Biology:Haplogroup R-M269
| Haplogroup R-M269 | |
|---|---|
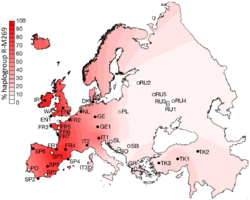 Projected spatial frequency distribution for haplogroup R-M269 in Europe.[1] | |
| Possible time of origin | 4,000–10,000 BP[2][3] |
| Possible place of origin | Eastern Europe, associated with Indo-European migrations[4] |
| Ancestor | R1b1a1a (R-P297) |
| Descendants | L23; L51/M412, L151/P310; Z2103 |
| Defining mutations | M269 |
Haplogroup R-M269 is the sub-clade of human Y-chromosome haplogroup R1b that is defined by the SNP marker M269. According to ISOGG 2020 it is phylogenetically classified as R1b1a1b. It underwent intensive research and was previously classified as R1b1a2 (2003 to 2005), R1b1c (2005 to 2008), R1b1b2 (2008 to 2011) and R1b1a1a2 (2011 to 2020).[5]
R-M269 is of particular interest for the genetic history of Western Europe, being the most common European haplogroup. It increases in frequency on an east to west gradient (its prevalence in Poland estimated at 22.7%, compared to Wales at 92.3%). It is carried by approximately 110 million European men (2010 estimate).[3] The age of the mutation M269 is estimated at 4,000 to 10,000 years ago.[2][3]
Origin
R-M269 had formerly been dated to the Upper Paleolithic,[6] but by about 2010 it was thought to have formed near the beginning of the Neolithic Revolution, about 10,000 years ago.[7][8][9] More recent archaeogenetics studies since 2015, however, strongly suggest an origin among Eneolithic hunter-gatherers from eastern Europe.[4][10]
Balaresque et al. (2010) based on the pattern of Y-STR diversity argued for a single source in the Near East and introduction to Europe via Anatolia in the Neolithic Revolution. In this scenario, Mesolithic hunter-gatherers in Europe would have been nearly replaced by the incoming farmers. By contrast, Busby et al. (2012) could not confirm the results of Balaresque et al. (2010) and could not make credible estimates of the age of R-M269 based on Y-STR diversity.[3][11] Furthermore, more recent studies have found that the Y-DNA of Early European Farmers is typically haplogroup G2a.[12]
According to a 2015 study,[4] a hunter-gatherer from Samara (dated 5640-5555 cal BCE) belonging to haplogroup R1b1(*) was ancestral for both haplogroups R-M269 and R-M478. According to the authors, the occurrence of basal forms of R1b in eastern European hunter-gatherers provides a "geographically plausible source" for haplogroup R-M269. Subclades of R-M269, such as R-Z2103, have been found to be prevalent in ancient DNA found in individuals associated with the Yamnaya culture and related populations,[4][13] and the dispersal of this haplogroup is associated with the spread of so-called "steppe ancestry" and at least some of the Indo-European languages.[4][14]
The subclade R-P311 is substantially confined to Western Europe in modern populations. R-P311 is absent from Neolithic-era ancient DNA found in Western Europe, strongly suggesting that its current distribution is due to population movements within Europe taking place after the end of the Neolithic. The three major subclades of P311 are U106 (S21), L21 (M529, S145), and U152 (S28). These show a clear articulation within Western Europe, with centers in the Low Countries, the British Isles and the Alps, respectively.[15] These lineages are associated with the non-Iberian steppe-related groups of the Bell Beaker culture, and demonstrate the relationship between steppe-related ancestry and R1b-M269 subclades,[13] which are "the major lineage associated with the arrival of Steppe ancestry in western Europe after 2500 BC".[16]
Distribution
This article's factual accuracy is disputed. (August 2020) (Learn how and when to remove this template message) |
European R1b is dominated by R-M269. It has been found at generally low frequencies throughout central Eurasia,[17] but with relatively high frequency among the Bashkirs of the Perm region (84.0%) and Baymaksky District (81.0%).[18] This marker is present in China and India at frequencies of less than one percent. The table below lists in more detail the frequencies of M269 in regions in Asia, Europe, and Africa.
