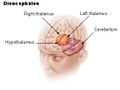
Biology:Hypothalamus
The hypothalamus (pl.: hypothalami; from grc ὑπό (hupó) 'under', and θάλαμος (thálamos) 'chamber') is a small part of the vertebrate brain that contains a number of nuclei with a variety of functions. One of the most important functions is to link the nervous system to the endocrine system via the pituitary gland. The hypothalamus is located below the thalamus and is part of the limbic system.[1] It forms the basal part of the diencephalon. All vertebrate brains contain a hypothalamus.[2] In humans, it is about the size of an almond.[3]
The hypothalamus has the function of regulating certain metabolic processes and other activities of the autonomic nervous system. It synthesizes and secretes certain neurohormones, called releasing hormones or hypothalamic hormones, and these in turn stimulate or inhibit the secretion of hormones from the pituitary gland. The hypothalamus controls body temperature, hunger, important aspects of parenting and maternal attachment behaviours, thirst,[4] fatigue, sleep, circadian rhythms, and is important in certain social behaviors, such as sexual and aggressive behaviors.[5][6]
Structure
The hypothalamus is divided into four regions (preoptic, supraoptic, tuberal, mammillary) in a parasagittal plane, indicating location anterior-posterior; and three zones (periventricular, intermediate, lateral) in the coronal plane, indicating location medial-lateral.[7] Hypothalamic nuclei are located within these specific regions and zones.[8] It is found in all vertebrate nervous systems. In mammals, magnocellular neurosecretory cells in the paraventricular nucleus and the supraoptic nucleus of the hypothalamus produce neurohypophysial hormones, oxytocin and vasopressin.[9] These hormones are released into the blood in the posterior pituitary.[10] Much smaller parvocellular neurosecretory cells, neurons of the paraventricular nucleus, release corticotropin-releasing hormone and other hormones into the hypophyseal portal system, where these hormones diffuse to the anterior pituitary.[citation needed]
Nuclei
The hypothalamic nuclei include the following:[11][12]
| Region | Area | Nucleus | Function[13] |
| Anterior (supraoptic) | Preoptic | Preoptic nucleus | |
| Ventrolateral preoptic nucleus | Sleep | ||
| Medial | Medial preoptic nucleus |
| |
| Supraoptic nucleus |
| ||
| Paraventricular nucleus |
| ||
| Anterior hypothalamic nucleus |
| ||
| Suprachiasmatic nucleus |
| ||
| Lateral | Lateral nucleus | See Lateral hypothalamus § Function – primary source of orexin neurons that project throughout the brain and spinal cord | |
| Middle (tuberal) | Medial | Dorsomedial hypothalamic nucleus |
|
| Ventromedial nucleus |
| ||
| Arcuate nucleus | |||
| Lateral | Lateral nucleus | See Lateral hypothalamus § Function – primary source of orexin neurons that project throughout the brain and spinal cord | |
| Lateral tuberal nuclei | |||
| Posterior (mammillary) | Medial | Mammillary nuclei (part of mammillary bodies) | |
| Posterior nucleus |
| ||
| Lateral | Lateral nucleus | See Lateral hypothalamus § Function – primary source of orexin neurons that project throughout the brain and spinal cord | |
| Tuberomammillary nucleus[17] |
-
Cross-section of the monkey hypothalamus displays two of the major hypothalamic nuclei on either side of the fluid-filled third ventricle.
-
Hypothalamic nuclei
-
Hypothalamic nuclei on one side of the hypothalamus, shown in a 3-D computer reconstruction[18]
Connections
The hypothalamus is highly interconnected with other parts of the central nervous system, in particular the brainstem and its reticular formation. As part of the limbic system, it has connections to other limbic structures including the amygdala and septum, and is also connected with areas of the autonomous nervous system. Lua error: not enough memory.
The hypothalamus receives many inputs from the brainstem, the most notable from the nucleus of the solitary tract, the locus coeruleus, and the ventrolateral medulla. Lua error: not enough memory.
Most nerve fibres within the hypothalamus run in two ways (bidirectional).
- Projections to areas caudal to the hypothalamus go through the medial forebrain bundle, the mammillotegmental tract and the dorsal longitudinal fasciculus.
- Projections to areas rostral to the hypothalamus are carried by the mammillothalamic tract, the fornix and terminal stria.
- Projections to areas of the sympathetic motor system (lateral horn spinal segments T1–L2/L3) are carried by the hypothalamospinal tract and they activate the sympathetic motor pathway.
Sexual dimorphism
Several hypothalamic nuclei are sexually dimorphic; i.e., there are clear differences in both structure and function between males and females.[19] Some differences are apparent even in gross neuroanatomy: most notable is the sexually dimorphic nucleus within the preoptic area,[19] in which the differences are subtle changes in the connectivity and chemical sensitivity of particular sets of neurons. The importance of these changes can be recognized by functional differences between males and females. For instance, males of most species prefer the odor and appearance of females over males, which is instrumental in stimulating male sexual behavior. If the sexually dimorphic nucleus is lesioned, this preference for females by males diminishes. Also, the pattern of secretion of growth hormone is sexually dimorphic;[20] this is why in many species, adult males are visibly distinct sizes from females.
