Biology:Fibroblast growth factor 21
 Generic protein structure example |
Fibroblast growth factor 21 (FGF-21) is a protein found in humans and other mammals that is encoded by the FGF21 gene.[1][2] This protein is a member of the fibroblast growth factor (FGF) family and its endocrine subfamily along with FGF23 and FGF15/19. FGF21 is the primary endogenous agonist of the FGF21 receptor, which is composed of the FGF receptor and co-receptor β-Klotho.[3]
Members of the FGF family are broad-spectrum mitogens important to survival activities. FGFs are involved in biological processes throughout the body including embryonic development, cell growth, morphogenesis, tissue repair, tumor growth and invasion.[2] FGFs act through a family of four FGF receptors. Binding is complicated and requires both interaction of the FGF molecule with an FGF receptor and binding to heparin through a heparin binding domain. Endocrine FGFs lack a heparin binding domain and thus can be released into the circulation.
FGF21 is a hepatokine, a hormone secreted primarily by the liver as shown by the Potthoff lab.[4][5] Among other activities, FGF21 regulates simple sugar intake and preferences for sweet foods via signaling through FGF21 receptors in the hypothalamus and correlates with reduced dopamine neurotransmission within the nucleus accumbens.[6][7][8] The Potthoff lab followed up on their discovery and later revealed that FGF21 signaling to the ventromedial hypothalamus suppresses sugar intake by enhancing the activity of glucose-excited and glucose-inhibited neurons in response to high, but not low, glucose levels.[9] The Potthoff lab was also the first to demonstrate that FGF21 has direct actions on adipose tissues, where it can increase acute insulin sensitivity and glucose uptake.[10][5] Initially thought of as a starvation hormone, FGF21 is now described as "an endocrine mediator of the intracellular stress response to various nutritional manipulations, including excess sugars and alcohol, caloric deficits, a ketogenic diet, and amino acid restriction".[11]
A single-nucleotide polymorphism of the FGF21 gene – the FGF21 rs838133 variant (frequency 44.7%) – has been identified as a genetic mechanism responsible for the sweet tooth behavioral phenotype, a trait associated with cravings for sweets and high sugar consumption, in both humans and mice.[12][13][14]
Regulation
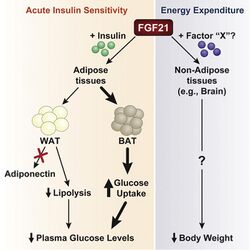
FGF21 has multiple sites of action in both the brain, where it acts to restrain intake of sweets and alcohol, and the periphery, where it acts to increase energy expenditure and body temperature. Initially, FGF21 was thought of as a starvation hormone.[5] FGF21 induction is termed paradoxical because FGF21 is induced both by fasting signals (PPARα and glucagon) and feeding signals (glucose and xylitol). This suggests that FGF21 is involved in adapting to multiple states of nutritional crisis, including both starvation and overfeeding.[5] FGF21 is now described as a "master sensitizer", modulating specific hormonal signals to regulate metabolism and reestablish energy and nutrient homeostasis.[5] A proposed mechanism to resolve paradoxes of FGF21 induction in liver is that glycerol-3-phosphate activates carbohydrate response element binding protein, which can explain induction via weight loss, exercise, ketogenic diet and starvation (via glycerol kinase), mitochondrial disease, NADH shuttle disruption and ethanol (via glycerol-3-phosphate dehydrogenase), and by excess of simple carbohydrates including fructose.[15]
FGF21 is expressed in numerous tissues, including liver, brown adipose tissue (BAT), white adipose tissue (WAT) and pancreas. Circulating levels of FGF21 are derived primarily from the liver in both mice and humans.[5] FGF21 levels in tissues tend to be basally low and expression can be induced by many different signals of nutritional and cellular stress. Response varies according to both tissue and nutritional context.[5][3] Transcriptional regulation is critical for FGF21 expression, but the regulation of circulating FGF21 also depends on mechanisms for the secretion and transport of FGF21.[16]
