Chemistry:Octanitrocubane
Octanitrocubane (molecular formula: C8(NO2)8) is a proposed high explosive that, like TNT, is shock-insensitive (not readily detonated by shock).[1] The octanitrocubane molecule has the same chemical structure as cubane (C8H8) except that each of the eight hydrogen atoms is replaced by a nitro group (NO2). As of 1998, octanitrocubane had not been produced in quantities large enough to test its performance as an explosive.[2]
It is, however, not as powerful an explosive as once thought, as the high-density theoretical crystal structure has not been achieved. For this reason, heptanitrocubane, the slightly less nitrated form, is believed to have marginally better performance, despite having a worse oxygen balance.
Octanitrocubane is thought to have 20–25% greater performance than HMX (octogen). This increase in power is due to its highly expansive breakdown into CO2 and N2, as well as to the presence of strained chemical bonds in the molecule which have stored potential energy. In addition, it produces no water vapor upon combustion, making it less visible, and both the chemical itself and its decomposition products (nitrogen and carbon dioxide) are considered to be non-toxic.
Octanitrocubane was first synthesized by Philip Eaton (who was also the first to synthesize cubane in 1964) and Mao-Xi Zhang at the University of Chicago in 1999, with the structure proven by crystallographer Richard Gilardi of the United States Naval Research Laboratory.[3][4]
Synthesis
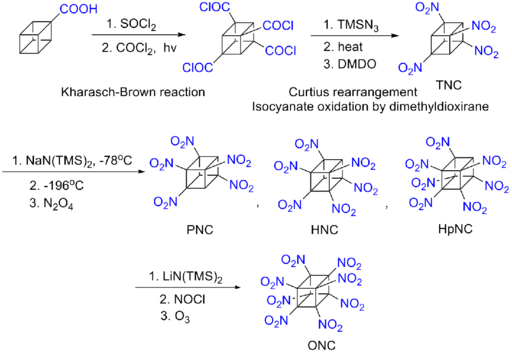
Although octanitrocubane is predicted to be one of the most effective explosives, the difficulty of its synthesis inhibits practical use. Philip Eaton's synthesis was difficult and lengthy, and required cubane (rare to begin with) as a starting point. As a result, octanitrocubane is more valuable, gram for gram, than gold.[5]
A proposed path to synthesis is the cyclotetramerization of the as yet undiscovered and presumably highly unstable dinitroacetylene.[6] [7]
See also
- Octaazacubane (N8)
- 4,4'-Dinitro-3,3'-diazenofuroxan (DDF)
- Hexanitrobenzene (HNB)
- Hexanitrohexaazaisowurtzitane (HNIW)
- HHTDD (Hexanitrohexaazatricyclododecanedione)
- Relative effectiveness factor
- TKX-50
References
- ↑ "Octanitrocubane: Easier said than done". University of Chicago News Office. March 20, 2001. http://www-news.uchicago.edu/releases/01/010320.explosive.shtml.
- ↑ Astakhov, A. M.; Stepanov, R. S.; Babushkin, A. Yu. (1998). "On the detonation parameters of octanitrocubane". Combustion, Explosion, and Shock Waves 34 (1): 85–87. doi:10.1007/BF02671823. Bibcode: 1998CESW...34...85A.
- ↑ Zhang, Mao-Xi; Eaton, Philip E.; Gilardi, Richard (2000). "Hepta- and Octanitrocubanes". Angewandte Chemie International Edition 39 (2): 401–404. doi:10.1002/(SICI)1521-3773(20000117)39:2<401::AID-ANIE401>3.0.CO;2-P. PMID 10649425.
- ↑ Eaton, Philip E.; Zhang, Mao-Xi; Gilardi, Richard; Gelber, Nat; Iyer, Sury; Surapaneni, Rao (2001). "Octanitrocubane: A New Nitrocarbon". Propellants, Explosives, Pyrotechnics 27 (1): 1–6. doi:10.1002/1521-4087(200203)27:1<1::AID-PREP1>3.0.CO;2-6.
- ↑ Krause, Horst H. (2004). "New Energetic Materials". in Teipel, Ulrich. Energetic Materials: Particle Processing and Characterization. pp. 1–25. ISBN 978-3-527-30240-6. http://www.wiley-vch.de/books/sample/3527302409_c01.pdf.
- ↑ Politzer, Peter; Lane, Pat; Wiener, John J. (8 June 1999). Cyclooligomerizations as Possible Routes to Cubane-Like Systems. ADA364287. OCLC 227895131. https://apps.dtic.mil/sti/pdfs/ADA364287.pdf.
- ↑ Harter, Lara; Bélanger-Chabot, Guillaume; Rahm, Martin (2025). "Dinitroacetylene: Can It be Made?". Propellants, Explosives, Pyrotechnics 50 (4). doi:10.1002/prep.12019. ISSN 0721-3115.
External links
 |