Responsiveness to ovarian steroids
Dimorphism is also found in physiological and behavioral responses to ovarian steroids in adults, where males and females respond to these hormones differently. For example, estrogen receptor sensitivity for different sets of neurons is dimorphic already early on in development[21]. Hypothalamic dimorphism underlies some known behavioral differences in mice[22], and has known physiological effects in humans, e.g. affecting thermoregulation[21] and metabolism[23]. Although human hypothalami exhibit various sex differences[24], it is not certain which behaviors are caused, predisposed, and not caused by these[25][26]. In addition to confounding environmental factors[27], the hypothalamus also contributes to dimorphic human behaviors where the hypothalamus does not itself cause dimorphism, but rather exhibits conditional, dimorphic responses as part of greater pathways, such as the HPG-axis[28][Note 1] or the HPA-axis[29][30][Note 2].
Male and female brains differ in the distribution of estrogen receptors; this is widely assumed[31] to be caused by neonatal estradiol exposure, with some mechanisms being proven[32], however the complete underlying mechanism remains uncertain[25]. Estrogen and progesterone receptors show differential expression where they are found in neurons of the anterior and mediobasal hypothalamus, notably:
- the preoptic area, where LHRH neurons are located, regulating dopamine responses and maternal behavior;[33]
- the periventricular nucleus, where somatostatin neurons are located, regulating stress levels;[34]
- the ventromedial hypothalamus, which regulates hunger and sexual arousal.
Development
In neonatal life, gonadal steroids are thought to influence the development of the hypothalamus. For instance, they correlate with the ability of females to exhibit a normal reproductive cycle, and of males and females to display appropriate reproductive behaviors in adult life:
- If a female rat is given testosterone in the first few days of postnatal life, during the "critical period" of sex-steroid influence in rats, the hypothalamus is irreversibly defeminized and masculinized; the adult rat will be incapable of generating an LH surge in response to estrogen as is characteristic of females, but will be capable of exhibiting male sexual behaviors e.g. mounting a sexually receptive female.[35]
- By contrast, a male rat castrated just after birth will be feminized, and the adult will show typical female "receptive" sexual behavior in response to estrogen, that is, lordosis behavior.[35]
- Masculinization and feminization can be distinguished from their complimentary de-feminization and de-masculinization, as neonatal treatment with COX2 inhibitors or PgE2 makes it possible to create rats which exhibit neither sexual behaviour, or both, respectively[25]. Some effects of combined masculinization and feminization on hypothalamic physiology are known[25][36], but outcomes where the processes oppose (e.g. proportions of cell types) remain unreported in vitro as of 2025.
In primates, the developmental influence of androgens is less clear, and the consequences are less understood. Within the brain, testosterone is aromatized (to estradiol), which is the principal active hormone for developmental influences. The human testis secretes high levels of testosterone from about week eight of fetal life until five to six months after birth (a similar perinatal surge in testosterone is observed in many species), a process that appears to underlie the male phenotype. Estrogen from the maternal circulation is relatively ineffective, partly because of the high circulating levels of steroid-binding proteins in pregnancy.[35]
Sex steroids are not the only important influences upon hypothalamic development; in particular, pre-pubertal stress in early life (of rats) determines the capacity of the adult hypothalamus to respond to an acute stressor.[37] Unlike gonadal steroid receptors, glucocorticoid receptors are very widespread throughout the brain; in the paraventricular nucleus, they mediate negative feedback control of CRF synthesis and secretion, but elsewhere their role is not well understood.
