Physics:Quantum molecular vibration
Quantum molecular vibration is the quantized periodic motion of atoms in a molecule relative to one another, with the center of mass of the molecule remaining unchanged. Molecular vibrations are central to molecular spectroscopy, because vibrational transitions produce characteristic infrared and Raman spectra that reveal bond strengths, molecular geometry, symmetry, and chemical structure.
Typical molecular vibrational frequencies range from less than 1013 Hz to about 1014 Hz, corresponding to wavenumbers of roughly 300–3000 cm−1 and wavelengths of about 30–3 µm. These energies usually lie in the infrared region of the electromagnetic spectrum.
Overview
For a diatomic molecule A–B, the vibrational frequency can be approximated by treating the bond as a spring. In this model,
where is the force constant and is the reduced mass,
The vibrational wavenumber is
where is the speed of light. Stronger bonds and lighter atoms generally give higher vibrational frequencies.
Vibrations of polyatomic molecules are described in terms of normal modes. These modes are independent collective motions of the nuclei, but each normal mode may involve simultaneous movement of several atoms. A nonlinear molecule with atoms has normal modes of vibration, while a linear molecule has modes because rotation about the molecular axis does not change the nuclear coordinates.[1]
Quantum description
A molecular vibration is excited when the molecule absorbs energy corresponding to the vibration frequency. The energy spacing is approximately
where is Planck's constant and is the vibrational frequency. A fundamental vibration occurs when one quantum of vibrational energy is absorbed from the ground vibrational state. When additional quanta are absorbed, overtones may be excited.
To a first approximation, the motion in a normal vibration can be described as simple harmonic motion. In this approximation, the vibrational energy is a quadratic function of the atomic displacement, and the energy levels are evenly spaced:
where is the vibrational quantum number.[2][3]
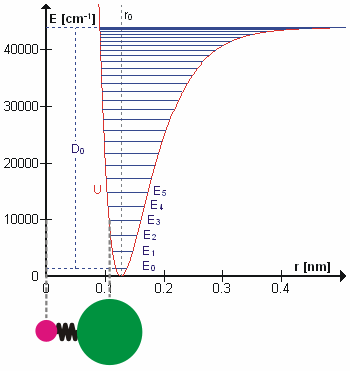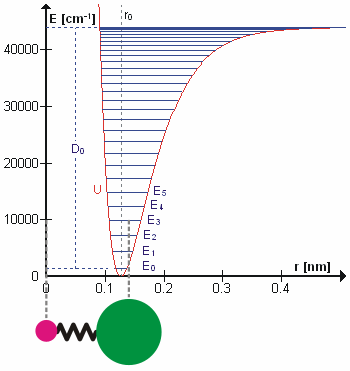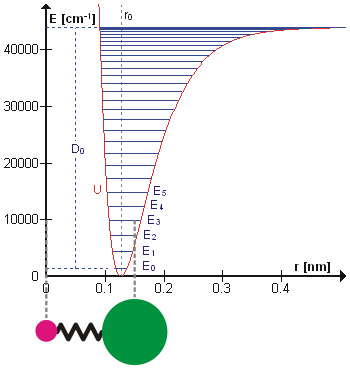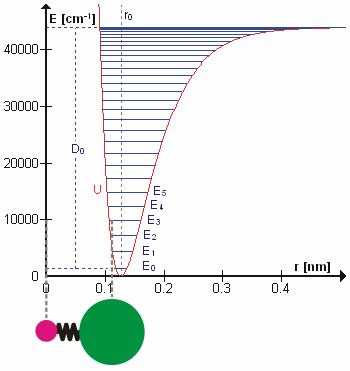
In real molecules, vibrations are anharmonic. The first overtone is therefore slightly less than twice the fundamental frequency, and higher overtones require progressively less additional energy. At sufficiently high vibrational excitation, the molecule may dissociate. The potential energy curve of a real bond is therefore better represented by a Morse potential than by a perfect parabola.
