Organization:Amino acid N-carboxyanhydride
N-Carboxyanhydrides | |
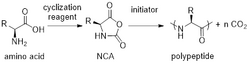 Formation and polymerization of an NCA. |
Amino acid N-carboxyanhydrides, also called Leuchs' anhydrides, are a family of heterocyclic organic compounds derived from amino acids. They are white, moisture-reactive solids. They have been evaluated for applications the field of biomaterials.[1][2][3]
Preparation
thumb|right|140px|Glycine N-carboxyanhydride is the parent member of the amino acid N-carboxyanhydrides.
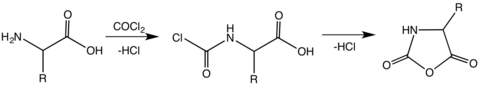
NCAs are typically prepared by phosgenation of amino acids:[4]
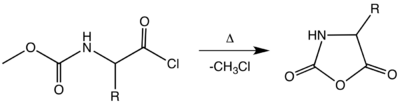
They were first synthesized by Hermann Leuchs by heating an N-ethoxycarbonyl or N-methoxycarbonyl amino acid chloride in a vacuum at 50-70 °C:[5][6]
A moisture-tolerant route to unprotected NCAs employs epoxides as scavengers of hydrogen chloride.[7]
This synthesis of NCAs is sometimes called the Template:Va. The relatively high temperatures necessary for this cyclization results in the decomposition of several NCAs. Of several improvements, one notable procedure involves treating an unprotected amino acid with phosgene or its trimer.[8][9][10]
Reactions
NCAs are prone to hydrolysis to the parent amino acid:
- RCHNHC(O)OC(O) + H2O → H2NCH(R)CO2H + CO2
Some derivatives however tolerate water briefly.[7]
NCAs convert to homopolypeptides ( [N(H)CH(R)CO)]n) through ring-opening polymerization:[1][2][3][11][12][13][14]
- n RCHNHC(O)OC(O) → [N(H)CH(R)CO)]n + n CO2
Poly-L-lysine has been prepared from N-carbobenzyloxy-α-N-carboxy-L-lysine anhydride, followed by deprotection with phosphonium iodide.[15] Peptide synthesis from NCAs does not require protection of the amino acid functional groups. N-Substituted NCAs, such as sulfenamide derivatives have also been examined.[16] The ring-opening polymerization of NCAs is catalyzed by metal catalysts.[17][3][6][11]
The polymerization of NCA’s have been considered as a prebiotic route to polypeptides.[3][18] [19]
Further reading
- "Aqueous Ring-Opening Polymerization-Induced Self-Assembly (ROPISA) of N-Carboxyanhydrides". Angewandte Chemie 59 (2): 622–626. January 2020. doi:10.1002/ange.201912028. PMID 31650664. https://hal.archives-ouvertes.fr/hal-02333682/file/ROPISA_HAL.pdf.
- "Ring-Opening Polymerization of N-Carboxyanhydride-Induced Self-Assembly for Fabricating Biodegradable Polymer Vesicles". ACS Macro Letters 8 (10): 1216–1221. 2019-10-15. doi:10.1021/acsmacrolett.9b00606. PMID 35651173.
- "Development of a liver-targeted siRNA delivery platform with a broad therapeutic window utilizing biodegradable polypeptide-based polymer conjugates". Journal of Controlled Release 183: 124–37. June 2014. doi:10.1016/j.jconrel.2014.03.028. PMID 24657948.
- "Chemically tunable mucin chimeras assembled on living cells". Proceedings of the National Academy of Sciences of the United States of America 112 (41): 12574–9. October 2015. doi:10.1073/pnas.1516127112. PMID 26420872. Bibcode: 2015PNAS..11212574K.
See also
- Dakin–West reaction
- Glycine N-carboxyanhydride, the parent NCA
References
- ↑ 1.0 1.1 Polypeptide Nanomaterials. 1-4. Soft Matter and Biomaterials on the Nanoscale: World Scientific. 2020. pp. 115–180. Bibcode: 2020smb3.book..115K.
- ↑ 2.0 2.1 "Self-assembly at all scales". Science 295 (5564): 2418–21. March 2002. doi:10.1126/science.1070821. PMID 11923529. Bibcode: 2002Sci...295.2418W.
- ↑ 3.0 3.1 3.2 3.3 "Polypeptides and 100 years of chemistry of alpha-amino acid N-carboxyanhydrides". Angewandte Chemie 45 (35): 5752–84. September 2006. doi:10.1002/anie.200600693. PMID 16948174.
