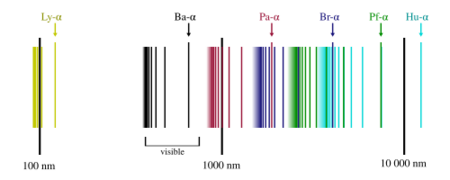Physics:Quantum molecular spectroscopy
Quantum molecular spectroscopy is the study of how molecules absorb, emit, or scatter radiation as they move between quantized molecular energy states. In quantum mechanics, molecular spectra arise because the allowed rotational, vibrational, and electronic states of a molecule are discrete or partly discrete. The observed spectral lines and bands therefore reveal information about bond lengths, force constants, molecular geometry, electronic structure, and interactions with the environment.[1][2]
Overview
A molecule has several kinds of quantum energy. Its electrons occupy molecular electronic states, the nuclei vibrate about equilibrium geometries, and the molecule can rotate. To a useful approximation, the total molecular energy can be written as a sum of electronic, vibrational, and rotational contributions,[1][2]
The energy spacing of these contributions is very different. Rotational transitions usually occur in the microwave or far-infrared region, vibrational transitions in the infrared, and electronic transitions in the visible or ultraviolet. Real spectra often combine these motions, producing rotational, vibrational, rovibrational, electronic, and vibronic structure.[3]
In quantum mechanics
In quantum mechanics, the discrete spectrum of an observable corresponds to the eigenvalues of the operator used to model that observable. According to the mathematical theory of such operators, its eigenvalues may form a discrete set of isolated points, which may be either finite or countable.
Discrete spectra are usually associated with systems that are bound or confined in some sense. The position and momentum operators have continuous spectra in an infinite domain, but a discrete spectrum in a compact or confined domain.[4] Similar spectral ideas apply to angular momentum, Hamiltonians, and other operators of quantum systems.[4]
The quantum harmonic oscillator and the hydrogen atom are examples of physical systems in which the Hamiltonian has a discrete spectrum. In the case of the hydrogen atom the spectrum has both a continuous and a discrete part, the continuous part corresponding to ionization.
-
The discrete part of the emission spectrum of hydrogen.
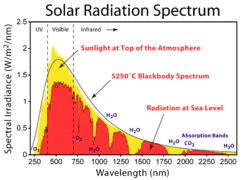
-
Spectrum of sunlight above the atmosphere and at sea level, showing absorption features with discrete and continuous parts.
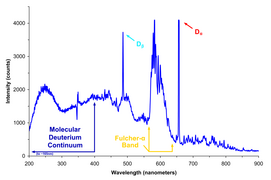
-
Spectrum of light emitted by a deuterium lamp, showing sharp peaks and a continuous background.
Rotational spectra
The simplest rotational model is the rigid rotor. For a diatomic molecule with reduced mass and bond length , the moment of inertia is
The rotational energy levels are
where is the rotational quantum number.[3] In spectroscopic notation the same levels are often written in terms of a rotational constant ,
Pure rotational absorption or emission requires a changing or permanent electric dipole moment. For a simple electric-dipole rotational transition, the selection rule is
Rotational spectra are especially useful for determining molecular moments of inertia and therefore bond lengths and molecular geometry.[1]
Vibrational spectra
Vibrational spectra arise from quantized nuclear motion. In the harmonic oscillator approximation, the vibrational energy levels of a diatomic molecule are
where is the vibrational quantum number and is the natural vibrational frequency of the bond.[3] In the simplest harmonic model, the main infrared selection rule is
Real molecular potentials are anharmonic, so weak overtone and combination bands may also appear. A vibration is infrared-active when it changes the molecular dipole moment. For this reason, homonuclear diatomic molecules such as and do not show ordinary pure vibrational infrared absorption, while heteronuclear molecules such as CO and HCl do.[5]
Rovibrational spectra
In gas-phase infrared spectra, a vibrational transition is often accompanied by rotational transitions. This produces rovibrational bands rather than a single isolated line. The rotational selection rule usually gives two main branches:
- the P branch, with ;
- the R branch, with .