Infrared and Raman spectroscopy
The vibrational states of a molecule can be probed in several ways. The most direct method is infrared spectroscopy, because vibrational transitions often require energies in the infrared region of the spectrum. A vibration is infrared-active when it changes the molecular dipole moment.
Raman spectroscopy can also measure vibrational frequencies. In Raman spectroscopy, the intensity of a band depends on the change of molecular polarizability with respect to the normal coordinate. Infrared and Raman spectroscopy are complementary: some vibrations that are weak or forbidden in infrared spectroscopy may appear strongly in Raman spectra, and vice versa.
Vibrational excitation can also occur together with electronic excitation. The combined transition is called a vibronic transition and gives vibrational fine structure to electronic spectra, especially for molecules in the gas phase.
Simultaneous excitation of vibration and rotation gives rise to vibration–rotation spectra, also called rovibrational spectra.
Number of vibrational modes
For a molecule with atoms, the positions of all nuclei depend on coordinates. These coordinates include translation, rotation, and vibration. Translation corresponds to motion of the center of mass and accounts for three degrees of freedom.
A nonlinear molecule can rotate about three mutually perpendicular axes and therefore has three rotational degrees of freedom. A linear molecule has only two rotational degrees of freedom because rotation about the molecular axis does not change the positions of the nuclei.[4][5]
Thus the number of vibrational modes is
for nonlinear molecules, and
for linear molecules.[4][5][6]
A diatomic molecule has one vibrational mode, corresponding to stretching and compression of the single bond.
Vibrational coordinates
The coordinate of a normal vibration is a combination of changes in the positions of atoms in the molecule. When the vibration is excited, the coordinate changes approximately sinusoidally with the vibrational frequency.
Internal coordinates
Internal coordinates describe molecular motion in terms of changes in bond lengths, bond angles, and relative group orientations. Common internal coordinates include:
- Stretching: a change in the length of a bond, such as C–H or C–C.
- Bending: a change in the angle between two bonds.
- Rocking: a change in angle between a group of atoms and the rest of the molecule.
- Wagging: motion of a group of atoms out of a reference plane.
- Twisting: a change in the angle between two planes defined by groups of atoms.
- Out-of-plane motion: movement of an atom or bond out of a molecular plane.
These internal coordinates do not necessarily correspond directly to normal modes. A normal mode may be a linear combination of several internal coordinates.
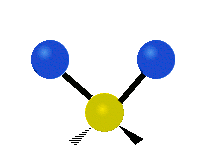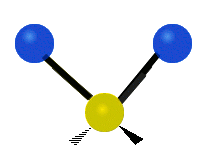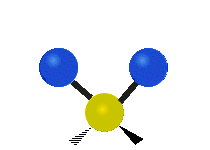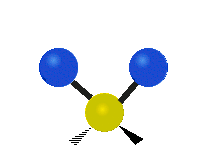
Methylene-group vibrations
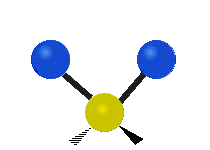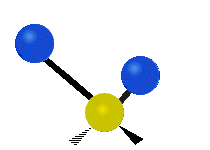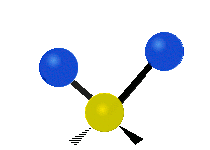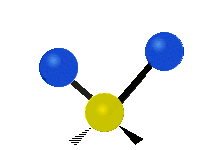
Within a CH2 group, the two hydrogen atoms can vibrate in several characteristic ways. These are commonly grouped as stretching, bending, rocking, wagging, and twisting motions.
