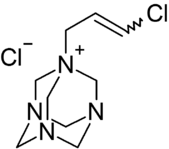
Chemistry:Quaternium-15
| |
| Names | |
|---|---|
| IUPAC name
1-(3-Chloroallyl)-3,5,7-triaza-1-azoniaadamantane chloride
| |
Other names
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| C9H16Cl2N4 | |
| Molar mass | 251.16 g·mol−1 |
| Hazards | |
| Safety data sheet | Sigma Aldrich[1] |
| GHS pictograms |    
|
| H228, H302, H315, H317, H361, H411[1] | |
| P210, P273, P280[1] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Quaternium-15 (systematic name: hexamethylenetetramine chloroallyl chloride) is a quaternary ammonium salt that has been used as a surfactant and preservative. It acts as an antimicrobial agent because it slowly releases formaldehyde, which is a preservative with biocidal properties.
Both quaternium-15 and formaldehyde release agents have been the subjects of controversy. They are often banned in US and Europe.[2][3]
It can be found under a variety of names, including Dow Chemical Company: Dowicil 200 (cis isomer only), Dowicil 75 and Dowicil 100 (both a mix of cis and trans isomers).
Synthesis
Quaternium-15 can be prepared by treating hexamethylenetetramine with 1,3-dichloropropene. A mixture of cis and trans isomers are produced.[citation needed]
Applications
The isolated cis-compound is used primarily in cosmetic applications, with a maximum permitted concentration in the EU of 0.2%. The mixed product (cis- and trans-) is used in a wider range of formulations such as: emulsifiable metal-cutting fluids, latex and emulsion paints, liquid floor polishes and floor waxes, and glues and adhesives.
Safety concerns
Quaternium-15 has been banned in the EU since 2017 and a bill was introduced in the US in 2017 to require the FDA to investigate its safety.[4][5]
Allergic reaction
Quaternium-15 is an allergen, and can cause dermatitis.[6] Many of those with an allergy to quaternium-15 are also allergic to formaldehyde. At low pHs, it would be expected to release significant amounts of formaldehyde due to acid hydrolysis via the Delepine reaction. Allergic sensitivity to quaternium-15 can be detected using a patch test.[7] It is the single most often found cause of allergic contact dermatitis of the hands (16.5% in 959 cases).[8] In 2005–06, it was the fourth-most-prevalent allergen in patch tests (10.3%).[9]
Although quaternium-15 releases low amounts of formaldehyde.[10] Even so, Johnson & Johnson announced plans to phase out its use of quaternium-15 in cosmetic products by 2015 in response to consumer pressure.[11][12]
See also
References
- ↑ 1.0 1.1 1.2 Sigma-Aldrich Co., 1-(cis-3-Chloroallyl)-3,5,7-triaza-1-azoniaadamantane chloride. Retrieved on 2014-10-07.
- ↑ de Groot, Anton C.; White, Ian R.; Flyvholm, Mari-Ann; Lensen, Gerda; Coenraads, Pieter-Jan (2010). "Formaldehyde-releasers in cosmetics: relationship to formaldehyde contact allergy". Contact Dermatitis 62 (1): 2–17. doi:10.1111/j.1600-0536.2009.01615.x. PMID 20136875.
- ↑ De Groot, Anton; Geier, Johannes; Flyvholm, Mari-Ann; Lensen, Gerda; Coenraads, Pieter-Jan (22 June 2010). "Formaldehyde-releasers: Relationship to formaldehyde contact allergy, Part 2: Metalworking fluids and remainder". Contact Dermatitis 63 (3): 129–139. doi:10.1111/j.1600-0536.2010.01715.x. PMID 20573163.
- ↑ "European Commission notifies bans, restrictions on CMRS in cosmetics". https://chemicalwatch.com/57303/european-commission-notifies-bans-restrictions-on-cmrs-in-cosmetics#overlay-strip.
- ↑ "The cosmetics industry has avoided strict regulation for over a century. Now rising health concerns has FDA inquiring". https://www.cnbc.com/2018/08/01/fda-begins-first-inquiry-of-lightly-regulated-cosmetics-industry.html.
- ↑ Cahill J, Nixon R. Allergic contact dermatitis to quaternium 15 in a moisturizing lotion. Australasia J Dermatol. 2005 Nov;46(4):284–5. PMID 16197434
- ↑ New Zealand Dermatological Society. "Quaternium-15 contact allergy". DermNet NZ. http://dermnetnz.org/dermatitis/quaternium-allergy.html. Retrieved 2007-05-31.
- ↑ E. Warshaw, et al. "Contact dermatitis of the hands: Cross-sectional analyses of North American Contact Dermatitis Group Data, 1994–2004". Journal of the American Academy of Dermatology, Volume 57, Issue 2, pp. 301–314
- ↑ Zug, KA; Warshaw, EM; Fowler, JF Jr; Maibach, HI; Belsito, DL; Pratt, MD; Sasseville, D; Storrs, FJ et al. (2009). "Patch-test results of the North American Contact Dermatitis Group 2005–2006". Dermatitis 20 (3): 149–60. doi:10.2310/6620.2009.08097. PMID 19470301.
- ↑ "Formaldehyde". http://www.cancer.org/cancer/cancercauses/othercarcinogens/intheworkplace/formaldehyde.
- ↑ "Johnson & Johnson to phase out potentially harmful chemicals by 2015". http://www.cbsnews.com/news/johnson-johnson-to-phase-out-potentially-harmful-chemicals-by-2015/.
- ↑ Thomas, Katie (17 January 2014). "The 'No More Tears' Shampoo, Now With No Formaldehyde". https://www.nytimes.com/2014/01/18/business/johnson-johnson-takes-first-step-in-removal-of-questionable-chemicals-from-products.html.
External links
 |
