Chemistry:Praseodymium arsenide
From HandWiki
| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| PrAs | |
| Molar mass | 215.83 g/mol |
| Density | 6.6 g/cm3 |
| Related compounds | |
Other anions
|
PrN, PrP, PrSb, PrBi, Pr2O3 |
Other cations
|
CeAs, NdAs |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Praseodymium arsenide is a binary inorganic compound of praseodymium and arsenic with the formula PrAs.
Preparation
Praseodymium arsenide can be prepared by heating praseodymium and arsenic:
- [math]\displaystyle{ \mathsf{ Pr + As \ \xrightarrow{T}\ PrAs } }[/math]
Physical properties
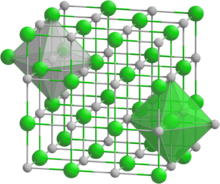
Praseodymium arsenide forms cubic crystals, space group F m3m, cell parameters a = 0.6009 nm, Z = 4, and structure like sodium chloride.[1][2][3][4] When heated, it decomposes into arsenic and Pr4As3.[2] At a pressure of 27 GPa, a phase transition to the tetragonal crystal system occurs.[5]
See also
References
- ↑ Справочник химика. 1 (2-е изд., испр ed.). М.-Л.: Химия. 1966.
- ↑ 2.0 2.1 Diagrammy sostojanija dvojnych metalličeskich sistem: spravočnik v trech tomach. 1. Moskva: Mašinostroenie. 1996. ISBN 978-5-217-02688-3.
- ↑ Gschneidner, K. A.; Calderwood, F. W. (August 1986). "The As−Pr (Arsenic-Praseodymium) system" (in en). Bulletin of Alloy Phase Diagrams 7 (4): 347–348. doi:10.1007/BF02873010. ISSN 0197-0216. http://link.springer.com/10.1007/BF02873010.
- ↑ Gschneidner, K. A.; Calderwood, F. W. (June 1986). "The arsenic-rare earth systems" (in en). Bulletin of Alloy Phase Diagrams 7 (3): 274–276. doi:10.1007/BF02869005. ISSN 0197-0216. http://link.springer.com/10.1007/BF02869005.
- ↑ Shirotani, Ichimin; Hayashi, Junichi; Yamanashi, Keigo; Hirano, Kouji; Adachi, Takafumi; Ishimatsu, Naoki; Shimomura, Osamu; Kikegawa, Takumi (June 2003). "X-ray study with synchrotron radiation of cerium and praseodymium monopnictides with the NaCl-type structure at high pressures" (in en). Physica B: Condensed Matter 334 (1–2): 167–174. doi:10.1016/S0921-4526(03)00042-5. Bibcode: 2003PhyB..334..167S. https://linkinghub.elsevier.com/retrieve/pii/S0921452603000425.
 |

