Biology:P53
p53, also known as tumor protein p53, TP53, cellular tumor antigen p53 (UniProt name), or transformation-related protein 53 (TRP53) is a regulatory transcription factor protein that is often mutated in human cancers. The p53 proteins (originally thought to be, and often spoken of as, a single protein) are crucial in vertebrates, where they prevent cancer formation.[1] As such, p53 has been described as "the guardian of the genome" because of its role in conserving stability by preventing genome mutation.[2] Hence TP53[note 1] is classified as a tumor suppressor gene.[3][4][5][6][7]
The TP53 gene is the most frequently mutated gene (>50%) in human cancer, indicating that the TP53 gene plays a crucial role in preventing cancer formation.[1] TP53 gene encodes proteins that bind to DNA and regulate gene expression to prevent mutations of the genome.[8] In addition to the full-length protein, the human TP53 gene encodes at least 12 protein isoforms.[9]
Comparative genomic studies found pathogenic mutations absent in some Neanderthal populations, while modern humans exhibit a expansion of over 1,000 mutated variations.[10] Evidence suggests that the vast majority of these protein-coding variants arose very recently in human history, specifically concentrated within a window of 5,000 to 10,000 years ago.[11][12]
Gene
In humans, the TP53 gene is located on the short arm of chromosome 17 (17p13.1).[3][4][5][6] The gene spans 20 kb, with a non-coding exon 1 and a very long first intron of 10 kb, overlapping the Hp53int1 gene. The coding sequence contains five regions showing a high degree of conservation in vertebrates, predominantly in exons 2, 5, 6, 7 and 8, but the sequences found in invertebrates show only distant resemblance to mammalian TP53.[13] TP53 orthologs[14] have been identified in most mammals for which complete genome data are available. Elephants, with 20 genes for TP53, rarely get cancer.[15]
Structure

The full-length p53 protein (p53α) comprises seven distinct protein domains:
- An acidic N-terminus transactivation domain (TAD), including activation domains 1 and 2 (AD1: residues 1–42; AD2: residues 43–63), which regulate transcription of several pro-apoptotic genes.[16]
- A proline-rich domain (residues 64–92), involved in apoptotic function and nuclear export via MAPK signaling.
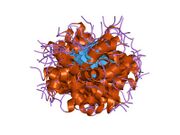
- A central DNA-binding domain (DBD; residues 102–292), containing a zinc atom and multiple arginine residues, essential for sequence-specific DNA interaction and co-repressor binding such as LMO3.[17]
- A nuclear localization sequence (NLS; residues 316–325), required for nuclear import.
- A homo-oligomerization domain (OD; residues 307–355), which mediates tetramerization—essential for p53 activity in vivo.
- A C-terminal regulatory domain (residues 356–393), which modulates the DNA-binding activity of the central domain.[18]
Most cancer-associated mutations in TP53 occur in the DBD, impairing DNA binding and transcriptional activation. These are typically recessive loss-of-function mutations. By contrast, mutations in the OD can exert dominant negative effects by forming inactive complexes with wild-type p53.
Wild-type p53 is a labile protein containing both folded and intrinsically disordered regions that act synergistically.[19]
Although designated as a 53 kDa protein by SDS-PAGE, the actual molecular weight of p53α is 43.7 kDa. The discrepancy is due to its high proline content, which slows electrophoretic migration.[20]
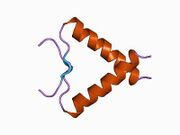
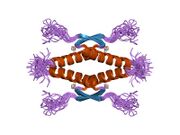
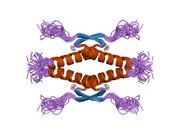
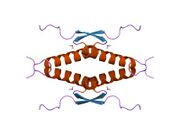
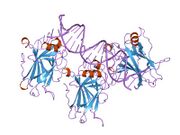
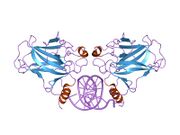
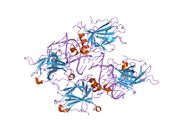
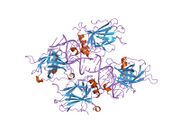
Tetramerization
p53 initially forms dimers cotranslationally during protein synthesis on ribosomes.[21] Each dimer consists of two p53 monomers joined through their oligomerization domains.[22]
The dimerization interface spans residues 325–356 and includes a beta-strand (residues 325–333), a alpha-helix (residues 335–356), and a sharp turn at the conserved hinge residue Gly334. This configuration links the beta-strand and alpha-helix to form a V-shaped monomer topology. The beta-strand contributes to the formation of an antiparallel intermolecular beta-sheet between two p53 monomers, stabilized by hydrophobic interactions involving Phe328, Leu330, and Ile332. The alpha-helix forms an antiparallel coiled-coil between the two monomers, with a packing angle of 156°. Helix–helix interactions are stabilized by hydrophobic contacts (e.g., Phe338, Phe341, Leu344) and electrostatic interactions, such as the Arg337–Asp352 salt bridge.
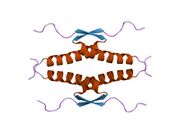
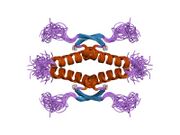
Following dimer formation, p53 dimers associate posttranslationally to form tetramers (dimers of dimers).[21][23] The tetramerization domain (residues 325–356) plays a central role in stabilizing the tetrameric structure.[23] In the tetramer, the two primary dimers associate at an angle described as "roughly orthogonal," with a helix bundle packing angle (θ) of approximately 80°.
Tetramers represent the active form of p53 for DNA binding and transcriptional regulation.[24][22]
Isoforms
Like 95% of human genes, TP53 encodes multiple proteins, collectively known as the p53 isoforms.[1] These vary in size from 3.5 to 43.7 kDa. Since their initial discovery in 2005, 12 human p53 isoforms have been identified: p53α, p53β, p53γ, ∆40p53α, ∆40p53β, ∆40p53γ, ∆133p53α, ∆133p53β, ∆133p53γ, ∆160p53α, ∆160p53β, and ∆160p53γ. Isoform expression is tissue-dependent, and p53α is never expressed alone.[7]
The isoforms differ by the inclusion or exclusion of specific domains. Some, such as Δ133p53β/γ and Δ160p53α/β/γ, lack the transactivation or proline-rich domains and are deficient in apoptosis induction, illustrating the functional diversity of TP53.[25][26]
Isoforms are generated through multiple mechanisms:
- Alternative splicing of intron 9 creates the β and γ isoforms with altered C-termini.
- An internal promoter in intron 4 produces the ∆133 and ∆160 isoforms, which lack part of the TAD and DBD.
- Alternative translation initiation at codons 40 or 160 results in ∆40p53 and ∆160p53 isoforms, respectively.[7]
Function
DNA damage and repair

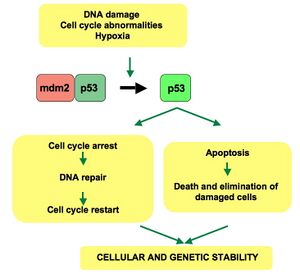
p53 regulates cell cycle progression, apoptosis, and genomic stability through multiple mechanisms:
- Activates DNA repair proteins in response to DNA damage,[27] suggesting a potential role in aging.[28]
- Arrests the cell cycle at the G1/S checkpoint upon DNA damage, allowing time for repair before progression.
- Initiates apoptosis if the damage is beyond repair.
- Essential for the senescence response triggered by short telomeres.
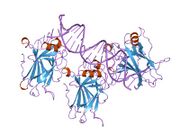
p53 functions as a transcription factor by binding DNA as a tetramer, a structure that is essential for its stability and effective DNA binding activity.[29] Once bound to DNA, p53 induces the transcription of numerous genes involved in DNA repair pathways. This includes components of base excision repair (BER) such as OGG1 and MUTYH, nucleotide excision repair (NER) factors like DDB2 and XPC, mismatch repair (MMR) genes such as MSH2 and MLH1, and elements of homologous recombination (HR) and non-homologous end-joining (NHEJ) repair.[30][31] These transcriptional responses are crucial for the DNA damage response (DDR), allowing cells to efficiently repair damaged DNA and maintain genomic integrity. While p53's role is most clearly defined in transcriptional activation of repair genes, it also participates in non-transcriptional regulation of DNA repair processes, particularly in HR and NHEJ, by modulating protein interactions and chromatin accessibility.[30][32]
p53 binds specific elements in the promoter of target genes, including CDKN1A, which encodes p21.[29][33] Upon activation by p53, p21 inhibits cyclin-dependent kinases, leading to cell cycle arrest and contributing to tumor suppression.[29][34] However, p21 can also be induced independently of p53 during processes such as differentiation, development, and in response to serum stimulation.[33]
p21 (WAF1) binds to cyclin-CDK complexes (notably CDK2, CDK1, CDK4, and CDK6), inhibiting their activity and blocking the G1/S transition.[35][36] This inhibition enforces a cell cycle pause that allows DNA repair to occur. In cells with functional p53, p21 is upregulated in response to DNA damage, ensuring this checkpoint control. In contrast, p53 mutations impair p21 induction and compromise this control.[29]
In human embryonic stem cells (hESCs), although p21 mRNA is upregulated following DNA damage, the protein is not detectable. This reflects a nonfunctional p53-p21 axis at the G1/S checkpoint.[37] This discrepancy is largely due to post-transcriptional repression, particularly by the miR-302 family of microRNAs, which inhibit p21 translation.[38] Although p53 binds the CDKN1A promoter in hESCs, it does not regulate miR-302, which is constitutively expressed and suppresses p21 expression.[38][37]
The p53 pathway is interconnected with the RB1 pathway via p14^ARF, which links the regulation of these key tumor suppressors.[39]
p53 expression can be induced by UV radiation, which also causes DNA damage. In this context, p53 activation can initiate processes that lead to melanin production and tanning.[40][41]
Stem cells
Levels of p53 play an important role in the maintenance of stem cells throughout development and the rest of human life.[42]
In human embryonic stem cells (hESCs)s, p53 is maintained at low inactive levels.[43] This is because activation of p53 leads to rapid differentiation of hESCs.[44] Studies have shown that knocking out p53 delays differentiation and that adding p53 causes spontaneous differentiation, showing how p53 promotes differentiation of hESCs and plays a key role in cell cycle as a differentiation regulator. When p53 becomes stabilized and activated in hESCs, it increases p21 to establish a longer G1. This typically leads to abolition of S-phase entry, which stops the cell cycle in G1, leading to differentiation. Work in mouse embryonic stem cells has recently shown however that the expression of P53 does not necessarily lead to differentiation.[45] p53 also activates miR-34a and miR-145, which then repress the hESCs pluripotency factors, further instigating differentiation.[43]
In adult stem cells, p53 regulation is important for maintenance of stemness in adult stem cell niches. Mechanical signals such as hypoxia affect levels of p53 in these niche cells through the hypoxia inducible factors, HIF-1α and HIF-2α. While HIF-1α stabilizes p53, HIF-2α suppresses it.[46] Suppression of p53 plays important roles in cancer stem cell phenotype, induced pluripotent stem cells and other stem cell roles and behaviors, such as blastema formation. Cells with decreased levels of p53 have been shown to reprogram into stem cells with a much greater efficiency than normal cells.[47][48] Papers suggest that the lack of cell cycle arrest and apoptosis gives more cells the chance to be reprogrammed. Decreased levels of p53 were also shown to be a crucial aspect of blastema formation in the legs of salamanders.[49] p53 regulation is very important in acting as a barrier between stem cells and a differentiated stem cell state, as well as a barrier between stem cells being functional and being cancerous.[50]
Other

Apart from the cellular and molecular effects above, p53 has a tissue-level anticancer effect that works by inhibiting angiogenesis.[51] As tumors grow they need to recruit new blood vessels to supply them, and p53 inhibits that by (i) interfering with regulators of tumor hypoxia that also affect angiogenesis, such as HIF1 and HIF2, (ii) inhibiting the production of angiogenic promoting factors, and (iii) directly increasing the production of angiogenesis inhibitors, such as arresten.[52][53]
p53 by regulating Leukemia Inhibitory Factor has been shown to facilitate implantation in the mouse and possibly human reproduction.[54]
The immune response to infection also involves p53 and NF-κB. Checkpoint control of the cell cycle and of apoptosis by p53 is inhibited by some infections such as Mycoplasma bacteria,[55] raising the specter of oncogenic infection.
