Chemistry:Sodium hexachloroosmate
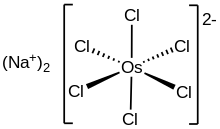 Two-dimensional representation of disodium hexachloroosmate salt
| |
 A solution of sodium hexachloroosmate
| |
| Identifiers | |
|---|---|
| |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| Cl6Na2Os | |
| Molar mass | 448.91 g·mol−1 |
| Appearance | red solid |
| Density | 3.221 g/cm3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Sodium hexachloroosmate is the inorganic compound with the formula Na
2OsCl
6. A red solid, it is the disodium salt of the osmium(IV) complex [OsCl
6]2−. The anion is an octahedral complex with Os-Cl distance of 2.325(3) Å, as established by X-ray crystallography.[1] The compound can be prepared by reaction of a suspension of osmium metal in molten sodium chloride with chlorine:[2]
- Os + 2 NaCl + 2 Cl
2 → Na
2OsCl
6
Hexachloroosmate is paramagnetic, with a low-spin d4 configuration.
Reactions
Sodium hexachloroosmate reacts with a solution of ammonium chloride to produce ammonium hexachloroosmate(IV). Upon exposure to strong alkali, hexachloroosmates will decompose to osmium dioxide. They react with excess ammonia to form so-called osmium diammine hydroxide, OsO(NH
3)
2(OH)
2 [3][4]
Sodium hexachloroosmate reacts with triphenylphosphine to form various complexes. When a solution of sodium hexachloroosmate and formalin is added to a boiling solution of triphenylphosphine in 2-methoxyethanol, OsHCl(CO)(PPh
3)
3 is formed.[5]
- Na
2OsCl
6•6H
2O + 3(C
6H
5)
3P formalin———→ OsHCl(CO)[P(C
6H
5)
3]
3
With an aqueous solution containing potassium hydroxide and formaldehyde they react to produce OsH
2(CO)(PPh
3)
3.[5]
- Na
2OsCl
6•6H
2O + 3(C
6H
5)
3P koh hcho———→ OsH
2(CO)[P(C
6H
5)
3]
3
With ethanol and sodium borohydride they react to produce OsH
4[P(C
6H
5)
3]
3.[5]
- Na
2OsCl
6•6H
2O + 3(C
6H
5)
3P borohydride————→ OsH
4[P(C
6H
5)
3]
3
With 2-methoxyethanol and n-methyl-n-nitrosotoluene sulfonamide they react to yield OsCl
3(NO)[P(C
6H
5)
3]
2.[5]
- Na
2OsCl
6•6H
2O + 2(C
6H
5)
3P + p-TolSO
2N(NO)(CH
3) → OsCl
3(NO)[P(C
6H
5)
3]
2.
References
- ↑ Rudnitskaya, O. V.; Kultyshkina, E. K.; Dobrokhotova, E. V.; Tereshina, T. A.; Popova, A. S.; Zubavichus, Ya. V.; Khrustalev, V. N. (2019). "Crystal Structure of Na2[OsCl6]". Journal of Structural Chemistry 60 (7): 1086–1090. doi:10.1134/S0022476619070096.
- ↑ H. L. Grube (1963). "Sodium Hexachloroosmate(VI)". in G. Brauer. Handbook of Preparative Inorganic Chemistry, 2nd Ed.. 2pages=1602. NY, NY: Academic Press.
- ↑ Prakash Satya (2013). "Platinum Metals-IV:Osmium". Advanced Chemistry of Rare Elements, 5th Ed.. India: S Chand and Company Limited. pp. 710.
- ↑ J. Newton Friend (1920). "Osmium and its compounds". A textbook of inorganic chemistry, vol.IX Part I Cobalt, Nickel, and The Elements of The Platinum Group. London: Charles Griffin and Company, Limited. pp. 216. https://dn790003.ca.archive.org/0/items/textbookinorgani00libg_516/textbookinorgani00libg_516.pdf. Retrieved 18 July 2025.
- ↑ 5.0 5.1 5.2 5.3 Parshall, George (1974). Inorganic Syntheses. 15. McGraw-Hill Book Company. pp. 53-58. https://sites.lsa.umich.edu/jbuss/wp-content/uploads/sites/811/2020/08/inorganic-synthesis15.pdf. Retrieved 17 July 2025.
 |
