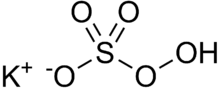
Chemistry:Potassium peroxymonosulfate
| |
| Names | |
|---|---|
| IUPAC name
Potassium peroxysulfate
| |
| Other names | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| KHSO5 | |
| Molar mass | 152.2 g/mol (614.76 g/mol as triple salt) |
| Appearance | Off-white powder |
| Decomposes | |
| Hazards | |
| Main hazards | Oxidant, corrosive |
| Safety data sheet | Fisher Scientific SDS |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related compounds
|
Potassium persulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Potassium peroxymonosulfate is widely used as an oxidizing agent, for example, in pools and spas (usually referred to as monopersulfate or "MPS") and as a biological disinfectant, Virkon. It is the potassium salt of peroxymonosulfuric acid.
Potassium peroxymonosulfate per se is rarely encountered. It is often confused with the triple salt 2KHSO
5 · KHSO
4 · K
2SO
4, known as Oxone.
The standard electrode potential for potassium peroxymonosulfate is +1.81 V with a half reaction generating the hydrogen sulfate (pH = 0):[3]
- HSO−
5 + 2H+
+ 2e−
→ HSO−
4 + H
2O
Oxone
Potassium peroxymonosulfate per se is a relatively obscure salt, but its derivative called Oxone is of commercial value. Oxone refers to the triple salt 2KHSO
5 · KHSO
4 · K
2SO
4. As such about one third by weight is potassium peroxymonosulfate. Oxone has a longer shelf life than does potassium peroxymonosulfate. A white, water-soluble solid, Oxone loses <1% of its oxidizing power per month.[4]
Oxone, which is commercially available, is produced from peroxysulfuric acid, which is generated in situ by combining oleum and hydrogen peroxide. Careful neutralization of this solution with potassium hydroxide allows the crystallization of the triple salt.
Uses
Cleaning
Oxone is used widely for cleaning. It whitens dentures,[5] oxidizes organic contaminants in swimming pools,[6] and cleans chips for the manufacture of microelectronics.[5][7][8]
Organic oxidations
Oxone is a versatile oxidant in organic synthesis. It oxidizes aldehydes to carboxylic acids; in the presence of alcoholic solvents, the esters may be obtained.[9] Internal alkenes may be cleaved to two carboxylic acids (see below), while terminal alkenes may be epoxidized. Sulfides give sulfones, tertiary amines give amine oxides, and phosphines give phosphine oxides.
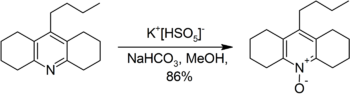
Further illustrative of the oxidative power of this salt is the conversion of an acridine derivative to the corresponding acridine-N-oxide.[10]
Oxone oxidizes sulfides to sulfoxides and then to sulfones.[11]
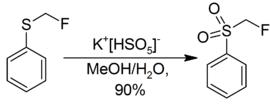
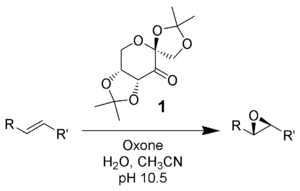
Oxone converts ketones to dioxiranes, which are used for diverse oxidations in organic synthesis. The dominant reagent dimethyldioxirane (DMDO) forms upon treatment of acetone with oxone. Dioxiranes are versatile, especially for the epoxidation of olefins.[12] Dioxiranes are also oxidize other unsaturated functionality, heteroatoms, and alkane C-H bonds.[13]
Oxone is used in the production of some organic periodinanes, notably the oxidation of 2-iodobenzoic acid to 2-iodoxybenzoic acid (IBX).[14]

Other uses
Oxone has been investigated for the delignification of wood.[15]
Ammonium, sodium, and potassium salts of HSO−
5 are used in the plastics industry as radical initiators for polymerization. They are also used as etchants, oxidative desizing agents for textile fabrics, and for decolorizing and deodorizing oils.
References
- ↑ Wu, Mingsong; Xu, Xinyang; Xu, Xun (November 2014). "Algicidal and Bactericidal Effect of Potassium Monopersulfate Compound on Eutrophic Water". Applied Mechanics and Materials 707: 259. doi:10.4028/www.scientific.net/AMM.707.259.