Regulation
Basal regulation
Under normal, unstressed conditions, p53 is maintained at low levels through continuous degradation mediated by the E3 ubiquitin ligase MDM2 (HDM2 in humans).[56] MDM2 binds p53, exports it from the nucleus, and targets it for proteasomal degradation. Notably, p53 transcriptionally activates MDM2, establishing a classic negative feedback loop.
This feedback loop gives rise to damped oscillations in p53 levels, as demonstrated both experimentally[57] and in mathematical models.[58][59] These oscillations may determine cell fate decisions between survival and apoptosis.[60]
Activation by cellular stress
p53 is activated in response to a range of cellular stressors, including DNA damage (for example from ultraviolet or ionizing radiation), osmotic shock, ribonucleotide depletion, oncogene activation, and some viral infections.[61][62]
Activation involves stabilization of the p53 protein, resulting in its accumulation in the nucleus, and regulatory changes that promote sequence-specific DNA binding and transcriptional activation of target genes.[62][63] These processes are initiated in part by phosphorylation of residues in the N-terminal transactivation domain by stress-activated kinases.[62][63] Phosphorylation of sites within the Mdm2-binding region (for example Ser20) can reduce binding to MDM2 and thereby decrease ubiquitin-mediated degradation of p53.[64][65]
Stress-responsive kinases
Kinases that regulate p53 phosphorylation can be divided into two broad groups. One group includes members of the MAPK pathways, including JNK1–3, ERK1/2 and p38 MAPK, which are activated by diverse cellular stresses such as oxidative stress and heat shock.[66] A second group comprises DNA damage response kinases, including ATM, ATR and DNA-PK, together with downstream checkpoint kinases such as CHK1 and CHK2, which are activated by DNA damage and replication stress and contribute to p53 regulation through phosphorylation-dependent signalling.[67][68]
Additional kinases implicated in p53 phosphorylation include the CDK-activating kinase (CAK; CDK7–cyclin H–MAT1), which has been shown to phosphorylate p53 (for example at Ser33) in vitro and in vivo,[69] and TP53RK (PRPK), which has been reported to phosphorylate p53 at Ser15.[70]
Oncogene-induced activation of p53 can also occur via p14ARF (ARF), which inhibits the p53 antagonist MDM2 and thereby stabilizes p53.[71][72]
Deubiquitination
Several deubiquitinating enzymes (DUBs) modulate p53 stability by removing ubiquitin chains. USP7, also known as HAUSP, can deubiquitinate both p53 and MDM2. In unstressed cells, HAUSP preferentially stabilizes MDM2, and its depletion may paradoxically increase p53 levels. USP42 is another DUB that stabilizes p53 and enhances its ability to respond to stress.[73] USP10 operates primarily in the cytoplasm, where it counteracts MDM2 by directly deubiquitinating p53. After DNA damage, USP10 translocates to the nucleus and further stabilizes p53. It does not interact with MDM2.[74]
Post-translational modifications and cofactors
Phosphorylation of the N-terminus not only prevents MDM2 binding but also facilitates the recruitment of cofactors. Pin1 enhances conformational changes in p53, while p300 and PCAF acetylate the C-terminus, exposing the DNA-binding domain and enhancing transcriptional activation. Conversely, deacetylases such as Sirt1 and Sirt7 remove these modifications, suppressing apoptosis and promoting cell survival.[75] Some oncogenes can also activate p53 indirectly by inhibiting MDM2.[76]
Dynamics
Both experimental evidence and mathematical modeling indicate that p53 levels oscillate over time in response to cellular signals. These oscillations become more pronounced in the presence of DNA damage, such as double-stranded breaks or UV exposure. Modeling approaches also help illustrate how mutations in p53 isoforms affect oscillatory behavior, potentially informing tissue-specific therapeutic development.[77][78][58]
Epigenetics
p53 function is also influenced by chromatin environment. The corepressor TRIM24 restricts p53 binding to epigenetically repressed loci by recognizing methylated histones. This interaction enables p53 to interpret local chromatin context and regulate gene expression in a locus-specific manner.[79] [80]
Role in disease
The TP53 gene can also be altered by mutagens—such as chemicals, radiation, or certain viruses—thereby increasing the likelihood of uncontrolled cell division. More than 50 percent of human tumors harbor a mutation or deletion of the TP53 gene.[81] Loss of p53 function leads to genomic instability, frequently resulting in an aneuploidy phenotype.[82]
Certain pathogens can also disrupt p53 activity. For example, human papillomavirus (HPV) produces the viral protein E6, which binds to and inactivates p53. In conjunction with the HPV protein E7, which inactivates the cell cycle regulator pRb, this promotes repeated cell division, clinically presenting as warts. High-risk HPV types, particularly types 16 and 18, can drive the progression from benign warts to low- or high-grade cervical dysplasia, reversible precancerous lesions. Persistent cervical infection can lead to irreversible changes, including carcinoma in situ and invasive cervical cancer. These outcomes are primarily driven by viral integration into the host genome and the continued expression of the E6 and E7 oncoproteins.[83]
Mutations
Most p53 mutations are detected by DNA sequencing. However, it is known that single missense mutations can have a large spectrum from rather mild to very severe functional effects.[78]

The large spectrum of cancer phenotypes due to mutations in the TP53 gene is also supported by the fact that different isoforms of p53 proteins have different cellular mechanisms for prevention against cancer. Mutations in TP53 can give rise to different isoforms, preventing their overall functionality in different cellular mechanisms and thereby extending the cancer phenotype from mild to severe. Recent studies show that p53 isoforms are differentially expressed in different human tissues, and the loss-of-function or gain-of-function mutations within the isoforms can cause tissue-specific cancer or provide cancer stem cell potential in different tissues.[7][26][85][86] TP53 mutation also hits energy metabolism and increases glycolysis in breast cancer cells.[87]
Codon 72 variations
A common human polymorphism in TP53 involves a substitution of arginine for proline at codon 72 of exon 4. Numerous studies have explored the relationship between this variation and cancer susceptibility, yielding mixed results. For instance, a 2009 meta-analysis found no association between the codon 72 polymorphism and cervical cancer risk.[88]
Other studies have identified possible associations between the codon 72 polymorphism and various cancers. A 2011 study reported that the proline variant significantly increased pancreatic cancer risk in males.[89] Another study found that proline homozygosity was associated with decreased breast cancer risk in Arab women.[90] Additional research suggested that TP53 codon 72 polymorphisms, in combination with MDM2 SNP309 and A2164G, may affect susceptibility and age of onset for non-oropharyngeal cancers in women.[91] A separate 2011 study linked the polymorphism to an increased risk of lung cancer in a Korean population.[92]
However, meta-analyses published in 2011 found no significant associations between the codon 72 variant and risks of either colorectal[93] or endometrial cancer.[94] A study of a Brazilian birth cohort found an association between the arginine variant and individuals without a family history of cancer.[95] Meanwhile, another study reported that individuals with the homozygous Pro/Pro genotype had a significantly increased risk of renal cell carcinoma.[96]
Therapeutic reactivation and gene therapy
While increasing p53 levels might appear beneficial for treating cancer, sustained p53 activation can cause premature aging.[97] A more promising approach involves restoring normal, endogenous p53 function. In some tumor types, this leads to regression via apoptosis or normalization of cell growth.[98][99]
The first commercial gene therapy, Gendicine, was approved in China in 2003 for head and neck squamous cell carcinoma. It delivers a functional copy of the TP53 gene using a modified adenovirus.[100]
The small-molecule inhibitor MI-63 can bind to MDM2, blocking its interaction with p53 and reactivating p53 in cancers where its function is suppressed.[101]
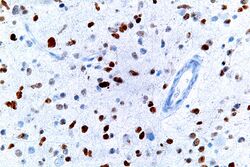
Diagnostic and prognostic significance
 This image shows different patterns of p53 expression in endometrial cancers on chromogenic immunohistochemistry, whereof all except wild-type are variably termed abnormal/aberrant/mutation-type and are strongly predictive of an underlying TP53 mutation:[102]
|

Discovery
p53 was identified in 1979 by Lionel Crawford, David P. Lane, Arnold Levine, and Lloyd Old, working at Imperial Cancer Research Fund (UK), Princeton University/UMDNJ (Cancer Institute of New Jersey), and Memorial Sloan Kettering Cancer Center, respectively. It had been hypothesized to exist before as the target of the SV40 virus, a strain that induced development of tumors. The name p53 is in fact a misnomer, as it describes the apparent molecular mass measured when it was first discovered, though it was later realised this was an overestimate: the correct molecular mass is only 43.7 kDa.[105]
The TP53 gene from the mouse was first cloned by Peter Chumakov of the Academy of Sciences of the USSR in 1982,[106] and independently in 1983 by Moshe Oren in collaboration with David Givol (Weizmann Institute of Science).[107][108] The human TP53 gene was cloned in 1984[3] and the full length clone in 1985.[109]
It was initially presumed to be an oncogene due to the use of mutated cDNA following purification of tumor cell mRNA. Its role as a tumor suppressor gene was revealed in 1989 by Bert Vogelstein at the Johns Hopkins School of Medicine and Arnold Levine at Princeton University.[110][111] p53 went on to be identified as a transcription factor by Guillermina Lozano working at MD Anderson Cancer Center.[112]
Warren Maltzman, of the Waksman Institute of Rutgers University first demonstrated that TP53 was responsive to DNA damage in the form of ultraviolet radiation.[113] In a series of publications in 1991–92, Michael Kastan of Johns Hopkins University, reported that TP53 was a critical part of a signal transduction pathway that helped cells respond to DNA damage.[114]
In 1993, p53 was voted molecule of the year by Science magazine.[115]
Interactions
p53 has been shown to interact with:
- AIMP2,[116]
- ANKRD2,[117]
- APTX,[118]
- ATM,[119][120][121][122][123]
- ATR,[119][120]
- ATF3,[124][125]
- AURKA,[126]
- BAK1,[127]
- BARD1,[128]
- BLM,[129][130][131][132]
- BRCA1,[128][133][134][135][136]
- BRCA2,[128][137]
- BRCC3,[128]
- BRE,[128]
- CEBPZ,[138]
- CDC14A,[139]
- Cdk1,[140][141]
- CFLAR,[142]
- CHEK1,[129][143][144]
- CCNG1,[145]
- CREBBP,[146][147][148]
- CREB1,[148]
- Cyclin H,[149]
- CDK7,[149][150]
- DNA-PKcs,[120][143][151]
- E4F1,[152][153]
- EFEMP2,[154]
- EIF2AK2,[155]
- ELL,[156]
- EP300,[147][157][158][159]
- ERCC6,[160][161]
- GNL3,[162]
- GPS2,[163]
- GSK3B,[164]
- HSP90AA1,[165][166][167]
- HIF1A,[168][169][170][171]
- HIPK1,[172]
- HIPK2,[173][174]
- HMGB1,[175][176]
- HSPA9,[177]
- Huntingtin,[178]
- ING1,[179][180]
- ING4,[181][182]
- ING5,[181]
- IκBα,[183]
- KPNB1,[165]
- LMO3,[17]
- Mdm2,[146][184][185][186]
- MDM4,[187][188]
- MED1,[189][190]
- MAPK9,[191][192]
- MNAT1,[150]
- NDN,[193]
- NCL,[194]
- NUMB,[195]
- NF-κB,[196]
- P16,[152][186][197]
- PARC,[198]
- PARP1,[118][199]
- PIAS1,[154][200]
- CDC14B,[139]
- PIN1,[201][202]
- PLAGL1,[203]
- PLK3,[204][205]
- PRKRA,[206]
- PHB,[207]
- PML,[184][208][209]
- PSME3,[210]
- PTEN,[185]
- PTK2,[211]
- PTTG1,[212]
- RAD51,[128][213][214]
- RCHY1,[215][216]
- RELA,[196]
- RPA1,[217][218]
- RPL11,[197]
- S100B,[219]
- SUMO1,[220][221]
- SMARCA4,[222]
- SMARCB1,[222]
- SMN1,[223]
- STAT3,[196]
- TBP,[224][225]
- TFAP2A,[226]
- TFDP1,[227]
- TIGAR,[228]
- TOP1,[229][230]
- TOP2A,[231]
- TP53BP1,[129][232][233][234][235][236][237]
- TP53BP2,[237][238]
- TOP2B,[231]
- TP53INP1,[239][240]
- TSG101,[241]
- UBE2A,[242]
- UBE2I,[154][220][243][244]
- UBC,[116][210][221][245][246][247][248][249]
- USP7,[250]
- USP10,[74]
- WRN,[132][251]
- WWOX,[252]
- XPB,[160]
- YBX1,[117][253]
- YPEL3,[254]
- YWHAZ,[255]
- Zif268,[256]
- ZNF148,[257]
- SIRT1,[258]
- circRNA_014511.[259]
See also
- Eprenetapopt, a reactivator of some mutant forms of p53
- Pifithrin, an inhibitor of p53
Notes
References
- ↑ 1.0 1.1 1.2 "Uncovering the role of p53 splice variants in human malignancy: a clinical perspective". OncoTargets and Therapy 7: 57–68. December 2013. doi:10.2147/OTT.S53876. PMID 24379683.
