Chemistry:Potassium trithiocarbonate
From HandWiki
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| K 2CS 3 | |
| Molar mass | 186.39 g·mol−1 |
| Appearance | White solid, often brown due to impurities |
| Related compounds | |
Related compounds
|
Trithiocarbonic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
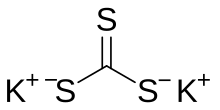
Potassium trithiocarbonate is the inorganic compound with the chemical formula K
2CS
3. It is the potassium salt of trithiocarbonic acid. It consists of two potassium cations K+
and the trigonal planar trithiocarbonate dianion CS2−
3. It is a white solid, although impure samples often appear brown. It is prepared by the reaction of potassium sulfide or potassium hydrosulfide with carbon disulfide.
- K
2S + CS
2 → K
2CS
3
Potassium trithiocarbonate reacts with alkylating agents to give trithiocarbonate esters:[1]
- K
2CS
3 + 2 RX → (RS)
2CS + 2 KX (X = halogen, R = monovalent organyl group)
References
- ↑ R. E. Strube (1959). "Trithiocarbodiglycolic Acid". Organic Syntheses 39: 77. doi:10.15227/orgsyn.039.0077.
 |