- ↑ "DuPont MSDS". http://www.sfm.state.or.us/CR2K_SubDB/MSDS/POTASSIUM_MONOPERSULFATE_POOL_CHEM.PDF.
- ↑ Spiro, M. (1979). "The standard potential of the peroxosulphate/sulphate couple". Electrochimica Acta 24 (3): 313–314. doi:10.1016/0013-4686(79)85051-3. ISSN 0013-4686.
- ↑ Crandall, Jack K.; Shi, Yian; Burke, Christopher P.; Buckley, Benjamin R. (2001) (in en). Encyclopedia of Reagents for Organic Synthesis. John Wiley & Sons, Ltd. doi:10.1002/047084289x.rp246.pub3. ISBN 978-0-470-84289-8.
- ↑ 5.0 5.1 Harald Jakob; Stefan Leininger; Thomas Lehmann; Sylvia Jacobi; Sven Gutewort. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_177.pub2.
- ↑ Pool School. Trouble Free Pool. p. PT4. https://books.google.com/books?id=WWBODwAAQBAJ&pg=PT4. Retrieved November 30, 2018.
- ↑ Peroxy Compounds Human Health and Ecological Draft Risk Assessment DP 455445, 455446 (Report). United States Environmental Protection Agency. 2020-03-11. p. 9-10. https://www.regulations.gov/search?filter=EPA-HQ-OPP-2017-0354-0006. Retrieved 2021-09-24.
- ↑ Wacławek, Stanisław; Lutze, Holger V.; Grübel, Klaudiusz; Padil, Vinod V.T.; Černík, Miroslav; Dionysiou, Dionysios. D. (2017-12-15). "Peroxy Compounds Human Health and Ecological Draft Risk Assessment DP 455445, 455446". Chemical Engineering Journal 330: 44–62. doi:10.1016/j.cej.2017.07.132.
- ↑ Benjamin R. Travis; Meenakshi Sivakumar; G. Olatunji Hollist; Babak Borhan (2003). "Facile Oxidation of Aldehydes to Acids and Esters with Oxone". Organic Letters 5 (7): 1031–4. doi:10.1021/ol0340078. PMID 12659566.
- ↑ Bell, Thomas W.; Cho, Young-Moon; Firestone, Albert; Healy, Karin; Liu, Jia; Ludwig, Richard; Rothenberger, Scott D. (1990). "9-n-Butyl-1,2,3,4,5,6,7,8-Octahydroacridin-4-ol". Organic Syntheses 69: 226. doi:10.15227/orgsyn.069.0226.
- ↑ McCarthy, James R.; Matthews, Donald P.; P. Paolini, John (1995). "Reaction of Sulfoxides with Diethylaminosulfur Trifluoride". Organic Syntheses 72: 209. doi:10.15227/orgsyn.072.0209.
- ↑ Adam, W.; Saha-Moller, C.; Zhao, C.-G. (2004). "Dioxirane Epoxidation of Alkenes". Org. React. 61: 219. doi:10.1002/0471264180.or061.02.
- ↑ Adam, W.; Zhao, C.-G.; Jakka, K. (2007). "Dioxirane Oxidations of Compounds other than Alkenes". Org. Reactions 69: 1. doi:10.1002/0471264180.or069.01.
- ↑ Frigerio, M.; Santagostino, M.; Sputore, S. (1999). "A User-Friendly Entry to 2-Iodoxybenzoic Acid (IBX)". J. Org. Chem. 64 (12): 4537–4538. doi:10.1021/jo9824596.
- ↑ Springer, E. L.; McSweeny, J. D. (1993). "Treatment of softwood kraft pulps with peroxymonosulfate before oxygen delignification". TAPPI Journal 76 (8): 194–199. ISSN 0734-1415. http://www.tappi.org/Bookstore/Technical-Papers/Journal-Articles/TAPPI-JOURNAL/Archives/1993/August/Treatment-of-softwood-kraft-pulps-with-peroxymonosulfate-before-oxygen-delignification-TAPPI-JOURNA.aspx. Retrieved 2011-05-14.
 |