| Symmetric stretching | Asymmetric stretching | Scissoring |
|---|---|---|
|
|
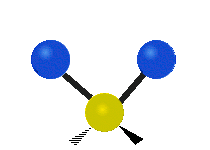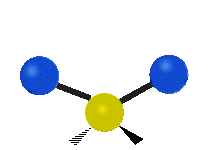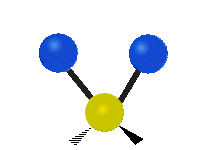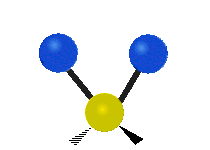
|
| Rocking | Wagging | Twisting |
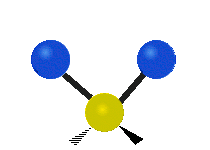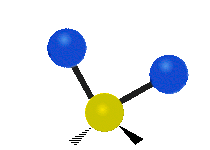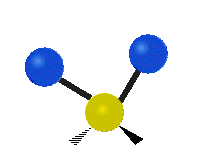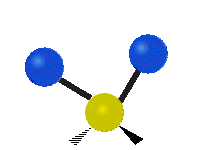
|
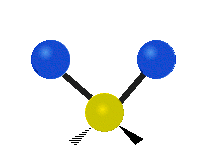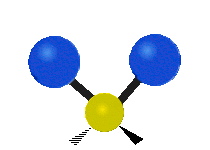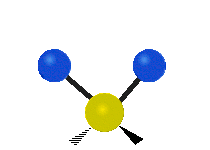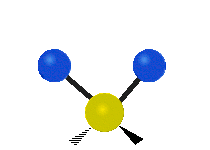
|
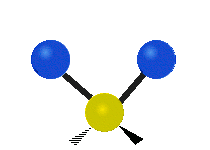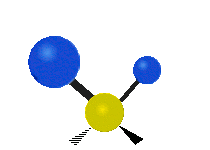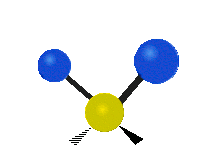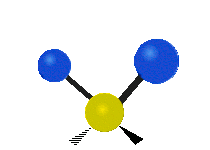
|
Symmetry-adapted coordinates
Symmetry-adapted coordinates are constructed by applying projection operators to internal coordinates. The projection operators are built from the character table of the molecular point group.[7]
For example, the four unnormalized C–H stretching coordinates of ethene may be combined as
Here to are internal coordinates for stretching of the four C–H bonds. Illustrations of symmetry-adapted coordinates for many small molecules can be found in Nakamoto.[8]
Normal coordinates
Normal coordinates, usually denoted , describe displacement along normal modes of vibration. Each normal mode has one normal coordinate. Normal modes diagonalize the matrix governing molecular vibrations, so each normal mode behaves as an independent vibration in the harmonic approximation.
If the molecule has symmetry, its normal modes transform according to irreducible representations of the molecular point group. For example, in carbon dioxide, the two C–O stretches are not independent local bond motions. They combine into a symmetric stretch and an asymmetric stretch:
- Symmetric stretching: the two C–O bond lengths change by the same amount.
- Asymmetric stretching: one C–O bond length increases while the other decreases.
When two or more normal coordinates have the same symmetry, mixing occurs and the coefficients of the linear combination cannot be determined by symmetry alone. A full normal-coordinate analysis is then required.
The Wilson GF method is a standard way to calculate normal modes and vibrational frequencies.[9]
Newtonian approximation
Although molecular vibrations are quantum mechanical, their approximate frequencies can often be calculated with Newtonian mechanics. In the harmonic approximation, each vibration is treated as if it were a spring obeying Hooke's law:
By Newton's second law,
so the equation of motion becomes
Its solution is
In this expression, is the amplitude, is the force constant, and is the reduced mass. For a diatomic molecule AB,
The use of the reduced mass ensures that the center of mass of the molecule is not affected by the vibration. In the harmonic approximation, the potential energy is quadratic in the normal coordinate:
The force constant is therefore related to the curvature of the potential energy surface:
When two or more normal vibrations have the same symmetry, a full normal coordinate analysis must be performed. In the Wilson GF method, vibrational frequencies are obtained from eigenvalues of the matrix product , where depends on masses and molecular geometry and depends on force constants.[9][10]
Quantum mechanics
In quantum mechanics, the harmonic approximation leads to the Schrödinger equation for each normal coordinate. The vibrational energy levels are
where is the vibrational quantum number. The difference between adjacent levels is
For a harmonic oscillator, the basic selection rule is
This selection rule explains why the fundamental vibrational transition is strongest in the ideal harmonic approximation. Overtone transitions such as become possible because real molecules are anharmonic. Anharmonicity also explains hot bands, where transitions begin from already excited vibrational states.
