Chemistry:Nitrourea
From HandWiki
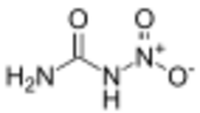
| |
| Names | |
|---|---|
| Preferred IUPAC name
Nitrourea | |
| Other names
N-Nitrocarbamide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C1H3N3O3 | |
| Molar mass | 105.05 g/mol |
| Appearance | White crystalline solid |
| Density | 1.73 g/cm3 |
| Boiling point | 155°C (decomposition) |
| Soluble | |
| Solubility | Soluble in ethanol, methanol, acetone Slightly soluble in benzene and chloroform |
| Explosive data | |
| Shock sensitivity | Medium |
| Friction sensitivity | Low |
| Detonation velocity | 6860 |
| RE factor | 1.01 |
| Related compounds | |
Related compounds
|
Urea nitrate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Nitrourea is a strong high explosive compound[1] synthesized by the nitration of urea or by way of a dehydration reaction of urea nitrate.[2]
References
- ↑ "Nitrourea". CAMEO Chemicals. NOAA. http://cameochemicals.noaa.gov/chemical/12124.
- ↑ Ingersoll, A. W.; Armendt, B. F. (1925). "Nitrourea". Organic Syntheses 5: 85. http://www.orgsyn.org/demo.aspx?prep=cv1p0417.; Collective Volume, 1, pp. 417
 |
