Chemistry:Surflon S-111
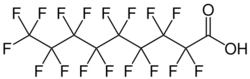
Surflon S-111 (CAS 72968-3-88) is a commercial product consisting of perfluorinated carboxylic acids (PFCAs) in ammonium salt form. It is commonly used as a polymerization aid in the production of fluoropolymers.[1]
The dominant chemical compound is perfluorononanoic acid (PFNA) at 74% by weight, followed by the 11 carbon perfluoroundecanoic acid (20%), and the 13 carbon perfluorotridecanoic acid (5%).[2]
Surflon S-111 is synthesized in Japan by oxidizing a mixture of fluorotelomer olefins.[2] Fluorotelomer olefins are synthesized using a telomerization of tetrafluoroethylene taxogens (monomers),[1][3] followed by an ethylene insertion.[3] The olefin is oxidized, removing one carbon to yield products with an odd number of carbons[2] with even-lengthed fluorocarbon chains plus a carboxylic acid group. As the fluorotelomer olefins are dominated by F(CF2)8CH=CH2,[2][4] PFNA is the major PFCA product.
Surflon S-111 "is described as Fatty acids, C7-C13, perfluoro, ammonium salts".[2]
See also
References
- ↑ 1.0 1.1 "PFOA in Norway TA-2354/2007". Norwegian Pollution Control Authority. 2007. pp. 74–75. http://www.sft.no/publikasjoner/2354/ta2354.pdf. Retrieved 2009-04-06.
- ↑ 2.0 2.1 2.2 2.3 2.4 "Sources, fate and transport of perfluorocarboxylates". Environ Sci Technol 40 (1): 32–44. January 2006. doi:10.1021/es0512475. PMID 16433330. Supporting Information (PDF).
- ↑ 3.0 3.1 Lehmler, HJ (Mar 2005). "Synthesis of environmentally relevant fluorinated surfactants—a review". Chemosphere 58 (11): 1471–96. doi:10.1016/j.chemosphere.2004.11.078. PMID 15694468.
- ↑ "PERFORCE — PERFLUORINATED ORGANIC COMPOUNDS IN THE EUROPEAN ENVIRONMENT". Institute for Biodiversity and Ecosystem Dynamics, Universiteit van Amsterdam. 2006-09-15. pp. 18. http://www.science.uva.nl/perforce/Final%20reportA.pdf. Retrieved 2009-02-15.
 |