- ↑ "The Guardian of the Genome Revisited: p53 Downregulates Genes Required for Telomere Maintenance, DNA Repair, and Centromere Structure". Cancers 10 (5): 135. May 2018. doi:10.3390/cancers10050135. PMID 29734785.
- ↑ 3.0 3.1 3.2 "Isolation and characterization of a human p53 cDNA clone: expression of the human p53 gene". The EMBO Journal 3 (13): 3257–62. December 1984. doi:10.1002/j.1460-2075.1984.tb02287.x. PMID 6396087.
- ↑ 4.0 4.1 "Localization of gene for human p53 tumour antigen to band 17p13". Nature 320 (6057): 84–5. 1986. doi:10.1038/320084a0. PMID 3456488. Bibcode: 1986Natur.320...84I.
- ↑ 5.0 5.1 "Identification of p53 as a sequence-specific DNA-binding protein". Science 252 (5013): 1708–11. June 1991. doi:10.1126/science.2047879. PMID 2047879. Bibcode: 1991Sci...252.1708K.
- ↑ 6.0 6.1 "The gene for human p53 cellular tumor antigen is located on chromosome 17 short arm (17p13)". Proceedings of the National Academy of Sciences of the United States of America 83 (1): 130–4. January 1986. doi:10.1073/pnas.83.1.130. PMID 3001719. Bibcode: 1986PNAS...83..130M.
- ↑ 7.0 7.1 7.2 7.3 "p53 isoforms can regulate p53 transcriptional activity". Genes & Development 19 (18): 2122–37. September 2005. doi:10.1101/gad.1339905. PMID 16131611.
- ↑ The p53 family. Cold Spring Harbor Perspectives in Biology. Cold Spring Harbor, N.Y.: Cold Spring Harbor Laboratory Press. 2010. ISBN 978-0-87969-830-0.
- ↑ "p53 Isoforms: An Intracellular Microprocessor?". Genes Cancer 2 (4): 453–65. April 2011. doi:10.1177/1947601911408893. PMID 21779513.
- ↑ "Pathogenic variation in human DNA damage repair genes was originated from the evolutionary process of modern humans". Genes & Diseases. November 2025. doi:10.1016/j.gendis.2025.101916.
- ↑ "Analysis of 6,515 exomes reveals the recent origin of most human protein-coding variants". Nature 493 (7431): 216–220. January 2013. doi:10.1038/nature11690. PMID 23201682. PMC 3676746. Bibcode: 2013Natur.493..216F. http://nrs.harvard.edu/urn-3:HUL.InstRepos:11717639.
- ↑ "Pathogenic variants in human DNA damage repair genes mostly arose in recent human history". BMC Cancer 24 (1). April 2024. doi:10.1186/s12885-024-12160-6. PMID 38575974.
- ↑ "Twenty years of p53 research: structural and functional aspects of the p53 protein". Oncogene 18 (53): 7621–36. December 1999. doi:10.1038/sj.onc.1203285. PMID 10618702.
- ↑ "OrthoMaM phylogenetic marker: TP53 coding sequence". http://www.orthomam.univ-montp2.fr/orthomam/data/cds/detailMarkers/ENSG00000141510_TP53.xml.
- ↑ "TP53 copy number expansion is associated with the evolution of increased body size and an enhanced DNA damage response in elephants". eLife 5. September 2016. doi:10.7554/eLife.11994. PMID 27642012. Bibcode: 2016eLife...511994S.
- ↑ "The requirement for the p53 proline-rich functional domain for mediation of apoptosis is correlated with specific PIG3 gene transactivation and with transcriptional repression". The EMBO Journal 17 (16): 4668–79. August 1998. doi:10.1093/emboj/17.16.4668. PMID 9707426.
- ↑ 17.0 17.1 "LMO3 interacts with p53 and inhibits its transcriptional activity". Biochemical and Biophysical Research Communications 392 (3): 252–7. February 2010. doi:10.1016/j.bbrc.2009.12.010. PMID 19995558. Bibcode: 2010BBRC..392..252L.
- ↑ "The C terminus of p53 family proteins is a cell fate determinant". Molecular and Cellular Biology 25 (5): 2014–30. March 2005. doi:10.1128/MCB.25.5.2014-2030.2005. PMID 15713654.
- ↑ "p53 contains large unstructured regions in its native state". Journal of Molecular Biology 322 (5): 917–27. October 2002. doi:10.1016/S0022-2836(02)00848-3. PMID 12367518.
- ↑ "Cell-free translations of proline-rich protein mRNAs". The Journal of Biological Chemistry 257 (18): 11176–80. September 1982. doi:10.1016/S0021-9258(18)33948-6. PMID 7107651.
- ↑ 21.0 21.1 "Biogenesis of p53 involves cotranslational dimerization of monomers and posttranslational dimerization of dimers. Implications on the dominant negative effect". The Journal of Biological Chemistry 277 (15): 12937–12945. April 2002. doi:10.1074/jbc.M108815200. PMID 11805092.
- ↑ 22.0 22.1 "TIMELESS-dependent positive and negative autoregulation in the Drosophila circadian clock". The EMBO Journal 18 (3): 675–686. February 1999. doi:10.1093/emboj/18.3.675. PMID 9927427.
- ↑ 23.0 23.1 "Ultraslow oligomerization equilibria of p53 and its implications". Proceedings of the National Academy of Sciences of the United States of America 106 (34): 14327–14332. August 2009. doi:10.1073/pnas.0907840106. PMID 19667193. Bibcode: 2009PNAS..10614327N.
- ↑ "Structure of the p53 core domain dimer bound to DNA". The Journal of Biological Chemistry 281 (29): 20494–20502. July 2006. doi:10.1074/jbc.M603634200. PMID 16717092.
- ↑ "Definition of the p53 functional domains necessary for inducing apoptosis". The Journal of Biological Chemistry 275 (51): 39927–34. December 2000. doi:10.1074/jbc.M005676200. PMID 10982799.
- ↑ 26.0 26.1 "p53 Isoforms: An Intracellular Microprocessor?". Genes & Cancer 2 (4): 453–65. April 2011. doi:10.1177/1947601911408893. PMID 21779513.
- ↑ 27.0 27.1 "Decoding p53 tumor suppression: a crosstalk between genomic stability and epigenetic control?". Cell Death and Differentiation 32 (1): 1–8. January 2025. doi:10.1038/s41418-024-01259-9. PMID 38379088.
 This article incorporates text from this source, which is available under the CC BY 4.0 license.
This article incorporates text from this source, which is available under the CC BY 4.0 license.
- ↑ Developmental Biology, 10th ed.. Sunderland, MA USA: Sinauer Associates, Inc. Publishers. p. 588.
- ↑ 29.0 29.1 29.2 29.3 "Cell cycle regulation: p53-p21-RB signaling". Cell Death and Differentiation 29 (5): 946–960. May 2022. doi:10.1038/s41418-022-00988-z. PMID 35361964.
- ↑ 30.0 30.1 "p53 in the DNA-Damage-Repair Process". Cold Spring Harbor Perspectives in Medicine 6 (5). May 2016. doi:10.1101/cshperspect.a026070. PMID 27048304.
- ↑ "p53 and regulation of DNA damage recognition during nucleotide excision repair". DNA Repair 2 (9): 947–54. September 2003. doi:10.1016/s1568-7864(03)00087-9. PMID 12967652.
- ↑ "p53 in recombination and repair". Cell Death and Differentiation 13 (6): 1003–16. June 2006. doi:10.1038/sj.cdd.4401903. PMID 16543940.
- ↑ 33.0 33.1 "Examination of the expanding pathways for the regulation of p21 expression and activity". Cellular Signalling 22 (7): 1003–12. July 2010. doi:10.1016/j.cellsig.2010.01.013. PMID 20100570.
- ↑ "Mechanisms of transcriptional regulation by p53". Cell Death and Differentiation 25 (1): 133–143. January 2018. doi:10.1038/cdd.2017.174. PMID 29125602.
- ↑ "The Role of the Cyclin Dependent Kinase Inhibitor p21cip1/waf1 in Targeting Cancer: Molecular Mechanisms and Novel Therapeutics". Cancers 11 (10): 1475. September 2019. doi:10.3390/cancers11101475. PMID 31575057.
