Chemistry:Rabacfosadine
 | |
| Clinical data | |
|---|---|
| Trade names | Tanovea |
| AHFS/Drugs.com | Veterinary Use |
| License data | |
| Routes of administration | Intravenous |
| ATCvet code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
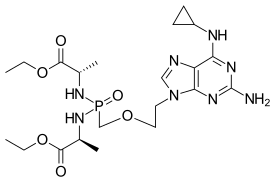
| Formula | C11H35N8O6P |
| Molar mass | 406.425 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Rabacfosadine, sold under the brand name Tanovea, is a guanine nucleotide analog used for the treatment of lymphoma in dogs. The drug was granted conditional approval by the U.S. Food and Drug Administration under application number 141-475 for use in treating canine lymphoma in December 2016[2] pending a full demonstration of effectiveness, and became the first drug to receive full approval for the treatment of canine lymphoma in July 2021.[3]
Originally developed by Gilead Sciences as GS-9219,[4] rabacfosadine is no longer being pursued for use in the treatment of lymphoma in humans.
The active form of rabacfosadine is a chain-terminating inhibitor of the major deoxyribonucleic acid (DNA) polymerases. In vitro studies have demonstrated that rabacfosadine inhibits DNA synthesis, resulting in S phase arrest and induction of apoptosis.[5] It also inhibits the proliferation of lymphocytes in dogs with naturally occurring lymphoma.[6]
Veterinary uses
In July 2021, the U.S. Food and Drug Administration (FDA) approved Tanovea to treat lymphoma in dogs.[7] Lymphoma, also called lymphosarcoma, is a type of cancer that can affect many species, including dogs.[7] Tanovea is the first conditionally approved new animal drug for dogs to achieve the FDA's full approval.[7]
Adverse effects
Common side effects of rabacfosadine are decreased white blood cell count, diarrhea, vomiting, decreased appetite or loss of appetite, weight loss, decreased activity level, and skin problems. Other side effects may occur.[2]
References
- ↑ "Tanovea-CA1- rabacfosadine succinate injection, powder, for solution". https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=407d42d7-2e23-4e90-aff0-b8b05d226e4b.
- ↑ 2.0 2.1 "FDA Conditionally Approves Tanovea-CA1, the first New Animal Drug for Treating Lymphoma in Dogs". U.S. Food and Drug Administration (FDA). 3 January 2017. https://www.fda.gov/animalveterinary/newsevents/cvmupdates/ucm535190.htm.
- ↑ Commissioner, Office of the (15 July 2021). "FDA Grants First Full Approval for Treatment of Lymphoma in Dogs". https://www.fda.gov/news-events/press-announcements/fda-grants-first-full-approval-treatment-lymphoma-dogs.
- ↑ "Tanovea® for the treatment of lymphoma in dogs". Biochemical Pharmacology 154: 265–269. August 2018. doi:10.1016/j.bcp.2018.05.010. PMID 29778492.
- ↑ "GS-9219--a novel acyclic nucleotide analogue with potent antineoplastic activity in dogs with spontaneous non-Hodgkin's lymphoma". Clinical Cancer Research 14 (9): 2824–32. May 2008. doi:10.1158/1078-0432.CCR-07-2061. PMID 18451250.
- ↑ "Use of 3'-deoxy-3'-[18F]fluorothymidine PET/CT for evaluating response to cytotoxic chemotherapy in dogs with non-Hodgkin's lymphoma". Veterinary Radiology & Ultrasound 50 (6): 660–8. 2009. doi:10.1111/j.1740-8261.2009.01612.x. PMID 19999354.
- ↑ 7.0 7.1 7.2 "FDA Grants First Full Approval for Treatment of Lymphoma in Dogs". U.S. Food and Drug Administration (FDA) (Press release). 15 July 2021. Retrieved 27 July 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
External links
- "Rabacfosadine". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/rabacfosadine.
 |
