Chemistry:Tellurophene
Tellurophene is the organotellurium compound with the formula C
4H
4Te. It is a heavy analogue of thiophene and selenophene. The compound is a pale yellow liquid. A number of substituted tellurophenes are known.[1]
Synthesis
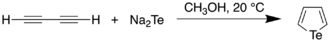
In 1966, Mack report a synthesis of an unsubstituted tellurophene through the reaction of sodium telluride with diacetylene in methanol. This method could be generalised to prepare 2,5-derivatives of tellurophene by selecting a suitably-substituted diacetylene precursor.[2] The product was obtained as a pale yellow liquid with a melting and boiling point of −36 °C and 148 °C, respectively. Taticchi et al. improved upon this synthesis by using a Schlenk line to exclude oxygen and moisture from the reaction vessel, using pure butadiyne (to decrease unwanted oxidation and polymerization side reactions), and by not using a vacuum to remove the methanol as it leads to loss of the product. This improved procedure allowed the tellurophene to be isolated in 47% yield.[3][4] Hydrogen telluride (HTe-) and tellurols (RTeH) are implicated in these conversions.
Structure and bonding
The geometry of tellurophene was first determined in 1973 through microwave spectroscopy and has been further refined through X-ray diffraction studies.[5] The Te–C bond length is 2.046 Å and the C–Te–C angle is 82°. These findings are consistent with decreased aromaticity vs that of selenophene and related heterocycles.[3][6]
Reactivity
Tellurophene forms poly(tellurophene) upon treatment with ferric chloride.[7]
- C
4H
4Te + 2 FeCl
3 → 1/n[C
4H
2Te]
n + 2 HCl + 2 FeCl
2
The conversion, an oxidative polymerization, is modeled after the corresponding synthesis of polythiophene. When treated with halogens, tellurophene gives a Te(IV) derivative:
- C
4H
4Te + Cl
2 → C
4H
4TeCl
2
Treatment of tellurophene with tert-butyllithium gives 2-lithiotellurophene.[8]
References
- ↑ Karapala, Vamsi Krishna; Shih, Hong-Pin; Han, Chien-Chung (2018). "Cascade and Effective Syntheses of Functionalized Tellurophenes". Organic Letters 20 (6): 1550–1554. doi:10.1021/acs.orglett.8b00279. ISSN 1523-7060. PMID 29494165.
- ↑ Mack, W. (1966). "Synthesis of Tellurophene and its 2,5-Disubstituted Derivatives". Angew. Chem. Int. Ed. 5 (10): 896. doi:10.1002/anie.196608961.
- ↑ 3.0 3.1 Fringuelli, Francesco; Marino, Gianlorenzo; Taticchi, Aldo (1977). "Tellurophene and Related Compounds". Advances in Heterocyclic Chemistry Volume 21. Advances in Heterocyclic Chemistry. 21. pp. 119–173. doi:10.1016/S0065-2725(08)60731-X. ISBN 9780120206216. https://books.google.com/books?id=1qIOmeTdiGcC&pg=PA119.
- ↑ Fringuelli, Francesco; Taticchi, Aldo (1972). "Tellurophen and some of its derivatives". Journal of the Chemical Society, Perkin Transactions 1: 199–203. doi:10.1039/P19720000199.
- ↑ Lukevics, E.; Arsenyan, P.; Belyakov, S.; Pudova, O. (2002). "Molecular Structure of Selenophenes and Tellurophenes". Chemistry of Heterocyclic Compounds 38 (7): 763–777. doi:10.1023/a:1020607300418. ISSN 0009-3122.
- ↑ Fringuelli, Francesco; Marino, Gianlorenzo; Taticchi, Aldo; Grandolini, Giuliano (1974). "A comparative study of the aromatic character of furan, thiophen, selenophen, and tellurophen". Journal of the Chemical Society, Perkin Transactions 2 1974 (4): 332–337. doi:10.1039/P29740000332.
- ↑ Sugimoto, Ryu-Ichi; Yoshino, Katsumi; Inoue, Shigehito; Tsukagoshi, Kunimitsu (1985). "Preparation and Property of Polytellurophene and Polyselenophene". Japanese Journal of Applied Physics 24 (6A): L425. doi:10.1143/JJAP.24.L425. Bibcode: 1985JaJAP..24L.425S.
- ↑ Jahnke, Ashlee A.; Howe, Graeme W.; Seferos, Dwight S. (2010). "Polytellurophenes with Properties Controlled by Tellurium-Coordination". Angewandte Chemie International Edition 49 (52): 10140–10144. doi:10.1002/anie.201005664. PMID 21105034.
 |
