Biology:WH1 domain
| WH1 domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | WH1 | ||||||||
| Pfam | PF00568 | ||||||||
| InterPro | IPR000697 | ||||||||
| SMART | WH1 | ||||||||
| SCOP2 | 1evh / SCOPe / SUPFAM | ||||||||
| CDD | cd01205 | ||||||||
| |||||||||
WH1 domain is an evolutionary conserved protein domain found on WASP proteins, which are often involved in actin polymerization.[1][2]
Function
WH1 domains are important for all cellular processes involving actin, this includes cell motility, cell trafficking, cell division and cytokinesis, cell signalling, and the establishment and maintenance of cell junctions and cell shape.[2]
Structure
Tertiary structure of the WH1 domain of the Mena protein revealed structure similarities with the pleckstrin (PH) domain. The overall fold consists of a compact parallel beta-sandwich, closed along one edge by a long alpha helix. A highly conserved cluster of three surface-exposed aromatic side-chains forms the recognition site for the molecule's target ligands.[3]
Interactions
The WASP protein family control actin polymerization by activating the Arp2/3 complex. WASP is defective in Wiskott–Aldrich syndrome (WAS) whereby in most patient cases, the majority of point mutations occur within the N-terminal WH1 domain. The metabotropic glutamate receptors mGluR1alpha and mGluR5 bind a protein called homer, which is a WH1 domain homologue.[4][5]
WASP family proteins contain an EVH1 (WH1) in their N-terminals which bind proline-rich sequences in the WASP interacting protein. Proteins of the RanBP1 family contain a WH1 domain in their N-terminal region, which seems to bind a different sequence motif present in the C-terminal part of RanGTP protein.[6][7]
Tertiary structure of the WH1 domain of the Mena protein revealed structure similarities with the pleckstrin homology (PH) domain. The overall fold consists of a compact parallel beta-sandwich, closed along one edge by a long alpha-helix. A highly conserved cluster of three surface-exposed aromatic side-chains forms the recognition site for the molecules target ligands.[3]
EVH1
| EVH1 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
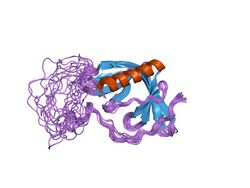 structure of the n-wasp evh1 domain-wip complex | |||||||||
| Identifiers | |||||||||
| Symbol | EVH1 | ||||||||
| Pfam | PF00568 | ||||||||
| Pfam clan | CL0266 | ||||||||
| InterPro | IPR000697 | ||||||||
| SMART | WH1 | ||||||||
| SCOP2 | 1evh / SCOPe / SUPFAM | ||||||||
| CDD | cd00837 | ||||||||
| |||||||||
A subset of WH1 domains has been termed the EVH1 domain bind a polyproline motif. The EVH1 domain (also known as the WH1, RanBP1-WASP, or enabled/VASP homology 1 domain) is also an evolutionary conserved protein domain. EVH1 domains recognise and bind the proline-rich motif FPPPP with low-affinity, further interactions then form between flanking residues.[5][8]
The EVH1 domain is found in multi-domain Ena/Vasp homology proteins implicated in a diverse range of signalling, nuclear transport and cytoskeletal events. This domain of around 115 amino acids is present in species ranging from yeast to mammals. Many EVH1-containing proteins associate with actin-based structures and play a role in cytoskeletal organisation. EVH1 domains recognise and bind the proline-rich motif FPPPP with low-affinity, further interactions then form between flanking residues.[8][9][10]
Examples
Human genes encoding proteins containing the WH1 (EVH1) domain include:
References
- ↑ "Wiskott-Aldrich syndrome protein, a novel effector for the GTPase CDC42Hs, is implicated in actin polymerization". Cell 84 (5): 723–34. March 1996. doi:10.1016/S0092-8674(00)81050-8. PMID 8625410.
- ↑ 2.0 2.1 "WASP family proteins: their evolution and its physiological implications.". Mol Biol Cell 21 (16): 2880–93. 2010. doi:10.1091/mbc.E10-04-0372. PMID 20573979.
- ↑ 3.0 3.1 "Structure of the enabled/VASP homology 1 domain-peptide complex: a key component in the spatial control of actin assembly". Cell 97 (4): 471–80. May 1999. doi:10.1016/S0092-8674(00)80757-6. PMID 10338211.
- ↑ "Identification of homer as a homologue of the Wiskott-Aldrich syndrome protein suggests a receptor-binding function for WH1 domains". J. Mol. Med. 75 (11–12): 769–71. 1997. doi:10.1007/s001090050166. PMID 9428607.
- ↑ 5.0 5.1 "A novel proline-rich motif present in ActA of Listeria monocytogenes and cytoskeletal proteins is the ligand for the EVH1 domain, a protein module present in the Ena/VASP family". EMBO J. 16 (17): 5433–44. September 1997. doi:10.1093/emboj/16.17.5433. PMID 9312002.
- ↑ "EVH1/WH1 domains of VASP and WASP proteins belong to a large family including Ran-binding domains of the RanBP1 family". FEBS Lett. 441 (2): 181–5. December 1998. doi:10.1016/S0014-5793(98)01541-5. PMID 9883880.
- ↑ "The Ran/TC4 GTPase-binding domain: identification by expression cloning and characterization of a conserved sequence motif". Proc. Natl. Acad. Sci. U.S.A. 92 (8): 3328–32. April 1995. doi:10.1073/pnas.92.8.3328. PMID 7724562.
- ↑ 8.0 8.1 "EVH1 domains: structure, function and interactions". FEBS Lett. 513 (1): 45–52. February 2002. doi:10.1016/S0014-5793(01)03291-4. PMID 11911879.
- ↑ Pawson T. "EVH1 Domain". The Pawson Lab. http://pawsonlab.mshri.on.ca/index.php?option=com_content&task=view&Itemid=64&id=217.
- ↑ PDB: 1QC6; "Structure of EVH1, a novel proline-rich ligand-binding module involved in cytoskeletal dynamics and neural function". Nat. Struct. Biol. 6 (7): 661–5. July 1999. doi:10.1038/10717. PMID 10404224.
External links
- Eukaryotic Linear Motif resource motif class LIG_EVH1_1
- Eukaryotic Linear Motif resource motif class LIG_EVH1_2
- Eukaryotic Linear Motif resource motif class LIG_WH1
 |