Function
Hormone release
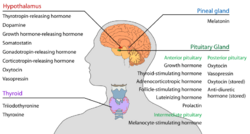
The hypothalamus has a central neuroendocrine function, most notably by its control of the anterior pituitary, which in turn regulates various endocrine glands and organs. Releasing hormones (also called releasing factors) are produced in hypothalamic nuclei then transported along axons to either the median eminence or the posterior pituitary, where they are stored and released as needed.[38]
- Anterior pituitary
In the hypothalamic–adenohypophyseal axis, releasing hormones, also known as hypophysiotropic or hypothalamic hormones, are released from the median eminence, a prolongation of the hypothalamus, into the hypophyseal portal system, which carries them to the anterior pituitary where they exert their regulatory functions on the secretion of adenohypophyseal hormones.[39] These hypophysiotropic hormones are stimulated by parvocellular neurosecretory cells located in the periventricular area of the hypothalamus. After their release into the capillaries of the third ventricle, the hypophysiotropic hormones travel through what is known as the hypothalamo-pituitary portal circulation. Once they reach their destination in the anterior pituitary, these hormones bind to specific receptors located on the surface of pituitary cells. Depending on which cells are activated through this binding, the pituitary will either begin secreting or stop secreting hormones into the rest of the bloodstream.[40]
| Secreted hormone | Abbreviation | Produced by | Effect |
|---|---|---|---|
| Thyrotropin-releasing hormone (Prolactin-releasing hormone) |
TRH, TRF, or PRH | Parvocellular neurosecretory cells of the paraventricular nucleus | Stimulate thyroid-stimulating hormone (TSH) release from anterior pituitary (primarily) Stimulate prolactin release from anterior pituitary |
| Corticotropin-releasing hormone | CRH or CRF | Parvocellular neurosecretory cells of the paraventricular nucleus | Stimulate adrenocorticotropic hormone (ACTH) release from anterior pituitary |
| Dopamine (Prolactin-inhibiting hormone) |
DA or PIH | Dopamine neurons of the arcuate nucleus | Inhibit prolactin release from anterior pituitary |
| Growth-hormone-releasing hormone | GHRH | Neuroendocrine neurons of the Arcuate nucleus | Stimulate growth-hormone (GH) release from anterior pituitary |
| Gonadotropin-releasing hormone | GnRH or LHRH | Neuroendocrine cells of the Preoptic area | Stimulate follicle-stimulating hormone (FSH) release from anterior pituitary Stimulate luteinizing hormone (LH) release from anterior pituitary |
| Somatostatin[41] (growth-hormone-inhibiting hormone) |
SS, GHIH, or SRIF | Neuroendocrine cells of the Periventricular nucleus | Inhibit growth-hormone (GH) release from anterior pituitary Inhibit (moderately) thyroid-stimulating hormone (TSH) release from anterior pituitary |
Other hormones secreted from the median eminence include vasopressin, oxytocin, and neurotensin.[42][43][44][45]
- Posterior pituitary
In the hypothalamic–pituitary–adrenal axis, neurohypophysial hormones are released from the posterior pituitary, which is actually a prolongation of the hypothalamus, into the circulation.
| Secreted hormone | Abbreviation | Produced by | Effect |
|---|---|---|---|
| Oxytocin | OXY or OXT | Magnocellular neurosecretory cells of the paraventricular nucleus and supraoptic nucleus | Uterine contraction Lactation (letdown reflex) |
| Vasopressin (antidiuretic hormone) |
ADH or AVP | Magnocellular and parvocellular neurosecretory cells of the paraventricular nucleus, magnocellular cells in supraoptic nucleus | Increase in the permeability to water of the cells of distal tubule and collecting duct in the kidney and thus allows water reabsorption and excretion of concentrated urine |
It is also known that hypothalamic–pituitary–adrenal axis (HPA) hormones are related to certain skin diseases and skin homeostasis. There is evidence linking hyperactivity of HPA hormones to stress-related skin diseases and skin tumors.[46]
Stimulation
The hypothalamus coordinates many hormonal and behavioural circadian rhythms, complex patterns of neuroendocrine outputs, complex homeostatic mechanisms, and important behaviours. The hypothalamus must, therefore, respond to many different signals, some of which are generated externally and some internally. Delta wave signalling arising either in the thalamus or in the cortex influences the secretion of releasing hormones; GHRH and prolactin are stimulated whilst TRH is inhibited. Lua error: Internal error: The interpreter exited with status 1.
The hypothalamus is responsive to:
- Light: daylength and photoperiod for regulating circadian and seasonal rhythms
- Olfactory stimuli, including pheromones
- Steroids, including gonadal steroids and corticosteroids
- Neurally transmitted information arising in particular from the heart, enteric nervous system (of the gastrointestinal tract),[47] and the reproductive tract. * Autonomic inputs
- Blood-borne stimuli, including leptin, ghrelin, angiotensin, insulin, pituitary hormones, cytokines, plasma concentrations of glucose and osmolarity etc.
- Stress
- Invading microorganisms by increasing body temperature, resetting the body's thermostat upward.
Olfactory stimuli
Olfactory stimuli are important for sexual reproduction and neuroendocrine function in many species. For instance, if a pregnant mouse is exposed to the urine of a 'strange' male during a critical period after coitus then the pregnancy fails (the Bruce effect). Thus, during coitus, a female mouse forms a precise 'olfactory memory' of her partner that persists for several days. Pheromonal cues aid synchronization of oestrus in many species; in women, synchronized menstruation may also arise from pheromonal cues, although the role of pheromones in humans is disputed. Lua error: Internal error: The interpreter exited with status 1.
Blood-borne stimuli
Findings have suggested that thyroid hormone (T4) is taken up by the hypothalamic glial cells in the infundibular nucleus/ median eminence, and that it is here converted into T3 by the type 2 deiodinase (D2). Subsequent to this, T3 is transported into the thyrotropin-releasing hormone (TRH)-producing neurons in the paraventricular nucleus. Thyroid hormone receptors have been found in these neurons, indicating that they are indeed sensitive to T3 stimuli. In addition, these neurons expressed MCT8, a thyroid hormone transporter, supporting the theory that T3 is transported into them. T3 could then bind to the thyroid hormone receptor in these neurons and affect the production of thyrotropin-releasing hormone, thereby regulating thyroid hormone production.[48]
The hypothalamus functions as a type of thermostat for the body.[49] It sets a desired body temperature, and stimulates either heat production and retention to raise the blood temperature to a higher setting or sweating and vasodilation to cool the blood to a lower temperature. All fevers result from a raised setting in the hypothalamus; elevated body temperatures due to any other cause are classified as hyperthermia.[49] Rarely, direct damage to the hypothalamus, such as from a stroke, will cause a fever; this is sometimes called a hypothalamic fever. However, it is more common for such damage to cause abnormally low body temperatures.[49]
Steroids
The hypothalamus contains neurons that react strongly to steroids and glucocorticoids (the steroid hormones of the adrenal gland, released in response to ACTH). It also contains specialized glucose-sensitive neurons (in the arcuate nucleus and ventromedial hypothalamus), which are important for appetite. The preoptic area contains thermosensitive neurons; these are important for TRH secretion. Lua error: Internal error: The interpreter exited with status 1.
