Chemistry:Vitamin A2
From HandWiki
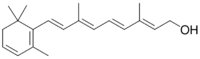
| |
| Names | |
|---|---|
| IUPAC name
3,4-Didehydroretinol
| |
| Preferred IUPAC name
(2E,4E,6E,8E)-3,7-Dimethyl-9-(2,6,6-trimethylcyclohexa-1,3-dien-1-yl)nona-2,4,6,8-tetraen-1-ol | |
| Other names
Retinol 2; 3,4-Dehydroretinol
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C20H28O | |
| Molar mass | 284.443 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Vitamin A2 is a subcategory of vitamin A.[1]
As with all vitamin A forms, A2 can exist as an aldehyde, Dehydroretinal (3,4-dehydroretinal), an alcohol, 3,4-dehydroretinol (vitamin A2 alcohol) or an acid, 3,4-dehydroretinoic acid (vitamin A2 acid). Many cold-blooded vertebrates use the aldehyde for their visual system to obtain a red-shifted sensitive spectrum.[2]
Human skin naturally contains the alcohol form.[3][4] In humans, CYP27C1 converts ordinary A1 (all-trans retinoids) to A2. The enzyme also converts 11-cis-retinal.[2]
Vitamin A2 was first identified by Richard Alan Morton using newly-developed absorption spectroscopy in 1941.[5]
References
- ↑ "The Biochemical Basis of Vitamin A3 Production in Arthropod Vision". ACS Chem Biol 11 (4): 1049–1057. 2016. doi:10.1021/acschembio.5b00967. PMID 26811964.
- ↑ 2.0 2.1 Kramlinger, VM; Nagy, LD; Fujiwara, R; Johnson, KM; Phan, TT; Xiao, Y; Enright, JM; Toomey, MB et al. (May 2016). "Human cytochrome P450 27C1 catalyzes 3,4-desaturation of retinoids.". FEBS Letters 590 (9): 1304–12. doi:10.1002/1873-3468.12167. PMID 27059013.
- ↑ "Biosynthesis of 3-dehydroretinol (vitamin A2) from all-trans-retinol (vitamin A1) in human epidermis". J Invest Dermatol 85 (6): 498–500. 1985. doi:10.1111/1523-1747.ep12277290. PMID 4067325.
- ↑ "The identification of dehydroretinol (vitamin A2) in human skin". Experientia 36 (3): 317–318. 1980. doi:10.1007/bf01952299. PMID 7371787.
- ↑ Goodwin, T W (1977). "R. A. Morton". Nature 266 (5600): 394. doi:10.1038/266394a0. Bibcode: 1977Natur.266..394G.
 |