For anharmonic oscillators, vibrational levels may be represented by expansions such as the Dunham expansion. These descriptions account for decreasing level spacings and the approach to molecular dissociation.
Intensities
In an infrared spectrum, the intensity of an absorption band is proportional to the change of the molecular dipole moment with respect to the normal coordinate.[11] A vibration is infrared-active when
In Raman spectroscopy, the intensity depends on the change in polarizability with respect to the normal coordinate:
This distinction explains why infrared and Raman spectra often provide complementary information about the same molecule.
Applications
Molecular vibration is used to interpret infrared and Raman spectra, identify functional groups, determine molecular symmetry, estimate bond strengths, and study molecular structure. It is important in chemical physics, atmospheric spectroscopy, astrochemistry, materials science, plasma diagnostics, and the study of molecular energy transfer.
See also
Table of contents (72 articles)
Index
Full contents
References
- ↑ Landau, L. D.; Lifshitz, E. M. (1976). Mechanics (3rd ed.). Pergamon Press. ISBN 0-08-021022-8. https://archive.org/details/mechanics00land.
- ↑ Hollas, J. M. (1996). Modern Spectroscopy (3rd ed.). John Wiley. p. 21. ISBN 0471965227.
- ↑ Atkins, P. W.; Paula, J. de (2006). Physical Chemistry (8th ed.). New York: W. H. Freeman. pp. 291 and 453. ISBN 0716787598. https://archive.org/details/atkinsphysicalch00pwat/page/291.
- ↑ 4.0 4.1 Hollas, J. M. (1996). Modern Spectroscopy (3rd ed.). John Wiley. p. 77. ISBN 0471965227.
- ↑ 5.0 5.1 Banwell, Colin N.; McCash, Elaine M. (1994). Fundamentals of Molecular Spectroscopy (4th ed.). McGraw Hill. p. 71. ISBN 0-07-707976-0. https://archive.org/details/isbn_9780077079765.
- ↑ Atkins, P. W.; Paula, J. de (2006). Physical Chemistry (8th ed.). New York: W. H. Freeman. p. 460. ISBN 0716787598. https://archive.org/details/atkinsphysicalch00pwat/page/460.
- ↑ Cotton, F. A. (1971). Chemical Applications of Group Theory (2nd ed.). New York: Wiley. ISBN 0471175706.
- ↑ Nakamoto, K. (1997). Infrared and Raman spectra of inorganic and coordination compounds, Part A (5th ed.). New York: Wiley. ISBN 0471163945.
- ↑ 9.0 9.1 Wilson, E. B.; Decius, J. C.; Cross, P. C. (1995). Molecular Vibrations. New York: Dover. ISBN 048663941X. https://archive.org/details/molecularvibrati00wils.
- ↑ Gans, P. (1971). Vibrating Molecules. New York: Chapman and Hall. ISBN 0412102900.
- ↑ Steele, D. (1971). Theory of vibrational spectroscopy. Philadelphia: W. B. Saunders. ISBN 0721685803. https://archive.org/details/theoryofvibratio0000stee.
Further reading
- Sherwood, P. M. A. (1972). Vibrational Spectroscopy of Solids. Cambridge University Press. ISBN 0521084822.