- ↑ "Multiple functions of p21 in cell cycle, apoptosis and transcriptional regulation after DNA damage". DNA Repair 42: 63–71. June 2016. doi:10.1016/j.dnarep.2016.04.008. PMID 27156098.
- ↑ 37.0 37.1 "An Updated View of the Roles of p53 in Embryonic Stem Cells". Stem Cells 40 (10): 883–891. October 2022. doi:10.1093/stmcls/sxac051. PMID 35904997.
- ↑ 38.0 38.1 "MicroRNAs regulate p21(Waf1/Cip1) protein expression and the DNA damage response in human embryonic stem cells". Stem Cells 30 (7): 1362–72. July 2012. doi:10.1002/stem.1108. PMID 22511267.
- ↑ "p14ARF links the tumour suppressors RB and p53". Nature 395 (6698): 124–5. September 1998. doi:10.1038/25867. PMID 9744267. Bibcode: 1998Natur.395..124B.
- ↑ "Genome's guardian gets a tan started". New Scientist. March 17, 2007. https://www.newscientist.com/channel/health/mg19325955.800-genomes-guardian-gets-a-tan-started.html. Retrieved 2007-03-29.
- ↑ "Central role of p53 in the suntan response and pathologic hyperpigmentation". Cell 128 (5): 853–64. March 2007. doi:10.1016/j.cell.2006.12.045. PMID 17350573. Bibcode: 2007Cell..128..853C.
- ↑ "Functions of p53 in pluripotent stem cells". Oxford Academic 11 (1): 71–78. 2020. doi:10.1007/s13238-019-00665-x. PMID 31691903.
- ↑ 43.0 43.1 "p53 regulates cell cycle and microRNAs to promote differentiation of human embryonic stem cells". PLOS Biology 10 (2). 2012. doi:10.1371/journal.pbio.1001268. PMID 22389628.
- ↑ "Activation of p53 by nutlin leads to rapid differentiation of human embryonic stem cells". Oncogene 27 (40): 5277–87. September 2008. doi:10.1038/onc.2008.166. PMID 18521083.
- ↑ "Critical role for P53 in regulating the cell cycle of ground state embryonic stem cells". Stem Cell Reports 14 (2): 175–183. February 2020. doi:10.1016/j.stemcr.2020.01.001. PMID 32004494.
- ↑ "HIF-2α suppresses p53 to enhance the stemness and regenerative potential of human embryonic stem cells". Stem Cells 30 (8): 1685–95. August 2012. doi:10.1002/stem.1142. PMID 22689594.
- ↑ "Context-dependent enhancement of induced pluripotent stem cell reprogramming by silencing Puma". Stem Cells 30 (5): 888–97. May 2012. doi:10.1002/stem.1054. PMID 22311782.
- ↑ "A p53-mediated DNA damage response limits reprogramming to ensure iPS cell genomic integrity". Nature 460 (7259): 1149–53. August 2009. doi:10.1038/nature08287. PMID 19668189. Bibcode: 2009Natur.460.1149M.
- ↑ "Regulation of p53 is critical for vertebrate limb regeneration". Proceedings of the National Academy of Sciences of the United States of America 110 (43): 17392–7. October 2013. doi:10.1073/pnas.1310519110. PMID 24101460. Bibcode: 2013PNAS..11017392Y.
- ↑ "p53: the barrier to cancer stem cell formation". FEBS Letters 588 (16): 2580–9. August 2014. doi:10.1016/j.febslet.2014.02.011. PMID 24560790. Bibcode: 2014FEBSL.588.2580A.
- ↑ 51.0 51.1 "Crosslink between p53 and metastasis: focus on epithelial-mesenchymal transition, cancer stem cell, angiogenesis, autophagy, and anoikis". Molecular Biology Reports 48 (11): 7545–7557. November 2021. doi:10.1007/s11033-021-06706-1. PMID 34519942.
- ↑ "Inhibition of tumor angiogenesis by p53: a new role for the guardian of the genome". Journal of Molecular Medicine 85 (11): 1175–1186. November 2007. doi:10.1007/s00109-007-0221-2. PMID 17589818.
- ↑ "p53 inhibits angiogenesis by inducing the production of Arresten". Cancer Research 72 (5): 1270–1279. March 2012. doi:10.1158/0008-5472.CAN-11-2348. PMID 22253229.
- ↑ "p53 regulates maternal reproduction through LIF". Nature 450 (7170): 721–4. November 2007. doi:10.1038/nature05993. PMID 18046411. Bibcode: 2007Natur.450..721H.
- ↑ "Effects of mycoplasma infection on the host organism response via p53/NF-κB signaling". Journal of Cellular Physiology 234 (1): 171–180. January 2018. doi:10.1002/jcp.26781. PMID 30146800.
- ↑ "Targeting mutant p53 for efficient cancer therapy". Nature Reviews. Cancer 18 (2): 89–102. February 2018. doi:10.1038/nrc.2017.109. PMID 29242642.
- ↑ "Oscillations and variability in the p53 system". Molecular Systems Biology 2. June 2006. doi:10.1038/msb4100068. PMID 16773083.
- ↑ 58.0 58.1 "Explaining oscillations and variability in the p53-Mdm2 system". BMC Systems Biology 2 (75). August 2008. doi:10.1186/1752-0509-2-75. PMID 18706112.
- ↑ "Mathematical modelling of p53 basal dynamics and DNA damage response". C-fACS 259 (20th International Congress on Mathematical Modelling and Simulation): 670–6. December 2013. doi:10.1016/j.mbs.2014.10.010. PMID 25433195.
- ↑ "p53 dynamics control cell fate". Science 336 (6087): 1440–1444. June 2012. doi:10.1126/science.1218351. PMID 22700930. Bibcode: 2012Sci...336.1440P.
- ↑ "Dial 9-1-1 for p53: mechanisms of p53 activation by cellular stress". Neoplasia 2 (3): 208–225. 2000. doi:10.1038/sj.neo.7900080. PMID 10935507.
- ↑ 62.0 62.1 62.2 "Post-translational modifications and activation of p53 by genotoxic stresses". European Journal of Biochemistry 268 (10): 2764–2772. May 2001. doi:10.1046/j.1432-1327.2001.02225.x. PMID 11358490. Bibcode: 2001EJBio.268.2764A.
- ↑ 63.0 63.1 "The complexity of p53 stabilization and activation". Cell Death and Differentiation 13 (6): 941–950. June 2006. doi:10.1038/sj.cdd.4401925. PMID 16601750.
- ↑ "Phosphorylation of Ser-20 mediates stabilization of human p53 in response to DNA damage". Proceedings of the National Academy of Sciences of the United States of America 96 (24): 13777–13782. November 1999. doi:10.1073/pnas.96.24.13777. PMID 10570149. Bibcode: 1999PNAS...9613777C.
- ↑ "p53 ubiquitination: Mdm2 and beyond". Molecular Cell 21 (3): 307–315. February 2006. doi:10.1016/j.molcel.2006.01.020. PMID 16455486.
- ↑ "Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases". Microbiology and Molecular Biology Reviews 75 (1): 50–83. March 2011. doi:10.1128/MMBR.00031-10. PMID 21372320.
- ↑ "ATM, ATR, and DNA-PK: The Trinity at the Heart of the DNA Damage Response". Molecular Cell 66 (6): 801–817. June 2017. doi:10.1016/j.molcel.2017.05.015. PMID 28622525.
- ↑ "p53 C-terminal phosphorylation by CHK1 and CHK2 participates in the regulation of DNA-damage-induced C-terminal acetylation". Molecular Biology of the Cell 16 (4): 1684–1695. April 2005. doi:10.1016/j.molcel.2004.12.023. PMID 15659650.
- ↑ "p53 is phosphorylated by CDK7-cyclin H in a p36MAT1-dependent manner". Molecular and Cellular Biology 17 (12): 7220–7229. December 1997. doi:10.1128/MCB.17.12.7220. PMID 9372954.
- ↑ "TP53RK (TP53-regulating kinase) (Homo sapiens)". https://www.uniprot.org/uniprotkb/Q96S44/entry.
- ↑ "Escape from p53-mediated tumor surveillance in neuroblastoma: switching off the p14(ARF)-MDM2-p53 axis". Cell Death and Differentiation 16 (12): 1563–1572. December 2009. doi:10.1038/cdd.2009.138. PMID 19779493.
- ↑ "Stabilization of p53 by p14ARF without relocation of MDM2 to the nucleolus". Nature Cell Biology 3 (5): 445–452. May 2001. doi:10.1038/35074506. PMID 11331871.
- ↑ "Regulation of p53 stability and function by the deubiquitinating enzyme USP42". The EMBO Journal 30 (24): 4921–30. November 2011. doi:10.1038/emboj.2011.419. PMID 22085928.
- ↑ 74.0 74.1 "USP10 Regulates p53 Localization and Stability by Deubiquitinating p53". Cell 140 (3): 384–396. February 2010. doi:10.1016/j.cell.2009.12.032. PMID 20096447.
- ↑ "Sirt7 increases stress resistance of cardiomyocytes and prevents apoptosis and inflammatory cardiomyopathy in mice". Circulation Research 102 (6): 703–10. March 2008. doi:10.1161/CIRCRESAHA.107.164558. PMID 18239138.
- ↑ "Transcription factors that interact with p53 and Mdm2". International Journal of Cancer 138 (7): 1577–85. April 2016. doi:10.1002/ijc.29663. PMID 26132471.
- ↑ "CellLine, a stochastic cell lineage simulator". Bioinformatics 23 (24): 3409–3411. December 2007. doi:10.1093/bioinformatics/btm491. PMID 17928303.
- ↑ 78.0 78.1 "Thermodynamic stability of wild-type and mutant p53 core domain". Proceedings of the National Academy of Sciences of the United States of America 94 (26): 14338–42. December 1997. doi:10.1073/pnas.94.26.14338. PMID 9405613. Bibcode: 1997PNAS...9414338B.
- ↑ "Readout of histone methylation by Trim24 locally restricts chromatin opening by p53". Nature Structural & Molecular Biology 30 (7): 948–57. June 2023. doi:10.1038/s41594-023-01021-8. PMID 37386214.
- ↑ "Li-Fraumeni syndrome: MedlinePlus Genetics" (in en). https://medlineplus.gov/genetics/condition/li-fraumeni-syndrome/.
- ↑ "p53 mutations in human cancers". Science 253 (5015): 49–53. July 1991. doi:10.1126/science.1905840. PMID 1905840. Bibcode: 1991Sci...253...49H. https://zenodo.org/record/1230948.
- ↑ "Dissecting p53 tumor suppressor functions in vivo". Cancer Cell 1 (3): 289–98. April 2002. doi:10.1016/S1535-6108(02)00047-8. PMID 12086865.
- ↑ "The Viral Etiology of AIDS-Associated Malignancies". HIV-1: Molecular Biology and Pathogenesis. Advances in Pharmacology. 56. 2008. pp. 509–57. doi:10.1016/S1054-3589(07)56016-3. ISBN 978-0-12-373601-7.
- ↑ 84.0 84.1 "Deciphering the significance of p53 mutant proteins". Trends in Cell Biology 35 (3): 258–268. July 2024. doi:10.1016/j.tcb.2024.06.003. PMID 38960851.
