and culture
Numerous products are sold online and in gyms and beauty salons as "melanotan" or "melanotan-1", "melanotan-2" or "Barbie drug" in their marketing.[18][19][20][21] Sale of these products for human use is illegal in many jurisdictions.[22][23][24][25]
Starting in 2007, health agencies in various countries began issuing warnings against their use.[26][27][28][29][30][31]
See also
- List of investigational sexual dysfunction drugs
- Decapaptide-12
- Methoxsalen
References
- ↑ 1.0 1.1 1.2 1.3 Javed, Muhammad; Yarrow, Jeremy; Hemington gorse, Sarah (2013-10-01). "Does melanotan injections (TAN JAB) cause melanoma? A systemic review of the effects of melanotan injections". International Journal of Surgery 11 (8): 677. doi:10.1016/j.ijsu.2013.06.485. ISSN 1743-9191.
- ↑ 2.0 2.1 2.2 2.3 2.4 Wensink, Debby; Wagenmakers, Margreet A.E.M.; Langendonk, Janneke G. (2021-02-01). "Afamelanotide for prevention of phototoxicity in erythropoietic protoporphyria". Expert Review of Clinical Pharmacology 14 (2): 151–160. doi:10.1080/17512433.2021.1879638. ISSN 1751-2433. PMID 33507118.
- ↑ Ryakhovsky, Vladimir V; Khachiyan, Georgy A; Kosovova, Nina F; Isamiddinova, Elena F; Ivanov, Andrey S (30 October 2008). "The first preparative solution phase synthesis of melanotan II". Beilstein Journal of Organic Chemistry 4: 39. doi:10.3762/bjoc.4.39. PMID 19043625.
- ↑ Wikberg, Jarl ES (2001). "Melanocortin receptors: new opportunities in drug discovery". Expert Opinion on Therapeutic Patents 11 (1): 61–76. doi:10.1517/13543776.11.1.61. ISSN 1354-3776.
- ↑ Norris, David O.; Lopez, Kristin H. (25 November 2010). Hormones and Reproduction of Vertebrates. Academic Press. pp. 4–. ISBN 978-0-08-095809-5. https://books.google.com/books?id=HKv3mC5Eb34C&pg=RA4-PA152.
- ↑ 6.0 6.1 6.2 6.3 "Melanocortin receptors, melanotropic peptides and penile erection". Curr Top Med Chem 7 (11): 1098–1106. 2007. doi:10.2174/1568026610707011111. PMID 17584130.
- ↑ Plant, Tony M.; Zeleznik, Anthony J. (15 November 2014). Knobil and Neill's Physiology of Reproduction: Two-Volume Set. Academic Press. pp. 2230–. ISBN 978-0-12-397769-4. https://books.google.com/books?id=I1ACBAAAQBAJ&pg=PA2230.
- ↑ Wein, Alan J.; Kavoussi, Louis R.; Novick, Andrew C.; Partin, Alan W.; Peters, Craig A. (28 September 2011). Campbell-Walsh Urology. Elsevier Health Sciences. pp. 743–. ISBN 978-1-4557-2298-3. https://books.google.com/books?id=W1aeyJD46kIC&pg=PA743.
- ↑ 9.0 9.1 "Discovery that a melanocortin regulates sexual functions in male and female humans". Peptides 26 (10): 1687–9. October 2005. doi:10.1016/j.peptides.2005.01.023. PMID 15996790.
- ↑ "EpiTan focuses on Melanotan, a potential blockbuster". The Pharma Letter. 1 November 2004. https://www.thepharmaletter.com/article/epitan-focuses-on-melanotan-a-potential-blockbuster.
- ↑ "Melanocortin peptide therapeutics: historical milestones, clinical studies and commercialization". Peptides 27 (4): 921–30. April 2006. doi:10.1016/j.peptides.2005.01.029. PMID 16412534.
- ↑ "Epitan changes name to Clinuvel, announces new clinical program". LabOnline. 27 February 2006. http://www.labonline.com.au/content/life-scientist/news/epitan-changes-name-to-clinuvel-announces-new-clinical-program-161764385.
- ↑ 13.0 13.1 "Press Release: Palatin Technologies Refutes Competitive Technologies Contention of Material Breach". Palatin Technologies via PR Newswire. September 12, 2007. http://www.prnewswire.com/news-releases/palatin-technologies-refutes-competitive-technologies-contention-of-material-breach-58006787.html.