Several signaling mechanisms for hepatic FGF21 have been described.[5][17] Two of the key transcription factors are Peroxisome proliferator-activated receptor alpha (PPARα) and Carbohydrate-responsive element-binding protein (ChREBP).[5][18][19] In addition, β-Klotho (KLB) is vital for FGFR activation, acting as a co‑receptor for the binding of FGF21.[20][21] The regulation of FGF21 expression in the liver is also mediated by transcription factors such as ATF4 and NRF.[5][18][19]
The FGF21 metabolic pathway responds to signals from adipose tissue, liver, and mitochondria. Hepatogenic FGF21 is released into the bloodstream, and can cross the blood-brain barrier. In the central nervous system (CNS), FGF21 interacts with its receptor complex, which contains both fibroblast growth factor receptor (e.g. FGFR1) and co-receptor (β-Klotho). The FGF21 receptor complex is found in brain regions such as the hypothalamus and hindbrain. Transcription factor PPARα mediates FGF21 secretion in the liver in response to fasting and high-fat diets. PPARα is also an intermediate in the upstream regulation of FGF21. Transcription factor ChREBP can stimulate hepatogenic FGF21 expression under conditions such as high carbohydrate intake. FGF21 mRNA is up-regulated under conditions of protein malnutrition.[16]
In this way signals are transmitted from the peripheral tissues to the brain, where they act in a variety of ways to influence regulation of metabolism and energy balance.[16] Major functions of FGF21 in regulating nutrient and energy homeostasis include the enhancement of insulin sensitivity, increases in energy expenditure and weight loss, decreases in hepatic triglycerides and regulation of macronutrient preferences.[5] Liver-derived FGF21 has been studied under nutritional conditions including fasting, ketogenic diet, protein restriction, high-carbohydrate diet, and consumption of alcohol.[5][17][18][19]
Function
| Parts of this biology (those related to section) need to be updated. Please update this biology to reflect recent events or newly available information. (August 2025) |
FGF21 was first identified as a major metabolic regulator based on its ability to lower glucose and lipid levels in rodents. FGF21 stimulates glucose uptake in adipocytes but not in other cell types.[22] This effect is additive to the activity of insulin. FGF21 treatment of adipocytes is associated with phosphorylation of FRS2, a protein linking FGF receptors to the Ras/MAP kinase pathway. FGF21 injection in ob/ob mice results in an increase in Glut1 in adipose tissue. FGF21 also protects animals from diet-induced obesity when overexpressed in transgenic mice and lowers blood glucose and triglyceride levels when administered to diabetic rodents.[22] Treatment of animals with FGF21 results in increased energy expenditure, fat utilization and lipid excretion.[23]
In cows, plasma FGF21 was nearly undetectable in late pregnancy (LP), peaked at parturition, and then stabilized at lower, chronically elevated concentrations during early lactation (EL). Plasma FGF21 was similarly increased in the absence of parturition when an energy-deficit state was induced by feed restricting late-lactating dairy cows, implicating energy insufficiency as a cause of chronically elevated FGF21 in EL. The liver was the major source of plasma FGF21 in early lactation with little or no contribution by WAT, skeletal muscle, and mammary gland. Meaningful expression of the FGF21 coreceptor β-Klotho was restricted to liver and WAT in a survey of 15 tissues that included the mammary gland. Expression of β-Klotho and its subset of interacting FGF receptors was modestly affected by the transition from LP to EL in liver but not in WAT.[24]
Clinical significance
| Parts of this biology (those related to section) need to be updated. Please update this biology to reflect recent events or newly available information. (August 2025) |
Serum FGF-21 levels were significantly increased in patients with type 2 diabetes mellitus (T2DM) which may indicate a role in the pathogenesis of T2DM.[25] Elevated levels also correlate with liver fat content in non-alcoholic fatty liver disease[26] and positively correlate with BMI in humans suggesting obesity as a FGF21-resistant state.[27]