 This article incorporates text from this source, which is available under the CC BY 4.0 license.
This article incorporates text from this source, which is available under the CC BY 4.0 license.
- ↑ "The relative mRNA expression of p53 isoforms in breast cancer is associated with clinical features and outcome". Carcinogenesis 35 (3): 586–96. March 2014. doi:10.1093/carcin/bgt411. PMID 24336193.
- ↑ "The p53 isoform Δ133p53β promotes cancer stem cell potential". Stem Cell Reports 4 (4): 531–40. April 2015. doi:10.1016/j.stemcr.2015.02.001. PMID 25754205.
- ↑ "TP53 mutation hits energy metabolism and increases glycolysis in breast cancer". Oncotarget 7 (41): 67183–67195. October 2016. doi:10.18632/oncotarget.11594. PMID 27582538.
- ↑ "TP53 codon 72 polymorphism and cervical cancer: a pooled analysis of individual data from 49 studies". The Lancet. Oncology 10 (8): 772–84. August 2009. doi:10.1016/S1470-2045(09)70187-1. PMID 19625214.
- ↑ "TP53 codon 72 polymorphism is associated with pancreatic cancer risk in males, smokers and drinkers". Molecular Medicine Reports 4 (3): 489–95. 2011. doi:10.3892/mmr.2011.449. PMID 21468597.
- ↑ "P53 gene polymorphisms and breast cancer risk in Arab women". Medical Oncology 28 (3): 709–15. September 2011. doi:10.1007/s12032-010-9505-4. PMID 20443084.
- ↑ "Effects of MDM2 promoter polymorphisms and p53 codon 72 polymorphism on risk and age at onset of squamous cell carcinoma of the head and neck". Molecular Carcinogenesis 50 (9): 697–706. September 2011. doi:10.1002/mc.20806. PMID 21656578.
- ↑ "p53 codon 72 polymorphism and the risk of lung cancer in a Korean population". Lung Cancer 73 (3): 264–7. September 2011. doi:10.1016/j.lungcan.2010.12.017. PMID 21316118.
- ↑ "TP53 codon 72 polymorphism and colorectal cancer susceptibility: a meta-analysis". Molecular Biology Reports 38 (8): 4847–53. November 2011. doi:10.1007/s11033-010-0619-8. PMID 21140221.
- ↑ "TP53 Arg72Pro polymorphism and endometrial cancer risk: a meta-analysis". Medical Oncology 28 (4): 1129–35. December 2011. doi:10.1007/s12032-010-9597-x. PMID 20552298.
- ↑ "TP53 gene polymorphism: importance to cancer, ethnicity and birth weight in a Brazilian cohort". Journal of Biosciences 36 (5): 823–31. December 2011. doi:10.1007/s12038-011-9147-5. PMID 22116280.
- ↑ "The polymorphisms of P53 codon 72 and MDM2 SNP309 and renal cell carcinoma risk in a low arsenic exposure area". Toxicology and Applied Pharmacology 257 (3): 349–55. December 2011. doi:10.1016/j.taap.2011.09.018. PMID 21982800. Bibcode: 2011ToxAP.257..349H.
- ↑ "p53 mutant mice that display early ageing-associated phenotypes". Nature 415 (6867): 45–53. January 2002. doi:10.1038/415045a. PMID 11780111. Bibcode: 2002Natur.415...45T.
- ↑ "Restoration of p53 function leads to tumour regression in vivo". Nature 445 (7128): 661–5. February 2007. doi:10.1038/nature05541. PMID 17251932.
- ↑ "Visualization and targeted disruption of protein interactions in living cells". Nature Communications 4. 2013. doi:10.1038/ncomms3660. PMID 24154492. Bibcode: 2013NatCo...4.2660H.
- ↑ "China approves first gene therapy". Nature Biotechnology 22 (1): 3–4. January 2004. doi:10.1038/nbt0104-3. PMID 14704685.
- ↑ "MI-63: a novel small-molecule inhibitor targets MDM2 and induces apoptosis in embryonal and alveolar rhabdomyosarcoma cells with wild-type p53". British Journal of Cancer 101 (5): 774–81. September 2009. doi:10.1038/sj.bjc.6605199. PMID 19707204.
- ↑ "Interpretation of P53 Immunohistochemistry in Endometrial Carcinomas: Toward Increased Reproducibility". International Journal of Gynecological Pathology 38 (Suppl 1): S123–S131. January 2019. doi:10.1097/PGP.0000000000000488. PMID 29517499.
 This article incorporates text available under the CC BY 4.0 license.
This article incorporates text available under the CC BY 4.0 license.
- ↑ Image is taken from following source, with some modification by Mikael Häggström, MD:
- "Altered p53/p16 expression is linked to urothelial carcinoma progression but largely unrelated to prognosis in muscle-invasive tumors.". Acta Oncol 62 (12): 1880–1889. 2023. doi:10.1080/0284186X.2023.2277344. PMID 37938166. - ↑ Kalantari MR, Ahmadnia H (2007). "P53 overexpression in bladder urothelial neoplasms: new aspect of World Health Organization/International Society of Urological Pathology classification.". Urol J 4 (4): 230–3. PMID 18270948. https://journals.sbmu.ac.ir/urolj/index.php/uj/article/view/103/102.
- ↑ "The first 30 years of p53: growing ever more complex". Nature Reviews. Cancer 9 (10): 749–758. October 2009. doi:10.1038/nrc2723. PMID 19776744. Bibcode: 2009NatRC...9..749L.
- ↑ "[Isolation of a plasmid clone containing the mRNA sequence for mouse nonviral T-antigen]" (in ru). Doklady Akademii Nauk SSSR 267 (5): 1272–5. 1982. PMID 6295732.
- ↑ "Molecular cloning of a cDNA specific for the murine p53 cellular tumor antigen". Proceedings of the National Academy of Sciences of the United States of America 80 (1): 56–9. January 1983. doi:10.1073/pnas.80.1.56. PMID 6296874. Bibcode: 1983PNAS...80...56O.
- ↑ "A single gene and a pseudogene for the cellular tumour antigen p53". Nature 306 (5943): 594–7. 1983. doi:10.1038/306594a0. PMID 6646235. Bibcode: 1983Natur.306..594Z.
- ↑ "Human p53 cellular tumor antigen: cDNA sequence and expression in COS cells". The EMBO Journal 4 (5): 1251–5. May 1985. doi:10.1002/j.1460-2075.1985.tb03768.x. PMID 4006916.
- ↑ "Chromosome 17 deletions and p53 gene mutations in colorectal carcinomas". Science 244 (4901): 217–21. April 1989. doi:10.1126/science.2649981. PMID 2649981. Bibcode: 1989Sci...244..217B.
- ↑ "The p53 proto-oncogene can act as a suppressor of transformation". Cell 57 (7): 1083–93. June 1989. doi:10.1016/0092-8674(89)90045-7. PMID 2525423.
- ↑ "Transcriptional activation by wild-type but not transforming mutants of the p53 anti-oncogene". Science 249 (4972): 1049–1051. August 1990. doi:10.1126/science.2144364. PMID 2144364. Bibcode: 1990Sci...249.1049R.
- ↑ "UV irradiation stimulates levels of p53 cellular tumor antigen in nontransformed mouse cells". Molecular and Cellular Biology 4 (9): 1689–94. September 1984. doi:10.1128/mcb.4.9.1689. PMID 6092932.
- ↑ "Control of G1 arrest after DNA damage". Environmental Health Perspectives 101 (Suppl 5): 55–8. December 1993. doi:10.2307/3431842. PMID 8013425.
- ↑ "Molecule of the year". Science 262 (5142): 1953. December 1993. doi:10.1126/science.8266084. PMID 8266084. Bibcode: 1993Sci...262.1953K.
- ↑ 116.0 116.1 "AIMP2/p38, the scaffold for the multi-tRNA synthetase complex, responds to genotoxic stresses via p53". Proceedings of the National Academy of Sciences of the United States of America 105 (32): 11206–11. August 2008. doi:10.1073/pnas.0800297105. PMID 18695251. Bibcode: 2008PNAS..10511206H.
- ↑ 117.0 117.1 "The Ankrd2 protein, a link between the sarcomere and the nucleus in skeletal muscle". Journal of Molecular Biology 339 (2): 313–25. May 2004. doi:10.1016/j.jmb.2004.03.071. PMID 15136035.
- ↑ 118.0 118.1 "Aprataxin, a novel protein that protects against genotoxic stress". Human Molecular Genetics 13 (10): 1081–93. May 2004. doi:10.1093/hmg/ddh122. PMID 15044383.
- ↑ 119.0 119.1 "BRCA1-BARD1 complexes are required for p53Ser-15 phosphorylation and a G1/S arrest following ionizing radiation-induced DNA damage". The Journal of Biological Chemistry 279 (30): 31251–8. July 2004. doi:10.1074/jbc.M405372200. PMID 15159397.
- ↑ 120.0 120.1 120.2 "Substrate specificities and identification of putative substrates of ATM kinase family members". The Journal of Biological Chemistry 274 (53): 37538–43. December 1999. doi:10.1074/jbc.274.53.37538. PMID 10608806.
- ↑ "Functional interaction of H2AX, NBS1, and p53 in ATM-dependent DNA damage responses and tumor suppression". Molecular and Cellular Biology 25 (2): 661–70. January 2005. doi:10.1128/MCB.25.2.661-670.2005. PMID 15632067.
- ↑ "ATM associates with and phosphorylates p53: mapping the region of interaction". Nature Genetics 20 (4): 398–400. December 1998. doi:10.1038/3882. PMID 9843217.
- ↑ "Genetic interactions between atm and p53 influence cellular proliferation and irradiation-induced cell cycle checkpoints". Cancer Research 57 (9): 1664–7. May 1997. PMID 9135004.
- ↑ "A human protein-protein interaction network: a resource for annotating the proteome". Cell 122 (6): 957–68. September 2005. doi:10.1016/j.cell.2005.08.029. PMID 16169070. Bibcode: 2005Cell..122..957S.
- ↑ "ATF3 represses 72-kDa type IV collagenase (MMP-2) expression by antagonizing p53-dependent trans-activation of the collagenase promoter". The Journal of Biological Chemistry 277 (13): 10804–12. March 2002. doi:10.1074/jbc.M112069200. PMID 11792711.
- ↑ "Suppression of the STK15 oncogenic activity requires a transactivation-independent p53 function". The EMBO Journal 21 (17): 4491–9. September 2002. doi:10.1093/emboj/cdf409. PMID 12198151.
- ↑ "Mitochondrial p53 activates Bak and causes disruption of a Bak-Mcl1 complex". Nature Cell Biology 6 (5): 443–50. May 2004. doi:10.1038/ncb1123. PMID 15077116.
- ↑ 128.0 128.1 128.2 128.3 128.4 128.5 "Regulation of BRCC, a holoenzyme complex containing BRCA1 and BRCA2, by a signalosome-like subunit and its role in DNA repair". Molecular Cell 12 (5): 1087–99. November 2003. doi:10.1016/S1097-2765(03)00424-6. PMID 14636569.
