Chemistry:Pyrosilicic acid
From HandWiki
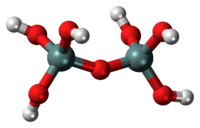
Short description: Silicic acid with the formula (HO)3–Si–O–Si–(OH)3
| |
| Names | |
|---|---|
| IUPAC name
Trihydroxysilyl trihydrogen orthosilicate
| |
| Other names
disilicic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| 26937 | |
PubChem CID
|
|
| |
| |
| Properties | |
| H6O7Si2 | |
| Molar mass | 174.211 g·mol−1 |
| Conjugate base | Pyrosilicate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Pyrosilicic acid is the chemical compound with formula H
6Si
2O
7 or (HO)
3SiOSi(OH)
3. It is one of the silicic acids and has pyrosilicate as its conjugate base.
It was synthesized, using nonaqueous solutions, in 2017.[1]
Pyrosilicic acid may be present in sea water and other natural waters at very low concentration.[2][3] Compounds formally derived from it, such as sodium pyrosilicate, are found in the sorosilicate minerals.
References
- ↑ Igarashi, Masayasu; Matsumoto, Tomohiro; Yagahashi, Fujio; Yamashita, Hiroshi; Ohhara, Takashi; Hanashima, Takayasu; Nakao, Akiko; Moyosh, Taketo et al. (2017). "Non-aqueous selective synthesis of orthosilicic acid and its oligomers". Nature Communications 8 (1): 140. doi:10.1038/s41467-017-00168-5. PMID 28747652. Bibcode: 2017NatCo...8..140I.
- ↑ Katsumi Goto (1956): "Effect of pH on Polymerization of Silicic Acid". Journal of Physical Chemistry, volume 60, issue 7, pages 1007–1008. doi:10.1021/j150541a046
- ↑ M. F. Bechtold (1955): "Polymerization and Properties of Dilute Aqueous Silicic Acid from Cation Exchange". Journal of Physical Chemistry, volume 59, issue 6, pages 532–541. doi:10.1021/j150528a013
 |