Neural
Oxytocin secretion in response to suckling or vagino-cervical stimulation is mediated by some of these pathways; vasopressin secretion in response to cardiovascular stimuli arising from chemoreceptors in the carotid body and aortic arch, and from low-pressure atrial volume receptors, is mediated by others. In the rat, stimulation of the vagina also causes prolactin secretion, and this results in pseudo-pregnancy following an infertile mating. In the rabbit, coitus elicits reflex ovulation. In the sheep, cervical stimulation in the presence of high levels of estrogen can induce maternal behavior in a virgin ewe. These effects are all mediated by the hypothalamus, and the information is carried mainly by spinal pathways that relay in the brainstem. Stimulation of the nipples stimulates release of oxytocin and prolactin and suppresses the release of LH and FSH. Lua error: Internal error: The interpreter exited with status 1.
Cardiovascular stimuli are carried by the vagus nerve. The vagus also conveys a variety of visceral information, including for instance signals arising from gastric distension or emptying, to suppress or promote feeding, by signalling the release of leptin or gastrin, respectively. Again, this information reaches the hypothalamus via relays in the brainstem. Lua error: Internal error: The interpreter exited with status 1.
In addition, hypothalamic function is responsive to—and regulated by—levels of all three classical monoamine neurotransmitters, noradrenaline, dopamine, and serotonin (5-hydroxytryptamine), in those tracts from which it receives innervation. For example, noradrenergic inputs arising from the locus coeruleus have important regulatory effects upon corticotropin-releasing hormone (CRH) levels. Lua error: Internal error: The interpreter exited with status 1.
Control of food intake
| Peptides that increase feeding behavior |
Peptides that decrease feeding behavior |
|---|---|
| Ghrelin | Leptin |
| Neuropeptide Y | (α,β,γ)-Melanocyte-stimulating hormones |
| Agouti-related peptide | Cocaine- and amphetamine-regulated transcript peptides |
| Orexins (A,B) | Corticotropin-releasing hormone |
| Melanin-concentrating hormone | Cholecystokinin |
| Galanin | Insulin |
| Glucagon-like peptide 1 |
The extreme lateral part of the ventromedial nucleus of the hypothalamus is responsible for the control of food intake. Stimulation of this area causes increased food intake. Bilateral lesion of this area causes complete cessation of food intake. Medial parts of the nucleus have a controlling effect on the lateral part. Bilateral lesion of the medial part of the ventromedial nucleus causes hyperphagia and obesity of the animal. Further lesion of the lateral part of the ventromedial nucleus in the same animal produces complete cessation of food intake.
There are different hypotheses related to this regulation:[51]
- Lipostatic hypothesis: This hypothesis holds that adipose tissue produces a humoral signal that is proportionate to the amount of fat and acts on the hypothalamus to decrease food intake and increase energy output. It has been evident that a hormone leptin acts on the hypothalamus to decrease food intake and increase energy output.
- Gutpeptide hypothesis: gastrointestinal hormones like Grp, glucagons, CCK and others claimed to inhibit food intake. The food entering the gastrointestinal tract triggers the release of these hormones, which act on the brain to produce satiety. The brain contains both CCK-A and CCK-B receptors.
- Glucostatic hypothesis: The activity of the satiety center in the ventromedial nuclei is probably governed by the glucose utilization in the neurons. It has been postulated that when their glucose utilization is low and consequently when the arteriovenous blood glucose difference across them is low, the activity across the neurons decrease. Under these conditions, the activity of the feeding center is unchecked and the individual feels hungry. Food intake is rapidly increased by intraventricular administration of 2-deoxyglucose therefore decreasing glucose utilization in cells.
- Thermostatic hypothesis: According to this hypothesis, a decrease in body temperature below a given set-point stimulates appetite, whereas an increase above the set-point inhibits appetite.