- ↑ 129.0 129.1 129.2 "Functional interaction between BLM helicase and 53BP1 in a Chk1-mediated pathway during S-phase arrest". The Journal of Cell Biology 166 (6): 801–13. September 2004. doi:10.1083/jcb.200405128. PMID 15364958.
- ↑ "Functional interaction of p53 and BLM DNA helicase in apoptosis". The Journal of Biological Chemistry 276 (35): 32948–55. August 2001. doi:10.1074/jbc.M103298200. PMID 11399766.
- ↑ "The Bloom syndrome protein interacts and cooperates with p53 in regulation of transcription and cell growth control". Oncogene 20 (57): 8276–80. December 2001. doi:10.1038/sj.onc.1205120. PMID 11781842.
- ↑ 132.0 132.1 "The processing of Holliday junctions by BLM and WRN helicases is regulated by p53". The Journal of Biological Chemistry 277 (35): 31980–7. August 2002. doi:10.1074/jbc.M204111200. PMID 12080066.
- ↑ "Functional and physical interactions between BRCA1 and p53 in transcriptional regulation of the IGF-IR gene". Hormone and Metabolic Research 35 (11–12): 758–62. 2003. doi:10.1055/s-2004-814154. PMID 14710355.
- ↑ "BRCA1 regulates p53-dependent gene expression". Proceedings of the National Academy of Sciences of the United States of America 95 (5): 2302–6. March 1998. doi:10.1073/pnas.95.5.2302. PMID 9482880. Bibcode: 1998PNAS...95.2302O.
- ↑ "The second BRCT domain of BRCA1 proteins interacts with p53 and stimulates transcription from the p21WAF1/CIP1 promoter". Oncogene 18 (1): 263–8. January 1999. doi:10.1038/sj.onc.1202323. PMID 9926942.
- ↑ "BRCA1 physically associates with p53 and stimulates its transcriptional activity". Oncogene 16 (13): 1713–21. April 1998. doi:10.1038/sj.onc.1201932. PMID 9582019.
- ↑ "The BRCA2 gene product functionally interacts with p53 and RAD51". Proceedings of the National Academy of Sciences of the United States of America 95 (23): 13869–74. November 1998. doi:10.1073/pnas.95.23.13869. PMID 9811893. Bibcode: 1998PNAS...9513869M.
- ↑ "Physical interaction of tumour suppressor p53/p73 with CCAAT-binding transcription factor 2 (CTF2) and differential regulation of human high-mobility group 1 (HMG1) gene expression". The Biochemical Journal 371 (Pt 2): 301–10. April 2003. doi:10.1042/BJ20021646. PMID 12534345.
- ↑ 139.0 139.1 "The human Cdc14 phosphatases interact with and dephosphorylate the tumor suppressor protein p53". The Journal of Biological Chemistry 275 (4): 2410–4. January 2000. doi:10.1074/jbc.275.4.2410. PMID 10644693.
- ↑ "The C-terminal regulatory domain of p53 contains a functional docking site for cyclin A". Journal of Molecular Biology 300 (3): 503–18. July 2000. doi:10.1006/jmbi.2000.3830. PMID 10884347.
- ↑ "Downregulation of the cdc2/cyclin B protein kinase activity by binding of p53 to p34(cdc2)". Biochemical and Biophysical Research Communications 283 (2): 507–12. May 2001. doi:10.1006/bbrc.2001.4792. PMID 11327730.
- ↑ "Cisplatin induces p53-dependent FLICE-like inhibitory protein ubiquitination in ovarian cancer cells". Cancer Research 68 (12): 4511–7. June 2008. doi:10.1158/0008-5472.CAN-08-0673. PMID 18559494.
- ↑ 143.0 143.1 "Regulatory interactions between the checkpoint kinase Chk1 and the proteins of the DNA-dependent protein kinase complex". The Journal of Biological Chemistry 278 (32): 29940–7. August 2003. doi:10.1074/jbc.M301765200. PMID 12756247.
- ↑ "Radiation-induced phosphorylation of Chk1 at S345 is associated with p53-dependent cell cycle arrest pathways". Neoplasia 4 (2): 171–80. 2002. doi:10.1038/sj.neo.7900219. PMID 11896572.
- ↑ "Cyclin G1 has growth inhibitory activity linked to the ARF-Mdm2-p53 and pRb tumor suppressor pathways". Molecular Cancer Research 1 (3): 195–206. January 2003. PMID 12556559.
- ↑ 146.0 146.1 "MDM2-HDAC1-mediated deacetylation of p53 is required for its degradation". The EMBO Journal 21 (22): 6236–45. November 2002. doi:10.1093/emboj/cdf616. PMID 12426395.
- ↑ 147.0 147.1 "p53 Transcriptional activity is mediated through the SRC1-interacting domain of CBP/p300". The Journal of Biological Chemistry 277 (11): 9054–61. March 2002. doi:10.1074/jbc.M108870200. PMID 11782467.
- ↑ 148.0 148.1 "p53 recruitment of CREB binding protein mediated through phosphorylated CREB: a novel pathway of tumor suppressor regulation". Molecular and Cellular Biology 20 (13): 4849–58. July 2000. doi:10.1128/MCB.20.13.4849-4858.2000. PMID 10848610.
- ↑ 149.0 149.1 "Regulation of CAK kinase activity by p53". Oncogene 17 (21): 2733–41. November 1998. doi:10.1038/sj.onc.1202504. PMID 9840937.
- ↑ 150.0 150.1 "p53 is phosphorylated by CDK7-cyclin H in a p36MAT1-dependent manner". Molecular and Cellular Biology 17 (12): 7220–9. December 1997. doi:10.1128/mcb.17.12.7220. PMID 9372954.
- ↑ "DNA end-independent activation of DNA-PK mediated via association with the DNA-binding protein C1D". Genes & Development 12 (14): 2188–99. July 1998. doi:10.1101/gad.12.14.2188. PMID 9679063.
- ↑ 152.0 152.1 "Association of p14ARF with the p120E4F transcriptional repressor enhances cell cycle inhibition". The Journal of Biological Chemistry 278 (7): 4981–9. February 2003. doi:10.1074/jbc.M210978200. PMID 12446718.
- ↑ "p53 is involved in the p120E4F-mediated growth arrest". Oncogene 19 (2): 188–99. January 2000. doi:10.1038/sj.onc.1203250. PMID 10644996.
- ↑ 154.0 154.1 154.2 "MBP1: a novel mutant p53-specific protein partner with oncogenic properties". Oncogene 18 (24): 3608–16. June 1999. doi:10.1038/sj.onc.1202937. PMID 10380882.
- ↑ "The double-stranded RNA activated protein kinase PKR physically associates with the tumor suppressor p53 protein and phosphorylates human p53 on serine 392 in vitro". Oncogene 18 (17): 2690–702. April 1999. doi:10.1038/sj.onc.1202620. PMID 10348343.
- ↑ "Physical interaction and functional antagonism between the RNA polymerase II elongation factor ELL and p53". The Journal of Biological Chemistry 274 (24): 17003–10. June 1999. doi:10.1074/jbc.274.24.17003. PMID 10358050.
- ↑ "p300/MDM2 complexes participate in MDM2-mediated p53 degradation". Molecular Cell 2 (4): 405–15. October 1998. doi:10.1016/S1097-2765(00)80140-9. PMID 9809062.
- ↑ "Ordered cooperative functions of PRMT1, p300, and CARM1 in transcriptional activation by p53". Cell 117 (6): 735–48. June 2004. doi:10.1016/j.cell.2004.05.009. PMID 15186775.
- ↑ "Regulation of transcription of the human presenilin-1 gene by ets transcription factors and the p53 protooncogene". The Journal of Biological Chemistry 275 (45): 34938–45. November 2000. doi:10.1074/jbc.M005411200. PMID 10942770.
- ↑ 160.0 160.1 "p53 modulation of TFIIH-associated nucleotide excision repair activity". Nature Genetics 10 (2): 188–95. June 1995. doi:10.1038/ng0695-188. PMID 7663514. http://repub.eur.nl/pub/54884.
- ↑ "Activation of p53 or loss of the Cockayne syndrome group B repair protein causes metaphase fragility of human U1, U2, and 5S genes". Molecular Cell 5 (5): 801–10. May 2000. doi:10.1016/S1097-2765(00)80320-2. PMID 10882116.
- ↑ "A nucleolar mechanism controlling cell proliferation in stem cells and cancer cells". Genes & Development 16 (23): 2991–3003. December 2002. doi:10.1101/gad.55671. PMID 12464630.
- ↑ "AMF1 (GPS2) modulates p53 transactivation". Molecular and Cellular Biology 21 (17): 5913–24. September 2001. doi:10.1128/MCB.21.17.5913-5924.2001. PMID 11486030.
- ↑ "Direct, activating interaction between glycogen synthase kinase-3beta and p53 after DNA damage". Proceedings of the National Academy of Sciences of the United States of America 99 (12): 7951–5. June 2002. doi:10.1073/pnas.122062299. PMID 12048243. Bibcode: 2002PNAS...99.7951W.
- ↑ 165.0 165.1 "A role for Hsc70 in regulating nucleocytoplasmic transport of a temperature-sensitive p53 (p53Val-135)". The Journal of Biological Chemistry 276 (18): 14649–57. May 2001. doi:10.1074/jbc.M100200200. PMID 11297531.
- ↑ "Phosphorylation and hsp90 binding mediate heat shock stabilization of p53". The Journal of Biological Chemistry 278 (3): 2066–71. January 2003. doi:10.1074/jbc.M206697200. PMID 12427754.
- ↑ "Inhibition of MDM2 by hsp90 contributes to mutant p53 stabilization". The Journal of Biological Chemistry 276 (44): 40583–90. November 2001. doi:10.1074/jbc.M102817200. PMID 11507088.
- ↑ "Direct interactions between HIF-1 alpha and Mdm2 modulate p53 function". The Journal of Biological Chemistry 278 (16): 13595–8. April 2003. doi:10.1074/jbc.C200694200. PMID 12606552.
- ↑ "Regulation of tumor angiogenesis by p53-induced degradation of hypoxia-inducible factor 1alpha". Genes & Development 14 (1): 34–44. January 2000. doi:10.1101/gad.14.1.34. PMID 10640274.
- ↑ "Two sequence motifs from HIF-1alpha bind to the DNA-binding site of p53". Proceedings of the National Academy of Sciences of the United States of America 99 (16): 10305–9. August 2002. doi:10.1073/pnas.122347199. PMID 12124396. Bibcode: 2002PNAS...9910305H.
- ↑ "Stabilization of wild-type p53 by hypoxia-inducible factor 1alpha". Nature 392 (6674): 405–8. March 1998. doi:10.1038/32925. PMID 9537326. Bibcode: 1998Natur.392..405A.
- ↑ "Characterization of cells and gene-targeted mice deficient for the p53-binding kinase homeodomain-interacting protein kinase 1 (HIPK1)". Proceedings of the National Academy of Sciences of the United States of America 100 (9): 5431–6. April 2003. doi:10.1073/pnas.0530308100. PMID 12702766. Bibcode: 2003PNAS..100.5431K.
- ↑ "Regulation of p53 activity by its interaction with homeodomain-interacting protein kinase-2". Nature Cell Biology 4 (1): 1–10. January 2002. doi:10.1038/ncb715. PMID 11740489.
