Chemistry:Oxatriquinane
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
(2as,4as,6as)-Octahydro-2a1λ4-oxacyclopenta[cd]pentalene-2a1(1H)-ylium | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| C9H15O | |||
| Molar mass | 139.218 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
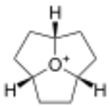
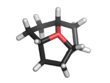
Oxatriquinane (oxoniaperhydrotriquinacene[1]) is an alkyl oxonium ion with formula (CH2CH2CH)3O+. It has a cyclononane backbone, with a tricoordinated oxygen connected to carbon 1, 4, and 7, forming three fused pentagonal rings. In contrast to most trialkyloxonium ions, oxatriquinane hydrolyzes slowly.
History
Oxatriquinane was first described in 2008. It five-step synthesis starts from 1,4,7-cyclononatriene.[2][3][4] Ita C–O bond lengths are 1.54 Å. The C−O−C angles are also acute.[2]
Reactions
Oxonium ions normally are strong alkylating agents and are hydrolytically sensitive. Oxatriquinane does not react with boiling water or with alcohols, thiols, halide ions, or amines, although it does react with stronger nucleophiles such as hydroxide, cyanide, and azide.[2] The ability of the oxygen to enter into a fourth covalent bond has been of some theoretical interest and was achieved using carborane acid.[5] As illustrated by the structures of most metal oxides, oxygen compounds routinely have bonds to >3 elements in other classes of compounds.
Analogues
Related species include oxatriquinacene,[2] the tri-unsaturated analogue, which is of interest as a possible precursor to oxaacepentalene, a neutral aromatic species. 1,4,7-tri-tert-butyloxatriquinane has also been synthesised; this compound contains significant amounts of intramolecular steric strain, resulting in further bond elongation to give C–O bond lengths of 1.622 Å, the longest recorded in any species.[6]
-
Oxatriquinacene
-
Oxaacepentalene
References
- ↑ Triquinane is more often used in the natural product literature to refer to three angularly fused cyclopentane rings sharing a common quaternary carbon in the center, a structure which contains one more carbon atom at the periphery. Moreover, substitution by O+ is more properly designated by the prefix oxonia. Nevertheless, the authors who first prepared the cation called it oxatriquinane, and the name has been perpetuated in the literature. A semisystematic name in line with standard nomenclature might be 2a1-oxonia-1,2,3,4,5,6-hexahydrotriquinacene.
- ↑ 2.0 2.1 2.2 2.3 Mark Mascal; Nema Hafezi; Nabin K. Meher; James C. Fettinger (2008). "Oxatriquinane and Oxatriquinacene: Extraordinary Oxonium Ions". Journal of the American Chemical Society 130 (41): 13532–13533. doi:10.1021/ja805686u. PMID 18798616.
- ↑ Rachel Petkewich (September 29, 2008). "Taming Alkyl Oxonium Ions: Fused tricyclic structure stabilizes famously reactive alkylating agents". Chemical and Engineering News 86 (39): 10. doi:10.1021/cen-v086n039.p010. http://pubs.acs.org/cen/news/86/i39/8639notw6.html.
- ↑ Tim Reid (3 October 2008). "Oxonium ions: Ring of stability". Nature Chemistry. doi:10.1038/nchem.70.
- ↑ Stoyanov, Evgenii S.; Gunbas, Gorkem; Hafezi, Nema; Mascal, Mark; Stoyanova, Irini V.; Tham, Fook S.; Reed, Christopher A. (11 January 2012). "The R3O+···H+ Hydrogen Bond: Toward a Tetracoordinate Oxadionium(2+) Ion". Journal of the American Chemical Society 134 (1): 707–714. doi:10.1021/ja209942s. PMID 22133059.
- ↑ Gunbas, Gorkem; Hafezi, Nema; Sheppard, William L.; Olmstead, Marilyn M.; Stoyanova, Irini V.; Tham, Fook S.; Meyer, Matthew P.; Mascal, Mark (18 November 2012). "Extreme oxatriquinanes and a record C–O bond length". Nature Chemistry 4 (12): 1018–1023. doi:10.1038/nchem.1502. PMID 23174982. Bibcode: 2012NatCh...4.1018G.
 |