Fear processing
The medial zone of hypothalamus is part of a circuitry that controls motivated behaviors, like defensive behaviors.[52] Analyses of Fos-labeling showed that a series of nuclei in the "behavioral control column" is important in regulating the expression of innate and conditioned defensive behaviors.[53]
- Antipredatory defensive behavior
Exposure to a predator (such as a cat) elicits defensive behaviors in laboratory rodents, even when the animal has never been exposed to a cat.[54] In the hypothalamus, this exposure causes an increase in Fos-labeled cells in the anterior hypothalamic nucleus, the dorsomedial part of the ventromedial nucleus, and in the ventrolateral part of the premammillary nucleus (PMDvl).[55] The premammillary nucleus has an important role in expression of defensive behaviors towards a predator, since lesions in this nucleus abolish defensive behaviors, like freezing and flight.[55][56] The PMD does not modulate defensive behavior in other situations, as lesions of this nucleus had minimal effects on post-shock freezing scores.[56] The PMD has important connections to the dorsal periaqueductal gray, an important structure in fear expression.[57][58] In addition, animals display risk assessment behaviors to the environment previously associated with the cat. Fos-labeled cell analysis showed that the PMDvl is the most activated structure in the hypothalamus, and inactivation with muscimol prior to exposure to the context abolishes the defensive behavior.[55] Therefore, the hypothalamus, mainly the PMDvl, has an important role in expression of innate and conditioned defensive behaviors to a predator.
- Social defeat
Likewise, the hypothalamus has a role in social defeat: nuclei in medial zone are also mobilized during an encounter with an aggressive conspecific. The defeated animal has an increase in Fos levels in sexually dimorphic structures, such as the medial pre-optic nucleus, the ventrolateral part of ventromedial nucleus, and the ventral premammilary nucleus.[6] Such structures are important in other social behaviors, such as sexual and aggressive behaviors. Moreover, the premammillary nucleus also is mobilized, the dorsomedial part but not the ventrolateral part.[6] Lesions in this nucleus abolish passive defensive behavior, like freezing and the "on-the-back" posture.[6]
Learning arbitrator
Recent research has questioned whether the lateral hypothalamus's role is only restricted to initiating and stopping innate behaviors and argued it learns about food-related cues. Specifically, that it opposes learning about information what is neutral or distant to food. According this view, the lateral hypothalamus is "a unique arbitrator of learning capable of shifting behavior toward or away from important events".[59]
Additional images
-
Human brain left dissected midsagittal view
-
Location of the hypothalamus
See also
- Ventrolateral preoptic nucleus
- Periventricular nucleus
- Copeptin
- Hypothalamic–pituitary–adrenal axis (HPA axis)
- Hypothalamic–pituitary–gonadal axis (HPG axis)
- Hypothalamic–pituitary–thyroid axis (HPT axis)
- Incertohypothalamic pathway
- Neuroendocrinology
- Neuroscience of sleep
Notes
- ↑ Sex hormones are directly involved in the feedback loop of the HPG axis.
- ↑ Sex hormones are not directly involved in the HPA axis, but nevertheless alter how the hypothalamus responds within the pathway.
Lua error: Internal error: The interpreter exited with status 1.
References
- ↑ "The Emotional Nervous System". http://webspace.ship.edu/cgboer/limbicsystem.html.
- ↑ "The hypothalamus predates the origin of vertebrates". Science Advances 7 (18). April 2021. doi:10.1126/sciadv.abf7452. PMID 33910896. Bibcode: 2021SciA....7.7452L.
- ↑ "Metabolic and Non-Cognitive Manifestations of Alzheimer's Disease: The Hypothalamus as Both Culprit and Target of Pathology". Cell Metabolism 22 (5): 761–776. November 2015. doi:10.1016/j.cmet.2015.08.016. PMID 26365177.
- ↑ "NCI Dictionary of Cancer Terms". https://www.cancer.gov/publications/dictionaries/cancer-terms.
- ↑ "Hypothalamic regulation of sleep and circadian rhythms". Nature 437 (7063): 1257–1263. October 2005. doi:10.1038/nature04284. PMID 16251950. Bibcode: 2005Natur.437.1257S.
- ↑ 6.0 6.1 6.2 6.3 "Dissecting the brain's fear system reveals the hypothalamus is critical for responding in subordinate conspecific intruders". Proceedings of the National Academy of Sciences of the United States of America 106 (12): 4870–5. March 2009. doi:10.1073/pnas.0900939106. PMID 19273843. Bibcode: 2009PNAS..106.4870M.
- ↑ Textbook of Clinical Neuroanatomy (2nd ed.). Elsevier Health Sciences. 2014. pp. 134. ISBN 9788131229811. https://books.google.com/books?id=LdCGBAAAQBAJ&dq=hypothalamus+regions++supraoptic%2C+tuberal%2C+mammillary&pg=PA134.
- ↑ Inderbir Singh (September 2011). Textbook of Anatomy: Volume 3: Head and Neck, Central Nervous System. JP Medical Ltd. pp. 1101–. ISBN 978-93-5025-383-0. https://books.google.com/books?id=8NJYL4ixFZQC&pg=PA1101.
- ↑ "Vasopressin and oxytocin gene expression in the human hypothalamus". The Journal of Comparative Neurology 337 (2): 295–306. November 1993. doi:10.1002/cne.903370210. PMID 8277003.
- ↑ Williams Textbook of Endocrinology (12th ed.). Saunders. 2011. pp. 107. ISBN 978-1437703245. https://www.elsevier.com/books/williams-textbook-of-endocrinology/melmed/978-1-4377-0324-5.