- ↑ "Identification and characterization of HIPK2 interacting with p73 and modulating functions of the p53 family in vivo". The Journal of Biological Chemistry 277 (35): 32020–8. August 2002. doi:10.1074/jbc.M200153200. PMID 11925430.
- ↑ "Interaction with p53 enhances binding of cisplatin-modified DNA by high mobility group 1 protein". The Journal of Biological Chemistry 276 (10): 7534–40. March 2001. doi:10.1074/jbc.M008143200. PMID 11106654.
- ↑ "HMGB1 interacts with many apparently unrelated proteins by recognizing short amino acid sequences". The Journal of Biological Chemistry 277 (9): 7021–8. March 2002. doi:10.1074/jbc.M108417200. PMID 11748221.
- ↑ "Hsp70 family member, mot-2/mthsp70/GRP75, binds to the cytoplasmic sequestration domain of the p53 protein". Experimental Cell Research 274 (2): 246–53. April 2002. doi:10.1006/excr.2002.5468. PMID 11900485.
- ↑ "The Huntington's disease protein interacts with p53 and CREB-binding protein and represses transcription". Proceedings of the National Academy of Sciences of the United States of America 97 (12): 6763–8. June 2000. doi:10.1073/pnas.100110097. PMID 10823891. Bibcode: 2000PNAS...97.6763S.
- ↑ "The candidate tumor suppressor ING1b can stabilize p53 by disrupting the regulation of p53 by MDM2". Cancer Research 62 (17): 4890–3. September 2002. PMID 12208736.
- ↑ "The candidate tumour suppressor p33ING1 cooperates with p53 in cell growth control". Nature 391 (6664): 295–8. January 1998. doi:10.1038/34675. PMID 9440695. Bibcode: 1998Natur.391..295G.
- ↑ 181.0 181.1 "p29ING4 and p28ING5 bind to p53 and p300, and enhance p53 activity". Cancer Research 63 (10): 2373–8. May 2003. PMID 12750254.
- ↑ "Two wobble-splicing events affect ING4 protein subnuclear localization and degradation". Experimental Cell Research 314 (17): 3130–41. October 2008. doi:10.1016/j.yexcr.2008.08.002. PMID 18775696.
- ↑ "The non-ankyrin C terminus of Ikappa Balpha physically interacts with p53 in vivo and dissociates in response to apoptotic stress, hypoxia, DNA damage, and transforming growth factor-beta 1-mediated growth suppression". The Journal of Biological Chemistry 277 (12): 10323–31. March 2002. doi:10.1074/jbc.M106607200. PMID 11799106.
- ↑ 184.0 184.1 "Cellular stress and DNA damage invoke temporally distinct Mdm2, p53 and PML complexes and damage-specific nuclear relocalization". Journal of Cell Science 116 (Pt 19): 3917–25. October 2003. doi:10.1242/jcs.00714. PMID 12915590.
- ↑ 185.0 185.1 "PTEN tumor suppressor regulates p53 protein levels and activity through phosphatase-dependent and -independent mechanisms". Cancer Cell 3 (2): 117–30. February 2003. doi:10.1016/S1535-6108(03)00021-7. PMID 12620407.
- ↑ 186.0 186.1 "ARF promotes MDM2 degradation and stabilizes p53: ARF-INK4a locus deletion impairs both the Rb and p53 tumor suppression pathways". Cell 92 (6): 725–34. March 1998. doi:10.1016/S0092-8674(00)81401-4. PMID 9529249.
- ↑ "MdmX is a RING finger ubiquitin ligase capable of synergistically enhancing Mdm2 ubiquitination". The Journal of Biological Chemistry 277 (51): 49668–75. December 2002. doi:10.1074/jbc.M208593200. PMID 12393902.
- ↑ "Isolation and identification of the human homolog of a new p53-binding protein, Mdmx". Genomics 43 (1): 34–42. July 1997. doi:10.1006/geno.1997.4775. PMID 9226370. https://repository.ubn.ru.nl/bitstream/2066/142231/1/142231.pdf.
- ↑ "RB18A, whose gene is localized on chromosome 17q12-q21.1, regulates in vivo p53 transactivating activity". Cancer Research 60 (23): 6585–9. December 2000. PMID 11118038.
- ↑ "Identification of RB18A, a 205 kDa new p53 regulatory protein which shares antigenic and functional properties with p53". Oncogene 15 (25): 3013–24. December 1997. doi:10.1038/sj.onc.1201492. PMID 9444950.
- ↑ "JNK1, JNK2 and JNK3 are p53 N-terminal serine 34 kinases". Oncogene 15 (19): 2277–87. November 1997. doi:10.1038/sj.onc.1201401. PMID 9393873.
- ↑ "Death-associated protein 4 binds MST1 and augments MST1-induced apoptosis". The Journal of Biological Chemistry 277 (50): 47991–8001. December 2002. doi:10.1074/jbc.M202630200. PMID 12384512.
- ↑ "Physical and functional interactions of neuronal growth suppressor necdin with p53". The Journal of Biological Chemistry 274 (23): 16242–8. June 1999. doi:10.1074/jbc.274.23.16242. PMID 10347180.
- ↑ "Stress-dependent nucleolin mobilization mediated by p53-nucleolin complex formation". Molecular and Cellular Biology 22 (16): 6014–22. August 2002. doi:10.1128/MCB.22.16.6014-6022.2002. PMID 12138209.
- ↑ "NUMB controls p53 tumour suppressor activity". Nature 451 (7174): 76–80. January 2008. doi:10.1038/nature06412. PMID 18172499. Bibcode: 2008Natur.451...76C.
- ↑ 196.0 196.1 196.2 "High-throughput sequencing identifies STAT3 as the DNA-associated factor for p53-NF-kappaB-complex-dependent gene expression in human heart failure". Genome Medicine 2 (6). June 2010. doi:10.1186/gm158. PMID 20546595.
- ↑ 197.0 197.1 "Ribosomal protein L11 negatively regulates oncoprotein MDM2 and mediates a p53-dependent ribosomal-stress checkpoint pathway". Molecular and Cellular Biology 23 (23): 8902–12. December 2003. doi:10.1128/MCB.23.23.8902-8912.2003. PMID 14612427.
- ↑ "Parc: a cytoplasmic anchor for p53". Cell 112 (1): 29–40. January 2003. doi:10.1016/S0092-8674(02)01255-2. PMID 12526791.
- ↑ "Poly(ADP-ribose) binds to specific domains of p53 and alters its DNA binding functions". The Journal of Biological Chemistry 273 (19): 11839–43. May 1998. doi:10.1074/jbc.273.19.11839. PMID 9565608.
- ↑ "Involvement of PIAS1 in the sumoylation of tumor suppressor p53". Molecular Cell 8 (3): 713–8. September 2001. doi:10.1016/S1097-2765(01)00349-5. PMID 11583632.
- ↑ "Role of Pin1 in the regulation of p53 stability and p21 transactivation, and cell cycle checkpoints in response to DNA damage". The Journal of Biological Chemistry 277 (50): 47976–9. December 2002. doi:10.1074/jbc.C200538200. PMID 12388558.
- ↑ "The prolyl isomerase Pin1 reveals a mechanism to control p53 functions after genotoxic insults". Nature 419 (6909): 853–7. October 2002. doi:10.1038/nature01120. PMID 12397362. Bibcode: 2002Natur.419..853Z.
- ↑ "Enhancement of p53-dependent gene activation by the transcriptional coactivator Zac1". Oncogene 20 (17): 2134–43. April 2001. doi:10.1038/sj.onc.1204298. PMID 11360197.
- ↑ "Plk3 functionally links DNA damage to cell cycle arrest and apoptosis at least in part via the p53 pathway". The Journal of Biological Chemistry 276 (46): 43305–12. November 2001. doi:10.1074/jbc.M106050200. PMID 11551930.
- ↑ "Mammalian Polo-like kinase 3 (Plk3) is a multifunctional protein involved in stress response pathways". Oncogene 21 (43): 6633–40. September 2002. doi:10.1038/sj.onc.1205850. PMID 12242661.
- ↑ "PACT: cloning and characterization of a cellular p53 binding protein that interacts with Rb". Oncogene 14 (2): 145–55. January 1997. doi:10.1038/sj.onc.1200825. PMID 9010216.
- ↑ "Prohibitin induces the transcriptional activity of p53 and is exported from the nucleus upon apoptotic signaling". The Journal of Biological Chemistry 278 (48): 47853–61. November 2003. doi:10.1074/jbc.M305171200. PMID 14500729.
- ↑ "Regulation of p53 activity in nuclear bodies by a specific PML isoform". The EMBO Journal 19 (22): 6185–95. November 2000. doi:10.1093/emboj/19.22.6185. PMID 11080164.
- ↑ "The function of PML in p53-dependent apoptosis". Nature Cell Biology 2 (10): 730–6. October 2000. doi:10.1038/35036365. PMID 11025664.
- ↑ 210.0 210.1 "Proteasome activator PA28 gamma regulates p53 by enhancing its MDM2-mediated degradation". The EMBO Journal 27 (6): 852–64. March 2008. doi:10.1038/emboj.2008.25. PMID 18309296.
- ↑ "Nuclear FAK promotes cell proliferation and survival through FERM-enhanced p53 degradation". Molecular Cell 29 (1): 9–22. January 2008. doi:10.1016/j.molcel.2007.11.031. PMID 18206965.
- ↑ "Human securin interacts with p53 and modulates p53-mediated transcriptional activity and apoptosis". Nature Genetics 32 (2): 306–11. October 2002. doi:10.1038/ng997. PMID 12355087.
- ↑ "p53 is linked directly to homologous recombination processes via RAD51/RecA protein interaction". The EMBO Journal 15 (8): 1992–2002. April 1996. doi:10.1002/j.1460-2075.1996.tb00550.x. PMID 8617246.
- ↑ "Interaction of p53 with the human Rad51 protein". Nucleic Acids Research 25 (19): 3868–74. October 1997. doi:10.1093/nar/25.19.3868. PMID 9380510.
- ↑ "Pirh2, a p53-induced ubiquitin-protein ligase, promotes p53 degradation". Cell 112 (6): 779–91. March 2003. doi:10.1016/S0092-8674(03)00193-4. PMID 12654245.
- ↑ "Molecular basis of Pirh2-mediated p53 ubiquitylation". Nature Structural & Molecular Biology 15 (12): 1334–42. December 2008. doi:10.1038/nsmb.1521. PMID 19043414.
- ↑ "The interaction of p53 with replication protein A mediates suppression of homologous recombination". Oncogene 23 (56): 9025–33. December 2004. doi:10.1038/sj.onc.1207982. PMID 15489903.
- ↑ "UV-induced DNA incision and proliferating cell nuclear antigen recruitment to repair sites occur independently of p53-replication protein A interaction in p53 wild type and mutant ovarian carcinoma cells". Carcinogenesis 22 (12): 1971–8. December 2001. doi:10.1093/carcin/22.12.1971. PMID 11751427.
- ↑ "Inhibiting S100B restores p53 levels in primary malignant melanoma cancer cells". The Journal of Biological Chemistry 279 (32): 34071–7. August 2004. doi:10.1074/jbc.M405419200. PMID 15178678.