- ↑ Madrigal, Alexis (2024-03-07). "Suntan Drug Greenlighted for Trials | WIRED". Wired. https://www.wired.com/2009/01/tan/. Retrieved 2025-01-21.
- ↑ "Press Release: Palatin Technologies Announces Litigation Settlement With Competitive Technologies". Palatin Technologies via PR Newswire. January 22, 2008. http://www.prnewswire.com/news-releases/palatin-technologies-announces-litigation-settlement-with-competitive-technologies-57107302.html.
- ↑ Wu, Jian-Ching; Tsai, Han-En; Hsiao, Yi-Hsiang; Wu, Ji-Syuan; Wu, Chieh-Shan; Tai, Ming-Hong (2020-01-20). "Topical MTII Therapy Suppresses Melanoma Through PTEN Upregulation and Cyclooxygenase II Inhibition" (in en). International Journal of Molecular Sciences 21 (2): 681. doi:10.3390/ijms21020681. PMID 31968661.
- ↑ Minakova, Elena; Lang, Jordan; Medel-Matus, Jesus-Servando; Gould, Georgianna G.; Reynolds, Ashley; Shin, Don; Mazarati, Andrey; Sankar, Raman (2019). "Melanotan-II reverses autistic features in a maternal immune activation mouse model of autism". PLOS ONE 14 (1). doi:10.1371/journal.pone.0210389. ISSN 1932-6203. PMID 30629642. Bibcode: 2019PLoSO..1410389M.
- ↑ "Believe It Or Not 'Tanorexia' A Very Real Problem". WCBS-TV, CBS. 2009-05-20. http://wcbstv.com/topstories/tanorexia.melanotan.self.2.1013828.html.
- ↑ "Fools Gold". Cosmopolitan (Australia). 2009-06-14. http://www.cosmopolitan.com.au/fool_gold.htm.
- ↑ Madrigal, Alexis (2009-01-29). "Suntan Drug Greenlighted for Trials". Wired. https://www.wired.com/wiredscience/2009/01/tan/.
- ↑ Habbema, Louis; Halk, Anne Berthe; Neumann, Martino; Bergman, Wilma (2017-03-17). "Risks of unregulated use of alpha-melanocyte-stimulating hormone analogues: a review" (in en). International Journal of Dermatology 56 (10): 975–980. doi:10.1111/ijd.13585. ISSN 0011-9059. https://onlinelibrary.wiley.com/doi/10.1111/ijd.13585.
- ↑ "Tanning drug a health risk". Herald Sun. 2009-10-31. http://www.heraldsun.com.au/news/tanning-drug-a-health-risk/story-e6frf7jo-1225792923321.
- ↑ "Melanotropic peptides: more than just 'Barbie drugs' and 'sun-tan jabs'?". The British Journal of Dermatology 163 (3): 451–5. September 2010. doi:10.1111/j.1365-2133.2010.09891.x. PMID 20545686.
- ↑ "Change in moles linked to use of unlicensed "sun tan jab"". BMJ 338: b277. January 2009. doi:10.1136/bmj.b277. PMID 19174439.
- ↑ "Risky tan jab warnings 'ignored'". BBC. 2009-02-18. https://news.bbc.co.uk/2/hi/health/7895366.stm.
- ↑ "Warning against the product Melanotan". Danish Medicines Agency. 2008. http://www.dkma.dk/1024/visUKLSArtikel.asp?artikelID=13865.
- ↑ ""Tan jab" is an unlicensed medicine and may not be safe". MHRA. 2008. http://www.mhra.gov.uk/NewsCentre/Pressreleases/CON031009.
- ↑ "US Lab Research Inc Warning letter". U.S. Food and Drug Administration. 2009-01-29. https://www.fda.gov/ICECI/EnforcementActions/WarningLetters/ucm152426.htm.
- ↑ "Melanotan Powder for Injection". Notice Information: – Warning – 27 February 2009. Irish Medicines Board. 2009. http://www.imb.ie/EN/Safety--Quality/Advisory-Warning--Recall-Notices/Human-Medicines/Melanotan-Powder-for-Injection-.aspx.
- ↑ "Legemiddelverket advarer mot bruk av Melanotan". Norwegian Medicines Agency. 2007-12-13. http://www.slk.no/templates/InterPage____65110.aspx.
- ↑ "Melanotan – farlig og ulovlig brunfarge". Norwegian Medicines Agency. 2009-01-23. http://www.slk.no/templates/InterPage____80434.aspx.
 |