- ↑ "Enlarged view of the hypothalamus". Jim Phelps. http://www.psycheducation.org/emotion/pics/big%20hypothalamus.htm.
- ↑ "Emotion and the limbic system". Lucien T. "Tres" Thompson, The University of Texas at Dallas. http://www.utdallas.edu/~tres/integ/hom3/display13_04.html.
- ↑ Guyton and Hall Textbook of Medical Physiology (12th ed.). Saunders/Elsevier. 2011. ISBN 978-1416045748.
- ↑ "Parallel preoptic pathways for thermoregulation". The Journal of Neuroscience 29 (38): 11954–64. September 2009. doi:10.1523/JNEUROSCI.2643-09.2009. PMID 19776281.
- ↑ "Dysfunctions of the paraventricular hypothalamic nucleus induce hypersomnia in mice". eLife 10. November 2021. doi:10.7554/eLife.69909. PMID 34787078.
- ↑ "Case Report: Dysfunction of the Paraventricular Hypothalamic Nucleus Area Induces Hypersomnia in Patients" (in English). Frontiers in Neuroscience 16. 2022-03-14. doi:10.3389/fnins.2022.830474. PMID 35360167.
- ↑ "Chapter 6: Widely Projecting Systems: Monoamines, Acetylcholine, and Orexin". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. 2009. pp. 175–176. ISBN 9780071481274. "Within the brain, histamine is synthesized exclusively by neurons with their cell bodies in the tuberomammillary nucleus (TMN) that lies within the posterior hypothalamus. There are approximately 64000 histaminergic neurons per side in humans. These cells project throughout the brain and spinal cord. Areas that receive especially dense projections include the cerebral cortex, hippocampus, neostriatum, nucleus accumbens, amygdala, and hypothalamus. ... While the best characterized function of the histamine system in the brain is regulation of sleep and arousal, histamine is also involved in learning and memory ... It also appears that histamine is involved in the regulation of feeding and energy balance."
- ↑ Brain Research Bulletin 35:323–327, 1994
- ↑ 19.0 19.1 "The sexually dimorphic nucleus of the preoptic area in the human brain: a comparative morphometric study". Journal of Anatomy 164: 55–72. June 1989. PMID 2606795.
- ↑ "Neural growth hormone: Regional regulation by estradiol and / or sex chromosome complement in male and female mice". Biology of Sex Differences 6. 2015. doi:10.1186/s13293-015-0026-x. PMID 25987976.
- ↑ 21.0 21.1 van Veen, J. Edward; Kammel, Laura G.; Bunda, Patricia C.; Shum, Michael; Reid, Michelle S.; Massa, Megan G.; Arneson, Douglas V.; Park, Jae W. et al. (2020-04-13). "Hypothalamic oestrogen receptor alpha establishes a sexually dimorphic regulatory node of energy expenditure" (in en). Nature Metabolism 2 (4): 351–363. doi:10.1038/s42255-020-0189-6. ISSN 2522-5812. PMID 32377634.
- ↑ Spiteri, Thierry; Musatov, Sergei; Ogawa, Sonoko; Ribeiro, Ana; Pfaff, Donald W.; Agmo, Anders (2010). "Estrogen-induced sexual incentive motivation, proceptivity and receptivity depend on a functional estrogen receptor alpha in the ventromedial nucleus of the hypothalamus but not in the amygdala". Neuroendocrinology 91 (2): 142–154. doi:10.1159/000255766. ISSN 1423-0194. PMID 19887773.
- ↑ McArthur, Simon; McHale, Emily; Gillies, Glenda E. (July 2007). "The size and distribution of midbrain dopaminergic populations are permanently altered by perinatal glucocorticoid exposure in a sex- region- and time-specific manner". Neuropsychopharmacology: Official Publication of the American College of Neuropsychopharmacology 32 (7): 1462–1476. doi:10.1038/sj.npp.1301277. ISSN 0893-133X. PMID 17164817.
- ↑ Cosgrove, Kelly P.; Mazure, Carolyn M.; Staley, Julie K. (2007-10-15). "Evolving knowledge of sex differences in brain structure, function, and chemistry". Biological Psychiatry 62 (8): 847–855. doi:10.1016/j.biopsych.2007.03.001. ISSN 0006-3223. PMID 17544382.
- ↑ 25.0 25.1 25.2 25.3 McCarthy, Margaret M. (2009-09-29). "Estradiol and the developing brain". Physiological Reviews 88 (1): 91–124. doi:10.1152/physrev.00010.2007. ISSN 0031-9333. PMID 18195084.
- ↑ Gillies, Glenda E.; McArthur, Simon (June 2010). "Estrogen actions in the brain and the basis for differential action in men and women: a case for sex-specific medicines". Pharmacological Reviews 62 (2): 155–198. doi:10.1124/pr.109.002071. ISSN 1521-0081. PMID 20392807.
- ↑ Shepard, Kathryn N.; Michopoulos, Vasiliki; Toufexis, Donna J.; Wilson, Mark E. (2009-05-25). "Genetic, epigenetic and environmental impact on sex differences in social behavior". Physiology & Behavior 97 (2): 157–170. doi:10.1016/j.physbeh.2009.02.016. ISSN 1873-507X. PMID 19250945.