- ↑ 220.0 220.1 "Covalent modification of p73alpha by SUMO-1. Two-hybrid screening with p73 identifies novel SUMO-1-interacting proteins and a SUMO-1 interaction motif". The Journal of Biological Chemistry 275 (46): 36316–23. November 2000. doi:10.1074/jbc.M004293200. PMID 10961991.
- ↑ 221.0 221.1 "p14ARF interacts with DAXX: effects on HDM2 and p53". Cell Cycle 7 (12): 1836–50. June 2008. doi:10.4161/cc.7.12.6025. PMID 18583933.
- ↑ 222.0 222.1 "SWI/SNF complex interacts with tumor suppressor p53 and is necessary for the activation of p53-mediated transcription". The Journal of Biological Chemistry 277 (25): 22330–7. June 2002. doi:10.1074/jbc.M111987200. PMID 11950834.
- ↑ "A direct interaction between the survival motor neuron protein and p53 and its relationship to spinal muscular atrophy". The Journal of Biological Chemistry 277 (4): 2852–9. January 2002. doi:10.1074/jbc.M108769200. PMID 11704667.
- ↑ "Wild-type p53 binds to the TATA-binding protein and represses transcription". Proceedings of the National Academy of Sciences of the United States of America 89 (24): 12028–32. December 1992. doi:10.1073/pnas.89.24.12028. PMID 1465435. Bibcode: 1992PNAS...8912028S.
- ↑ "Pax-6 interactions with TATA-box-binding protein and retinoblastoma protein". Investigative Ophthalmology & Visual Science 40 (7): 1343–50. June 1999. PMID 10359315.
- ↑ "Tumor suppressor activity of AP2alpha mediated through a direct interaction with p53". The Journal of Biological Chemistry 277 (47): 45028–33. November 2002. doi:10.1074/jbc.M208924200. PMID 12226108.
- ↑ "Functional interaction between DP-1 and p53". Molecular and Cellular Biology 16 (10): 5888–95. October 1996. doi:10.1128/mcb.16.10.5888. PMID 8816502.
- ↑ "p53 and metabolism: Inside the TIGAR". Cell 126 (1): 30–2. July 2006. doi:10.1016/j.cell.2006.06.032. PMID 16839873.
- ↑ "The interaction between p53 and DNA topoisomerase I is regulated differently in cells with wild-type and mutant p53". Proceedings of the National Academy of Sciences of the United States of America 96 (18): 10355–60. August 1999. doi:10.1073/pnas.96.18.10355. PMID 10468612. Bibcode: 1999PNAS...9610355G.
- ↑ "Subnuclear distribution of topoisomerase I is linked to ongoing transcription and p53 status". Proceedings of the National Academy of Sciences of the United States of America 99 (3): 1235–40. February 2002. doi:10.1073/pnas.022631899. PMID 11805286. Bibcode: 2002PNAS...99.1235M.
- ↑ 231.0 231.1 "Human topoisomerase IIalpha and IIbeta interact with the C-terminal region of p53". Experimental Cell Research 255 (1): 86–94. February 2000. doi:10.1006/excr.1999.4772. PMID 10666337.
- ↑ "Crystal structure of human 53BP1 BRCT domains bound to p53 tumour suppressor". The EMBO Journal 21 (14): 3863–72. July 2002. doi:10.1093/emboj/cdf383. PMID 12110597.
- ↑ "Comparison of BRCT domains of BRCA1 and 53BP1: a biophysical analysis". Protein Science 13 (3): 617–25. March 2004. doi:10.1110/ps.03461404. PMID 14978302.
- ↑ "The 8-kDa dynein light chain binds to p53-binding protein 1 and mediates DNA damage-induced p53 nuclear accumulation". The Journal of Biological Chemistry 280 (9): 8172–9. March 2005. doi:10.1074/jbc.M411408200. PMID 15611139.
- ↑ "Structure of the 53BP1 BRCT region bound to p53 and its comparison to the Brca1 BRCT structure". Genes & Development 16 (5): 583–93. March 2002. doi:10.1101/gad.959202. PMID 11877378.
- ↑ "Purification, crystallization and preliminary X-ray analysis of the BRCT domains of human 53BP1 bound to the p53 tumour suppressor". Acta Crystallographica D 58 (Pt 10 Pt 2): 1826–9. October 2002. doi:10.1107/S0907444902010910. PMID 12351827. Bibcode: 2002AcCrD..58.1826D.
- ↑ 237.0 237.1 "Two cellular proteins that bind to wild-type but not mutant p53". Proceedings of the National Academy of Sciences of the United States of America 91 (13): 6098–102. June 1994. doi:10.1073/pnas.91.13.6098. PMID 8016121. Bibcode: 1994PNAS...91.6098I.
- ↑ "The p53-binding protein 53BP2 also interacts with Bc12 and impedes cell cycle progression at G2/M". Molecular and Cellular Biology 16 (7): 3884–92. July 1996. doi:10.1128/MCB.16.7.3884. PMID 8668206.
- ↑ "TP53INP1s and homeodomain-interacting protein kinase-2 (HIPK2) are partners in regulating p53 activity". The Journal of Biological Chemistry 278 (39): 37722–9. September 2003. doi:10.1074/jbc.M301979200. PMID 12851404.
- ↑ "p53DINP1, a p53-inducible gene, regulates p53-dependent apoptosis". Molecular Cell 8 (1): 85–94. July 2001. doi:10.1016/S1097-2765(01)00284-2. PMID 11511362.
- ↑ "A TSG101/MDM2 regulatory loop modulates MDM2 degradation and MDM2/p53 feedback control". Proceedings of the National Academy of Sciences of the United States of America 98 (4): 1619–24. February 2001. doi:10.1073/pnas.98.4.1619. PMID 11172000. Bibcode: 2001PNAS...98.1619L.
- ↑ "Supramolecular complex formation between Rad6 and proteins of the p53 pathway during DNA damage-induced response". Molecular and Cellular Biology 23 (7): 2463–75. April 2003. doi:10.1128/MCB.23.7.2463-2475.2003. PMID 12640129.
- ↑ "Associations of UBE2I with RAD52, UBL1, p53, and RAD51 proteins in a yeast two-hybrid system". Genomics 37 (2): 183–6. October 1996. doi:10.1006/geno.1996.0540. PMID 8921390. https://zenodo.org/record/1229705.
- ↑ "Structural basis for E2-mediated SUMO conjugation revealed by a complex between ubiquitin-conjugating enzyme Ubc9 and RanGAP1". Cell 108 (3): 345–56. February 2002. doi:10.1016/S0092-8674(02)00630-X. PMID 11853669. Bibcode: 2002Cell..108..345B.
- ↑ "Identification of c-Cbl as a new ligase for insulin-like growth factor-I receptor with distinct roles from Mdm2 in receptor ubiquitination and endocytosis". Cancer Research 68 (14): 5669–77. July 2008. doi:10.1158/0008-5472.CAN-07-6364. PMID 18632619.
- ↑ "The tumour suppressor RASSF1A promotes MDM2 self-ubiquitination by disrupting the MDM2-DAXX-HAUSP complex". The EMBO Journal 27 (13): 1863–74. July 2008. doi:10.1038/emboj.2008.115. PMID 18566590.
- ↑ "CARPs enhance p53 turnover by degrading 14-3-3sigma and stabilizing MDM2". Cell Cycle 7 (5): 670–82. March 2008. doi:10.4161/cc.7.5.5701. PMID 18382127.
- ↑ "Hedgehog signaling overrides p53-mediated tumor suppression by activating Mdm2". Proceedings of the National Academy of Sciences of the United States of America 105 (12): 4838–43. March 2008. doi:10.1073/pnas.0712216105. PMID 18359851. Bibcode: 2008PNAS..105.4838A.
- ↑ "Specific inhibition of Mdm2-mediated neddylation by Tip60". Cell Cycle 7 (2): 222–31. January 2008. doi:10.4161/cc.7.2.5185. PMID 18264029.
- ↑ "Deubiquitination of p53 by HAUSP is an important pathway for p53 stabilization". Nature 416 (6881): 648–53. April 2002. doi:10.1038/nature737. PMID 11923872. Bibcode: 2002Natur.416..648L.
- ↑ "p53 Modulates the exonuclease activity of Werner syndrome protein". The Journal of Biological Chemistry 276 (37): 35093–102. September 2001. doi:10.1074/jbc.M103332200. PMID 11427532.
- ↑ "Hyaluronidase induction of a WW domain-containing oxidoreductase that enhances tumor necrosis factor cytotoxicity". The Journal of Biological Chemistry 276 (5): 3361–70. February 2001. doi:10.1074/jbc.M007140200. PMID 11058590.
- ↑ "Direct interaction of p53 with the Y-box binding protein, YB-1: a mechanism for regulation of human gene expression". Oncogene 19 (54): 6194–202. December 2000. doi:10.1038/sj.onc.1204029. PMID 11175333.
- ↑ "YPEL3, a p53-regulated gene that induces cellular senescence". Cancer Research 70 (9): 3566–75. May 2010. doi:10.1158/0008-5472.CAN-09-3219. PMID 20388804.
- ↑ "ATM-dependent activation of p53 involves dephosphorylation and association with 14-3-3 proteins". Nature Genetics 19 (2): 175–8. June 1998. doi:10.1038/542. PMID 9620776.
- ↑ "Physical interaction between p53 and primary response gene Egr-1". International Journal of Oncology 18 (4): 863–70. April 2001. doi:10.3892/ijo.18.4.863. PMID 11251186.
- ↑ "ZBP-89 promotes growth arrest through stabilization of p53". Molecular and Cellular Biology 21 (14): 4670–83. July 2001. doi:10.1128/MCB.21.14.4670-4683.2001. PMID 11416144.
- ↑ "MiR-34, SIRT1 and p53: the feedback loop". Cell Cycle 8 (5): 712–5. March 2009. doi:10.4161/cc.8.5.7753. PMID 19221490.
- ↑ "CircRNA_014511 affects the radiosensitivity of bone marrow mesenchymal stem cells by binding to miR-29b-2-5p". Bosnian Journal of Basic Medical Sciences 19 (2): 155–163. May 2019. doi:10.17305/bjbms.2019.3935. PMID 30640591.
External links
- "p53 Knowledgebase". Lane Group at the Institute of Molecular and Cell Biology (IMCB), Singapore. http://p53.bii.a-star.edu.sg/.
- GeneReviews/NCBI/NIH/UW entry on Li-Fraumeni Syndrome
- TUMOR PROTEIN p53 @ OMIM
- p53 restoration of function
- p53 @ The Atlas of Genetics and Cytogenetics in Oncology and Haematology
- TP53 Gene @ GeneCards
- p53 News provided by insciences organisation
- "p53 Tumor Suppressor". Molecule of the Month. RCSB Protein Data Bank. 2002-07-01. http://pdb101.rcsb.org/motm/31.
- "p53 Web Site". http://p53.free.fr/.
- Living LFS A non-profit Li-Fraumeni Syndrome patient support organization
- The George Pantziarka TP53 Trust A support group from the UK for people with Li-Fraumeni Syndrome or other TP53-related disorders
- IARC TP53 Somatic Mutations database maintained at IARC, Lyon, by Magali Olivier
- PDBe-KB provides an overview of all the structure information available in the PDB for Human P53.
- scientific animation conformational changes of p53 upon binding to DNA
 |