A single-nucleotide polymorphism (SNP) of the FGF21 gene – the FGF21 rs838133 variant (frequency 44.7%) – has been identified as a genetic mechanism responsible for the sweet tooth behavioral phenotype, a trait associated with cravings for sweets and high sugar consumption, in both humans and mice.[12][13][14]
Animal studies
| Parts of this biology (those related to section) need to be updated. Please update this biology to reflect recent events or newly available information. (August 2025) |
Mice lacking FGF21 fail to fully induce PGC-1α expression in response to a prolonged fast and have impaired gluconeogenesis and ketogenesis.[28]
FGF21 stimulates phosphorylation of fibroblast growth factor receptor substrate 2 and ERK1/2 in the liver. Acute FGF21 treatment induced hepatic expression of key regulators of gluconeogenesis, lipid metabolism, and ketogenesis including glucose-6-phosphatase, phosphoenol pyruvate carboxykinase, 3-hydroxybutyrate dehydrogenase type 1, and carnitine palmitoyltransferase 1α. In addition, injection of FGF21 was associated with decreased circulating insulin and free fatty acid levels. FGF21 treatment induced mRNA and protein expression of PGC-1α, but in mice PGC-1α expression was not necessary for the effect of FGF21 on glucose metabolism.[29]
In mice FGF21 is strongly induced in liver by prolonged fasting via PPAR-alpha and in turn induces the transcriptional coactivator PGC-1α and stimulates hepatic gluconeogenesis, fatty acid oxidation, and ketogenesis. FGF21 also blocks somatic growth and sensitizes mice to a hibernation-like state of torpor, playing a key role in eliciting and coordinating the adaptive starvation response. FGF21 expression is also induced in white adipose tissue by PPAR-gamma, which may indicate it also regulates metabolism in the fed state.[30] FGF21 is induced in both rodents and humans consuming a low protein diet.[31][32] FGF21 expression is also induced by diets with reduced levels of the essential dietary amino acids methionine[33][34] or threonine,[35] or with reduced levels of branched-chain amino acids.[36]
Activation of AMPK and SIRT1 by FGF21 in adipocytes enhanced mitochondrial oxidative capacity as demonstrated by increases in oxygen consumption, citrate synthase activity, and induction of key metabolic genes. The effects of FGF21 on mitochondrial function require serine/threonine kinase 11 (STK11/LKB1), which activates AMPK. Inhibition of AMPK, SIRT1, and PGC-1α activities attenuated the effects of FGF21 on oxygen consumption and gene expression, indicating that FGF21 regulates mitochondrial activity and enhances oxidative capacity through an LKB1-AMPK-SIRT1-PGC-1α-dependent mechanism in adipocytes, resulting in increased phosphorylation of AMPK, increased cellular NAD+ levels and activation of SIRT1 and deacetylation of SIRT1 targets PGC-1α and histone 3.[37]
Acutely, the rise in FGF21 in response to alcohol consumption inhibits further drinking. Chronically, the rise in FGF21 expression in the liver may protect against liver damage.[3]
References
- ↑ "Identification of a novel FGF, FGF-21, preferentially expressed in the liver". Biochimica et Biophysica Acta (BBA) - Gene Structure and Expression 1492 (1): 203–206. June 2000. doi:10.1016/S0167-4781(00)00067-1. PMID 10858549.
- ↑ 2.0 2.1 "Entrez Gene: FGF21 fibroblast growth factor 21". https://www.ncbi.nlm.nih.gov/gene?Db=gene&Cmd=ShowDetailView&TermToSearch=26291.
- ↑ 3.0 3.1 3.2 "Fibroblast Growth Factor 21: A Versatile Regulator of Metabolic Homeostasis.". Annual Review of Nutrition 38: 173–196. May 2018. doi:10.1146/annurev-nutr-071816-064800. PMID 29727594.
- ↑ "Circulating FGF21 is liver derived and enhances glucose uptake during refeeding and overfeeding". Diabetes 63 (12): 4057–63. 2014. doi:10.2337/db14-0595. ISSN 0012-1797. PMID 25008183.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 "Metabolic Messengers: FGF21". Nature Metabolism 3 (3): 309–317. 2021. doi:10.1038/s42255-021-00354-2. ISSN 2522-5812. PMID 33758421.
- ↑ "FGF21 Mediates Endocrine Control of Simple Sugar Intake and Sweet Taste Preference by the Liver". Cell Metabolism 23 (2): 335–343. February 2016. doi:10.1016/j.cmet.2015.12.003. PMID 26724858.
- ↑ "FGF21 Regulates Sweet and Alcohol Preference". Cell Metabolism 23 (2): 344–349. February 2016. doi:10.1016/j.cmet.2015.12.008. PMID 26724861.
