Chemistry:SN-38
SN-38 is an antineoplastic drug. It is the active metabolite of irinotecan (an analog of camptothecin - a topoisomerase I inhibitor) but has 1000 times more activity than irinotecan itself. In vitro cytotoxicity assays show that the potency of SN-38 relative to irinotecan varies from 2- to 2000-fold.[1]
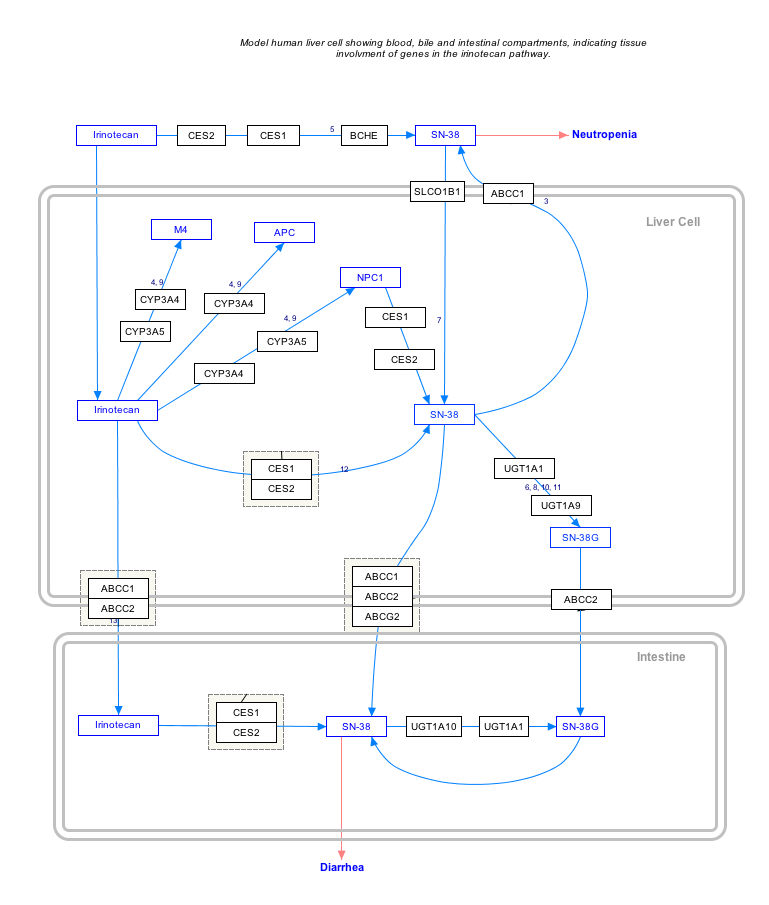
SN38 is formed via hydrolysis of irinotecan by carboxylesterases and metabolized via glucuronidation by UGT1A1.
The variant of UGT1A1 in ~10% of Caucasians which leads to poor metabolism of SN-38 predicts irinotecan toxicity, as it is then less easily excreted from the body in its SN-38 glucuronide form.[2]
SN-38 and its glucuronide are lost into the bile and intestines. It can cause the symptoms of diarrhoea and myelosuppression experienced by ~25% of the patients administered irinotecan.
Use in antibody-drug conjugates
SN-38 has been utilized as the cytotoxic payload in the antibody-drug conjugate (ADC) sacituzumab govitecan. Unlike ADCs that employ ultra-toxic payloads with picomolar IC50 values, SN-38 is considered a moderately toxic agent with IC50 in the nanomolar range, potentially allowing for a lower off-target toxicity profile.[3]
In sacituzumab govitecan, SN-38 is conjugated to an anti-Trop2 antibody via a cleavable CL2A linker, achieving a high drug-to-antibody ratio (DAR) of approximately 7.6. This high DAR allows for efficient delivery of the chemotherapeutic agent to tumor cells. The cleavable linker also facilitates the release of SN-38 in both the extracellular space and the cytoplasm. Once released, SN-38's membrane permeability enables it to diffuse out of target cancer cells and exert cytotoxic effects on neighboring cells regardless of their Trop2 expression status, a phenomenon known as the "bystander killing effect".[3]
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ↑ The interactive pathway map can be edited at WikiPathways: "IrinotecanPathway_WP46359". http://www.wikipathways.org/index.php/Pathway:WP46359.
See also
- NK012, a nanodevice formulation of SN-38
- Sacituzumab govitecan, an antibody-drug conjugate that uses SN-38 as the cytotoxic drug.
References
- ↑ "CAMPTOSAR- irinotecan hydrochloride injection, solution Pharmacia & Upjohn Company LLC". http://labeling.pfizer.com/ShowLabeling.aspx?id=533.
- ↑ O'Dwyer PJ, Catalano RB (October 2006). "Uridine diphosphate glucuronosyltransferase (UGT) 1A1 and irinotecan: practical pharmacogenomics arrives in cancer therapy". J. Clin. Oncol. 24 (28): 4534–8. doi:10.1200/JCO.2006.07.3031. PMID 17008691.
- ↑ 3.0 3.1 "Shedding light on triple-negative breast cancer with Trop2-targeted antibody-drug conjugates". American Journal of Cancer Research 12 (4): 1671–1685. April 2022. PMID 35530295.
 |