- ↑ Wibral, Matthias; Dohmen, Thomas; Klingmüller, Dietrich; Weber, Bernd; Falk, Armin (2012-10-10). Krueger, Frank. ed. "Testosterone Administration Reduces Lying in Men" (in en). PLOS ONE 7 (10): e46774. doi:10.1371/journal.pone.0046774. ISSN 1932-6203. PMID 23071635. Bibcode: 2012PLoSO...746774W.
- ↑ Heck, Ashley L.; Handa, Robert J. (2018-08-01). "Sex differences in the hypothalamic–pituitary–adrenal axis' response to stress: an important role for gonadal hormones" (in en). Neuropsychopharmacology 44 (1): 45–58. doi:10.1038/s41386-018-0167-9. ISSN 1740-634X. PMID 30111811. https://www.nature.com/articles/s41386-018-0167-9.
- ↑ Sofer, Yael; Osher, Esther; Abu Ahmad, Wiessam; Yacobi Bach, Michal; Even Zohar, Naomi; Zaid, Dana; Golani, Nehama; Moshe, Yaffa et al. (2024). "Gender-affirming hormone therapy effect on cortisol levels in trans males and trans females" (in en). Clinical Endocrinology 100 (2): 164–169. doi:10.1111/cen.14985. ISSN 1365-2265. PMID 37933843. https://onlinelibrary.wiley.com/doi/abs/10.1111/cen.14985.
- ↑ McCarthy, Margaret M.; Konkle, Anne T. M. (2005-09-01). "When is a sex difference not a sex difference?". Frontiers in Neuroendocrinology 26 (2): 85–102. doi:10.1016/j.yfrne.2005.06.001. ISSN 0091-3022. https://www.sciencedirect.com/science/article/pii/S0091302205000282.
- ↑ Todd, Brigitte J.; Schwarz, Jaclyn M.; McCarthy, Margaret M. (December 2005). "Prostaglandin-E2: a point of divergence in estradiol-mediated sexual differentiation". Hormones and Behavior 48 (5): 512–521. doi:10.1016/j.yhbeh.2005.07.011. ISSN 0018-506X. PMID 16126205.
- ↑ "Luteinizing hormone-releasing hormone distribution in the anterior hypothalamus of the female rats". ISRN Anatomy 2013: 1–6. 2013. doi:10.5402/2013/870721. PMID 25938107.
- ↑ "Estrogen receptor beta in the paraventricular nucleus of hypothalamus regulates the neuroendocrine response to stress and is regulated by corticosterone". Neuroscience 121 (4): 837–45. 2003. doi:10.1016/S0306-4522(03)00561-X. PMID 14580933.
- ↑ 35.0 35.1 35.2 "Sex differences in the brain: the not so inconvenient truth". The Journal of Neuroscience 32 (7): 2241–7. February 2012. doi:10.1523/JNEUROSCI.5372-11.2012. PMID 22396398.
- ↑ Speert, Debra B.; Konkle, Anne T. M.; Zup, Susan L.; Schwarz, Jaclyn M.; Shiroor, Chaitanya; Taylor, Michael E.; McCarthy, Margaret M. (July 2007). "Focal adhesion kinase and paxillin: novel regulators of brain sexual differentiation?". Endocrinology 148 (7): 3391–3401. doi:10.1210/en.2006-0845. ISSN 0013-7227. PMID 17412802.
- ↑ "Stress history and pubertal development interact to shape hypothalamic–pituitary–adrenal axis plasticity". Endocrinology 147 (4): 1664–74. April 2006. doi:10.1210/en.2005-1432. PMID 16410296.
- ↑ "Overview of Hypothalamic and Pituitary Hormones". http://www.vivo.colostate.edu/hbooks/pathphys/endocrine/hypopit/overview.html.
- ↑ "Disorders of the anterior pituitary and hypothalamus". Harrison's Principles of Internal Medicine (16th ed.). New York, NY: McGraw-Hill. 2005. pp. 2076–97. ISBN 978-0-07-139140-5. https://archive.org/details/harrisonsprincip00kasp.
- ↑ "Hypothalamic Control of the Anterior Pituitary". Neuroscience: Exploring the Brain (4th ed.). Philadelphia: Wolters Kluwer. 2016. p. 528. ISBN 978-0-7817-7817-6.
- ↑ "Pituitary somatostatin receptor signaling". Trends in Endocrinology and Metabolism 21 (3): 123–33. March 2010. doi:10.1016/j.tem.2009.12.003. PMID 20149677.
- ↑ "Oxytocin and vasopressin in rat hypophysial portal blood: experimental studies in normal and Brattleboro rats". The Journal of Endocrinology 104 (2): 211–24. February 1985. doi:10.1677/joe.0.1040211. PMID 3968510.
- ↑ "Distribution of orexin/hypocretin in the rat median eminence and pituitary". Brain Research. Molecular Brain Research 76 (1): 1–6. March 2000. doi:10.1016/s0169-328x(99)00317-4. PMID 10719209.