In many simple diatomic spectra the direct line is absent, leaving a gap between the P and R branches. Rovibrational spectra can be used to determine bond lengths, rotational constants, vibrational frequencies, and anharmonic corrections.[6]
Electronic spectra
Electronic spectra involve transitions between molecular electronic states. In a molecular-orbital picture, electronic states are described by molecular orbitals extending over the molecule rather than orbitals localized on a single atom. Electronic transitions often occur together with changes in vibrational and rotational state, producing vibronic bands.[3]
The intensities of electronic-vibrational transitions are often described using the Franck–Condon principle. Because electrons move much faster than nuclei, an electronic transition occurs so rapidly that the nuclear geometry is approximately fixed during the transition. The strongest transitions therefore occur between vibrational states whose wavefunctions overlap strongly at the same nuclear geometry.[1][5]
Selection rules and transition moments
A transition is observed only if the interaction between the molecule and the radiation gives a nonzero transition probability. In electric-dipole spectroscopy this is expressed through the transition dipole moment,
where and are the initial and final molecular states and is the dipole-moment operator. If this matrix element vanishes by symmetry, the transition is forbidden in the electric-dipole approximation.[5]
Selection rules are therefore not arbitrary rules but consequences of molecular symmetry, angular-momentum conservation, and the form of the radiation-matter interaction. Forbidden transitions may still appear weakly when approximations break down, for example through spin-orbit coupling, vibronic coupling, collisions, or external fields.[1]
Spectral lines and molecular information
The position, spacing, and intensity of molecular spectral lines encode physical information. Rotational line spacings can give moments of inertia and bond lengths. Vibrational frequencies reveal bond strengths and force constants. Electronic bands reveal molecular electronic structure, dissociation energies, and excited-state geometry. Isotopic substitution changes reduced masses and therefore shifts rotational and vibrational levels, providing a sensitive test of molecular models.[2][1]
Spectral lines also have finite widths. Line shapes may be affected by natural lifetime broadening, Doppler broadening, pressure broadening, instrumental resolution, and interactions with surrounding matter. These effects are important in laboratory spectroscopy, atmospheric physics, astrophysics, plasma diagnostics, and chemical analysis.[1]
Applications
Molecular spectroscopy is used to identify molecules, measure molecular constants, determine chemical composition, and probe physical conditions. Important applications include infrared absorption spectroscopy, Raman spectroscopy, microwave spectroscopy, atmospheric remote sensing, combustion diagnostics, astrochemistry, plasma diagnostics, and the study of molecular structure and reaction dynamics.[1][3]
In quantum physics, molecular spectroscopy also provides direct evidence for quantized molecular motion. It connects the abstract operator language of quantum mechanics to measurable frequencies, intensities, and line shapes.
See also
Table of contents (72 articles)
Index
Full contents
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Bernath, Peter F. (2016). Spectra of Atoms and Molecules (3rd ed.). Oxford University Press. ISBN 978-0-19-938257-6.
- ↑ 2.0 2.1 2.2 Herzberg, Gerhard (1950). Molecular Spectra and Molecular Structure: I. Spectra of Diatomic Molecules. Van Nostrand Reinhold. ISBN 978-0-442-03385-8.
- ↑ 3.0 3.1 3.2 3.3 3.4 "Spectroscopy - Theory of molecular spectra". https://www.britannica.com/science/spectroscopy/Theory-of-molecular-spectra.
- ↑ 4.0 4.1 Landau, L. D.; Lifshitz, E. M. (1965). Quantum Mechanics: Non-Relativistic Theory. Course of Theoretical Physics. 3. Pergamon Press. https://archive.org/details/QuantumMechanics_104.
- ↑ 5.0 5.1 5.2 "Selection rules and transition moment integral". https://chem.libretexts.org/Core/Physical_and_Theoretical_Chemistry/Spectroscopy/Fundamentals/Selection_rules_and_transition_moment_integral.
- ↑ "Rovibrational Spectroscopy". https://chem.libretexts.org/Core/Physical_and_Theoretical_Chemistry/Spectroscopy/Rotational_Spectroscopy/Rovibrational_Spectroscopy.
```