- ↑ Lyndon C. Xavier, Julie J. Mohan, David J. Mathre, Andrew S. Thompson, James D. Carroll, Edward G. Corley, and Richard Desmond (1997). "(S)-Tetrahydro-1-methyl-3,3-diphenyl-1h,3h-pyrrolo-[1,2-c] [1,3,2]oxazaborole-borane Complex". Organic Syntheses 74: 50. doi:10.15227/orgsyn.074.0050.
- ↑ "Ueber die Glycin-carbonsäure" (in German). Berichte der Deutschen Chemischen Gesellschaft 39: 857–61. 1906. doi:10.1002/cber.190603901133. https://zenodo.org/record/1426172.
- ↑ 6.0 6.1 "Synthetic polypeptides for biomedical applications". Prog. Polym. Sci. 32 (8–9): 858–875. 2007. doi:10.1016/j.progpolymsci.2007.05.010.
- ↑ 7.0 7.1 "A Moisture-Tolerant Route to Unprotected α/β-Amino acid N-Carboxyanhydrides and Facile Synthesis of Hyperbranched Polypeptides". Nature Communications 12 (1): 5810. October 2021. doi:10.1038/s41467-021-25689-y. PMID 34608139. Bibcode: 2021NatCo..12.5810T.
- ↑ "Amide bond formation and peptide coupling". Tetrahedron 61 (46): 10827–52. 2005. doi:10.1016/j.tet.2005.08.031.
- ↑ "(S)-Tetrahydro-1-methyl-3,3-diphenyl-1h,3h-pyrrolo-[1,2-c] [1,3,2]oxazaborole-borane Complex". Org. Synth. 74: 50. 1997. doi:10.15227/orgsyn.074.0050.
- ↑ "Improved synthesis of O-linked, and first synthesis of S- linked, carbohydrate functionalised N-carboxyanhydrides (glycoNCAs)". Organic & Biomolecular Chemistry 5 (17): 2756–7. September 2007. doi:10.1039/b707563d. PMID 17700840.
- ↑ 11.0 11.1 "Synthesis of nearly monodisperse polystyrene-polypeptide block copolymers via polymerisation of N-carboxyanhydrides". Chemical Communications (23): 2944–2945. December 2003. doi:10.1039/b308990h. PMID 14680253.
- ↑ "Synthetic Polypeptides: from Polymer Design to Supramolecular Assembly and Biomedical Application". Chemical Society Reviews 46 (21): 6570–6599. October 2017. doi:10.1039/C7CS00460E. PMID 28944387.
- ↑ "Smart and genetically engineered biomaterials and drug delivery systems". European Journal of Pharmaceutical Sciences 20 (1): 1–16. September 2003. doi:10.1016/S0928-0987(03)00164-7. PMID 13678788.
- ↑ "Recent Advances in Amino Acid N-Carboxyanhydrides and Synthetic Polypeptides: Chemistry, Self-Assembly and Biological Applications". Chemical Communications 50 (2): 139–55. January 2014. doi:10.1039/c3cc46317f. PMID 24217557.
- ↑ "My contributions to science and society". The Journal of Biological Chemistry 280 (17): 16529–41. April 2005. doi:10.1074/jbc.X400013200. PMID 15718236.
- ↑ "Peptide synthesis using o-nitrophenylsulfenyl N-carboxy alpha-amino acid anhydrides". The Journal of Organic Chemistry 40 (19): 2697–2702. September 1975. doi:10.1021/jo00907a001. PMID 1177065.
- ↑ "Amino Acid Derived Nickelacycles: Intermediates in Nickel-Mediated Polypeptide Synthesis". Journal of the American Chemical Society 120 (17): 4240–4241. 1998. doi:10.1021/ja980313i.
- ↑ "N-carbamoyl-alpha-amino acids rather than free alpha-amino acids formation in the primitive hydrosphere: a novel proposal for the emergence of prebiotic peptides". Origins of Life and Evolution of the Biosphere 28 (1): 61–77. February 1998. doi:10.1023/A:1006566810636. PMID 11536856. Bibcode: 1998OLEB...28...61T.
- ↑ "Synthesis of long prebiotic oligomers on mineral surfaces". Nature 381 (6577): 59–61. May 1996. doi:10.1038/381059a0. PMID 8609988. Bibcode: 1996Natur.381...59F.
 |