Chemistry:Methyldiphenylphosphine
From HandWiki
Revision as of 11:23, 14 November 2021 by imported>NBrushPhys (change)
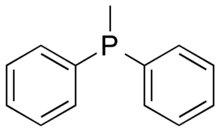
| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| 743075 | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C13H13P | |
| Molar mass | 200.221 g·mol−1 |
| Appearance | colorless liquid |
| Density | 1.0779 |
| Melting point | 117–118 °C (243–244 °F; 390–391 K) |
| Boiling point | 284 °C (543 °F; 557 K) |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H302, H315, H319, H335 | |
| P261, P264, P270, P271, P280, P301+312, P302+352, P304+340, P305+351+338, P312, P321, P330, P332+313, P337+313, P362, P403+233, P405, P501 | |
| Related compounds | |
Related compounds
|
Phenyldimethylphosphine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Methyldiphenylphosphine is the organophosphine with the formula CH3(C6H5)2P, often abbreviated PMePh2. It is a colorless, viscous liquid. It is a member of series (CH3)3-n(C6H5)2P that also includes n = 0, n = 1, and n = 3 that are often employed as ligands in metal phosphine complexes.
Methyldiphenylphosphine is prepared by reaction of chlorodiphenylphosphine with methyl Grignard reagent:
- Cl(C6H5)2P + CH3MgBr → CH3(C6H5)2P + MgBrCl
Selected derivatives:
- The phosphine oxide OPMePh2, prepared by treatment with hydrogen peroxide.[1]
- The coordination complex MoH4(PMePh2)4, prepared by treatment of MoCl4(PMePh2)2 with sodium borohydride in the presence of excess ligand.[2]
- The coordination complex CoCl2(PMePh2)2, prepared by treating cobalt(II) chloride with the phosphine.[3]
- The phosphine-borane H3BPMePh2 prepared by treating the phosphine with borane.[4]
References
- ↑ Denniston, Michael L.; Martin, Donald R. (1977). Methyldiphenylphosphine Oxide and Dimethylphenylphosphine Oxide. Inorganic Syntheses. 17. pp. 183–185. doi:10.1002/9780470132487.ch50. ISBN 9780470132487.
- ↑ Pennella, Filippo (1974). "Tetrahydridotetrakis(methyldiphenylphosphine)molybdenum(IV)". Inorganic Syntheses. Inorganic Syntheses. 15. pp. 42–44. doi:10.1002/9780470132463.ch12. ISBN 9780470132463.
- ↑ Dolcetti, G.; Ghedim, M.; Reed, C. A. (1976). "Bis(Methyldiphenylphosphine)Dichloro-Nitrosylcobalt". Inorganic Syntheses. Inorganic Syntheses. 16. pp. 29–32. doi:10.1002/9780470132470.ch8. ISBN 9780470132470.
- ↑ Mathur, M. A.; Myers, W. H.; Sisler, H. H.; Ryschkewitsch, G. E. (1974). "Methyldiphenylphosphine-Borane and Dimethylphenylphosphine-Borane". Inorganic Syntheses. Inorganic Syntheses. 15. pp. 128–133. doi:10.1002/9780470132463.ch29. ISBN 9780470132463.
 |