- ↑ "FGF21 Is a Sugar-Induced Hormone Associated with Sweet Intake and Preference in Humans". Cell Metabolism 25 (5): 1045–1053.e6. May 2017. doi:10.1016/j.cmet.2017.04.009. PMID 28467924.
- ↑ "FGF21 Signals to Glutamatergic Neurons in the Ventromedial Hypothalamus to suppress Carbohydrate Intake". Cell Metabolism 4 (32): 273–286. August 2020. doi:10.1016/j.cmet.2020.06.008. PMID 32640184.
- ↑ "FGF21 Regulates Metabolism Through Adipose-Dependent and -Independent Mechanisms". Cell Metabolism 4 (25): 935–944. April 2017. doi:10.1016/j.cmet.2017.03.005. PMID 28380381.
- ↑ "Fibroblast Growth Factor 21 Facilitates the Homeostatic Control of Feeding Behavior.". Journal of Clinical Medicine 11 (3): 580. 24 January 2022. doi:10.3390/jcm11030580. ISSN 2077-0383. PMID 35160033.
- ↑ 12.0 12.1 "A Common Allele in FGF21 Associated with Sugar Intake Is Associated with Body Shape, Lower Total Body-Fat Percentage, and Higher Blood Pressure". Cell Reports 23 (2): 327–336. April 2018. doi:10.1016/j.celrep.2018.03.070. PMID 29641994.
- ↑ 13.0 13.1 "Liver: FGF21 - the cause of having a 'sweet tooth'?". Nature Reviews. Endocrinology 13 (7): 378. July 2017. doi:10.1038/nrendo.2017.62. PMID 28497814.
- ↑ 14.0 14.1 "Neuroendocrinology: FGF21 influences a 'sweet tooth' in mice". Nature Reviews. Endocrinology 12 (3): 123. March 2016. doi:10.1038/nrendo.2016.8. PMID 26822924.
- ↑ "Glycerol-3-phosphate activates ChREBP, FGF21 transcription and lipogenesis in citrin deficiency". Nature Metabolism 7 (11): 2284–2299. November 2025. doi:10.1038/s42255-025-01399-3. PMID 41238906.
- ↑ 16.0 16.1 16.2 "Fibroblast growth factor 21: An emerging pleiotropic regulator of lipid metabolism and the metabolic network.". Genes & Diseases 11 (3). May 2024. doi:10.1016/j.gendis.2023.06.033. PMID 38292170.
- ↑ 17.0 17.1 "The Nuanced Metabolic Functions of Endogenous FGF21 Depend on the Nature of the Stimulus, Tissue Source, and Experimental Model" (in English). Frontiers in Endocrinology 12. 3 January 2022. doi:10.3389/fendo.2021.802541. ISSN 1664-2392. PMID 35046901.
- ↑ 18.0 18.1 18.2 "Distinct Fgf21 Expression Patterns in Various Tissues in Response to Different Dietary Regimens Using a Reporter Mouse Model.". Nutrients 17 (7): 1179. 28 March 2025. doi:10.3390/nu17071179. ISSN 2072-6643. PMID 40218937.
- ↑ 19.0 19.1 19.2 "Functions of FGF21 and its role in cardiac hypertrophy". Journal of Advanced Research. 13 March 2025. doi:10.1016/j.jare.2025.03.007. ISSN 2090-1232. PMID 40089060. https://www.sciencedirect.com/science/article/pii/S2090123225001481. Retrieved 6 August 2025.
- ↑ "Aberrant acetylated modification of FGF21‑KLB signaling contributes to hepatocellular carcinoma metastasis through the β‑catenin pathway". International Journal of Oncology 63 (2): 1–13. 1 August 2023. doi:10.3892/ijo.2023.5539. ISSN 1019-6439. PMID 37350415. https://www.spandidos-publications.com/10.3892/ijo.2023.5539?text=fulltext. Retrieved 6 August 2025.
- ↑ "BetaKlotho is required for metabolic activity of fibroblast growth factor 21". Proceedings of the National Academy of Sciences of the United States of America 104 (18): 7432–7437. May 2007. doi:10.1073/pnas.0701600104. PMID 17452648. Bibcode: 2007PNAS..104.7432O.
