Chemistry:Stictic acid
From HandWiki
| |
| |
| Names | |
|---|---|
| Systematic IUPAC name
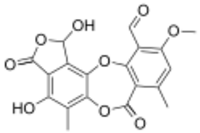
1,4-Dihydroxy-10-methoxy-5,8-dimethyl-3,7-dioxo-1,3-dihydro-7H-2,6,12-trioxabenzo[5,6]cyclohepta[1,2-e]indene-11-carbaldehyde | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C19H14O9 | |
| Molar mass | 386.312 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Stictic acid is an aromatic organic compound, a product of secondary metabolism in some species of lichens.[1]
Stictic acid is the subject of preliminary biomedical research. Stictic acid has cytotoxic and apoptotic effects in vitro.[2] Computational studies suggest stictic acid may also stimulate reactivation of mutant p53.[3]
See also
- Chemistry:Constictic acid
- Chemistry:Hypostictic acid – Lichen-derived depsidone
- Chemistry:Norstictic acid
References
- ↑ Lohézic-Le Dévéhat, Françoise; Tomasi, Sophie; Elix, John A.; Bernard, Aurélie; Rouaud, Isabelle; Uriac, Philippe; Boustie, Joël (2007). "Stictic Acid Derivatives from the Lichen Usnea articulata and Their Antioxidant Activities". Journal of Natural Products 70 (7): 1218–20. doi:10.1021/np070145k. PMID 17629329. Bibcode: 2007JNAtP..70.1218L.
- ↑ Correché, ER; Enriz, RD; Piovano, M; Garbarino, J; Gómez-Lechón, MJ (2004). "Cytotoxic and apoptotic effects on hepatocytes of secondary metabolites obtained from lichens". Alternatives to Laboratory Animals 32 (6): 605–15. doi:10.1177/026119290403200611. PMID 15757498.
- ↑ Wassman, Christopher D.; Baronio, Roberta; Demir, Özlem; Wallentine, Brad D.; Chen, Chiung-Kuang; Hall, Linda V.; Salehi, Faezeh; Lin, Da-Wei et al. (2013). "Computational identification of a transiently open L1/S3 pocket for reactivation of mutant p53". Nature Communications 4. doi:10.1038/ncomms2361. PMID 23360998. Bibcode: 2013NatCo...4.1407W.
 |