- ↑ "In vivo release of neurotensin from the median eminence of ovariectomized estrogen-primed rats as estimated by push-pull perfusion: correlation with luteinizing hormone and prolactin surges". Neuroendocrinology 57 (4): 760–4. April 1993. doi:10.1159/000126434. PMID 8367038.
- ↑ "Orexins in the regulation of the hypothalamic–pituitary–adrenal axis". Pharmacological Reviews 58 (1): 46–57. March 2006. doi:10.1124/pr.58.1.4. PMID 16507882.
- ↑ "Expression of Hypothalamic–Pituitary–Adrenal Axis in Common Skin Diseases: Evidence of its Association with Stress-related Disease Activity". National Research Foundation of Korea. 2013. http://web.b.ebscohost.com/ehost/pdfviewer/pdfviewer?sid=8239d5d4-8cd4-48b0-b25c-e1218229f462%40sessionmgr115&vid=11&hid=122.
- ↑ "Gut feelings: the emerging biology of gut-brain communication". Nature Reviews. Neuroscience 12 (8): 453–66. July 2011. doi:10.1038/nrn3071. PMID 21750565.
- ↑ "Functional neuroanatomy of thyroid hormone feedback in the human hypothalamus and pituitary gland". Molecular and Cellular Endocrinology 251 (1–2): 1–8. June 2006. doi:10.1016/j.mce.2006.03.042. PMID 16707210.
- ↑ 49.0 49.1 49.2 Fauci, Anthony (2008). Harrison's Principles of Internal Medicine (17 ed.). McGraw-Hill Professional. pp. 117–121. ISBN 978-0-07-146633-2. https://archive.org/details/harrisonsprincip00asfa.
- ↑ "Chapter 10: Neural and Neuroendocrine Control of the Internal Milieu – Table 10:3". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. 2009. p. 263. ISBN 9780071481274.
- ↑ "Anorexia-producing intermediary metabolites". The American Journal of Clinical Nutrition 29 (5): 552–8. May 1976. doi:10.1093/ajcn/29.5.552. PMID 178168.
- ↑ "Cerebral hemisphere regulation of motivated behavior". Brain Research 886 (1–2): 113–164. December 2000. doi:10.1016/S0006-8993(00)02905-X. PMID 11119693.
- ↑ Canteras, N.S. (2002). "The medial hypothalamic defensive system:Hodological organization and functional implications". Pharmacology Biochemistry and Behavior 71 (3): 481–491. doi:10.1016/S0091-3057(01)00685-2. PMID 11830182.
- ↑ "An alternative experimental procedure for studying predator-related defensive responses". Neuroscience and Biobehavioral Reviews 29 (8): 1255–63. 2005. doi:10.1016/j.neubiorev.2005.04.006. PMID 16120464.
- ↑ 55.0 55.1 55.2 "Hypothalamic sites responding to predator threats--the role of the dorsal premammillary nucleus in unconditioned and conditioned antipredatory defensive behavior". The European Journal of Neuroscience 28 (5): 1003–15. September 2008. doi:10.1111/j.1460-9568.2008.06392.x. PMID 18691328.
- ↑ 56.0 56.1 Blanchard, D.C. (2003). "Dorsal premammillary nucleus differentially modulates defensive behaviors induced by different threat stimuli in rats". Neuroscience Letters 345 (3): 145–148. doi:10.1016/S0304-3940(03)00415-4. PMID 12842277.
- ↑ "The dorsal premammillary nucleus: an unusual component of the mammillary body". Proceedings of the National Academy of Sciences of the United States of America 89 (21): 10089–93. November 1992. doi:10.1073/pnas.89.21.10089. PMID 1279669. Bibcode: 1992PNAS...8910089C.
- ↑ "Functional characteristics of the midbrain periaqueductal gray". Progress in Neurobiology 46 (6): 575–605. August 1995. doi:10.1016/0301-0082(95)00009-K. PMID 8545545.
- ↑ "The cognitive (lateral) hypothalamus". Trends in Cognitive Sciences 28 (1): 18–29. January 2024. doi:10.1016/j.tics.2023.08.019. PMID 37758590.
Lua error: Internal error: The interpreter exited with status 1.
Further reading
- "Sex differences in the brain: the relation between structure and function". Hormones and Behavior 55 (5): 589–96. May 2009. doi:10.1016/j.yhbeh.2009.03.012. PMID 19446075.
External links
- Stained brain slice images which include the "Hypothalamus" at the BrainMaps project
- The Hypothalamus and Pituitary at endotexts.org
- NIF Search - Hypothalamus via the Neuroscience Information Framework
- Space-filling and cross-sectional diagrams of hypothalamic nuclei: right hypothalamus, anterior, tubular, posterior.
Lua error: Internal error: The interpreter exited with status 1. Lua error: Internal error: The interpreter exited with status 1.
Lua error: Internal error: The interpreter exited with status 1.


![Hypothalamic nuclei on one side of the hypothalamus, shown in a 3-D computer reconstruction[18]](/wiki/images/thumb/0/04/3D-Hypothalamus.JPG/120px-3D-Hypothalamus.JPG)