- ↑ 22.0 22.1 "FGF-21 as a novel metabolic regulator". The Journal of Clinical Investigation 115 (6): 1627–1635. June 2005. doi:10.1172/JCI23606. PMID 15902306. Bibcode: 2005JCliI.115.1627K.
- ↑ "Fibroblast growth factor 21 corrects obesity in mice". Endocrinology 149 (12): 6018–6027. December 2008. doi:10.1210/en.2008-0816. PMID 18687777.
- ↑ "Plasma FGF21 is elevated by the intense lipid mobilization of lactation". Endocrinology 152 (12): 4652–4661. December 2011. doi:10.1210/en.2011-1425. PMID 21990311.
- ↑ "Serum FGF-21 levels in type 2 diabetic patients". Endocrine Research 36 (4): 142–148. 2011. doi:10.3109/07435800.2011.558550. PMID 21973233.
- ↑ "Circulating fibroblast growth factor 21 levels are closely associated with hepatic fat content: a cross-sectional study". PLOS ONE 6 (9). 2011. doi:10.1371/journal.pone.0024895. PMID 21949781. Bibcode: 2011PLoSO...624895Y.
- ↑ "Fibroblast growth factor 21: effects on carbohydrate and lipid metabolism in health and disease". Current Opinion in Clinical Nutrition and Metabolic Care 14 (4): 354–359. July 2011. doi:10.1097/MCO.0b013e328346a326. PMID 21505329.
- ↑ "FGF21 induces PGC-1alpha and regulates carbohydrate and fatty acid metabolism during the adaptive starvation response". Proceedings of the National Academy of Sciences of the United States of America 106 (26): 10853–10858. June 2009. doi:10.1073/pnas.0904187106. PMID 19541642. Bibcode: 2009PNAS..10610853P.
- ↑ "Integrated regulation of hepatic metabolism by fibroblast growth factor 21 (FGF21) in vivo". Endocrinology 152 (8): 2996–3004. August 2011. doi:10.1210/en.2011-0281. PMID 21712364.
- ↑ "Fibroblast growth factor 21: from pharmacology to physiology". The American Journal of Clinical Nutrition 91 (1): 254S–257S. January 2010. doi:10.3945/ajcn.2009.28449B. PMID 19906798.
- ↑ "FGF21 is an endocrine signal of protein restriction". The Journal of Clinical Investigation 124 (9): 3913–3922. September 2014. doi:10.1172/JCI74915. PMID 25133427.
- ↑ "Decreased Consumption of Branched-Chain Amino Acids Improves Metabolic Health". Cell Reports 16 (2): 520–530. July 2016. doi:10.1016/j.celrep.2016.05.092. PMID 27346343.
- ↑ "Methionine restriction restores a younger metabolic phenotype in adult mice with alterations in fibroblast growth factor 21". Aging Cell 13 (5): 817–827. October 2014. doi:10.1111/acel.12238. PMID 24935677.
- ↑ "Short-term methionine deprivation improves metabolic health via sexually dimorphic, mTORC1-independent mechanisms". FASEB Journal 32 (6): 3471–3482. January 2018. doi:10.1096/fj.201701211R. PMID 29401631.
- ↑ "Restriction of essential amino acids dictates the systemic metabolic response to dietary protein dilution". Nature Communications 11 (1). 2020-06-09. doi:10.1038/s41467-020-16568-z. ISSN 2041-1723. PMID 32518324. Bibcode: 2020NatCo..11.2894Y.
- ↑ "Restoration of metabolic health by decreased consumption of branched-chain amino acids". The Journal of Physiology 596 (4): 623–645. February 2018. doi:10.1113/JP275075. PMID 29266268.
- ↑ "Fibroblast growth factor 21 regulates energy metabolism by activating the AMPK-SIRT1-PGC-1alpha pathway". Proceedings of the National Academy of Sciences of the United States of America 107 (28): 12553–12558. July 2010. doi:10.1073/pnas.1006962107. PMID 20616029. Bibcode: 2010PNAS..10712553C.
External links
- "FGF21". Gene Cards. Weizmann Institute of Science. https://www.genecards.org/cgi-bin/carddisp.pl?gene=FGF21.
- Got a sweet tooth? Blame your liver Phys.org, 2017
This article incorporates text from the United States National Library of Medicine, which is in the public domain.
 |