Distribution of R-M269 in Europe increases in frequency from east to west. It peaks at the national level in Wales at a rate of 92%, at 82% in Ireland, 70% in Scotland, 68% in Spain , 60% in France (76% in Normandy), about 60% in Portugal,[19] 50% in Germany , 50% in the Netherlands, 47% in Italy,[20] 45% in Eastern England , 43% in Denmark and 42% in Iceland. It is as high as 95% in parts of Ireland. It is also found in some areas of North Africa, where its frequency peaks at 10% in some parts of Algeria.[21] M269 has likewise been observed among 8% of the Herero in Namibia.[22] The R-M269 subclade has been found in ancient Guanche (Bimbapes) fossils excavated in Punta Azul, El Hierro, Canary Islands, which are dated to the 10th century (~44%).[23] In western Asia, R-M269 has been reported in 29.2% of Assyrian males from Iran.[24] Haplogroup R1b1 and its subclades in Asia.[25] M269* (xL23) is found at highest frequency in the central Balkans notably Kosovo with 7.9%, North Macedonia 5.1% and Serbia 4.4%.[19] Kosovo is notable in having a high percentage of descendant L23* or L23(xM412) at 11.4% unlike most other areas with significant percentages of M269* and L23* except for Poland with 2.4% and 9.5% and the Bashkirs of southeast Bashkortostan with 2.4% and 32.2% respectively.[19] Notably this Bashkir population also has a high percentage of M269 sister branch M73 at 23.4%.[19] Five individuals out of 110 tested in the Ararat Valley, Armenia belonged to R1b1a2* and 36 to L23*, with none belonging to known subclades of L23.[26] Trofimova et al. (2015) found a surprising high frequency of R1b-L23 (Z2105/2103) among the peoples of the Idel-Ural. 21 out of 58 (36.2%) of Burzyansky District Bashkirs, 11 out of 52 (21.2%) of Udmurts, 4 out of 50 (8%) of Komi, 4 out of 59 (6.8%) of Mordvins, 2 out of 53 (3.8%) of Besermyan and 1 out of 43 (2.3%) of Chuvash were R1b-L23 (Z2105/2103),[27] the type of R1b found in the recently analyzed Yamna remains of the Samara Oblast and Orenburg Oblast.[28]
Especially Western European R1b is dominated by specific sub-clades of R-M269 (with some small amounts of other types found in areas such as Sardinia[19][29]). Within Europe, R-M269 is dominated by R-M412, also known as R-L51, which according to Myres et al. (2010) is "virtually absent in the Near East, the Caucasus and West Asia." This Western European population is further divided between R-P312/S116 and R-U106/S21, which appear to spread from the western and eastern Rhine river basin respectively. Myres et al. note further that concerning its closest relatives, in R-L23*, it is "instructive" that these are often more than 10% of the population in the Caucasus, Turkey, and some southeast European and circum-Uralic populations.
In Western Europe it is present but in generally much lower levels apart from "an instance of 27% in Switzerland's Upper Rhone Valley."[19] In addition, the sub-clade distribution map, Figure 1h titled "L11(xU106,S116)", in Myres et al. shows that R-P310/L11* (or as yet undefined subclades of R-P310/L11) occurs only in frequencies greater than 10% in Central England with surrounding areas of England and Wales having lower frequencies.[19] This R-P310/L11* is almost non-existent in the rest of Eurasia and North Africa with the exception of coastal lands fringing the western and southern Baltic (reaching 10% in Eastern Denmark and 6% in northern Poland) and in Eastern Switzerland and surrounds.[19]
| M269 (R1b1a1a2)[30] |
| ||||||||||||||||||||||||||||||||||||||||||
In 2009, DNA extracted from the femur bones of 6 skeletons in an early-medieval burial place in Ergolding (Bavaria, Germany) dated to around c. 670 yielded the following results: 4 were found to be haplogroup R1b with the closest matches in modern populations of Germany, Ireland and the USA while 2 were in Haplogroup G2a.[31]
Population studies which test for M269 have become more common in recent years, while in earlier studies men in this haplogroup are only visible in the data by extrapolation of what is likely. The following gives a summary of most of the studies which specifically tested for M269, showing its distribution (as a percentage of total population) in Europe, North Africa, the Middle East and Central Asia as far as China and Nepal.
| Country | Sampling | sample | R-M269 | Source |
|---|---|---|---|---|
| Wales | National | 65 | 92.3% | Balaresque et al. (2009)[3] |
| Spain | Basques | 116 | 87.1% | Balaresque et al. (2009)[3] |
| Ireland | National | 796 | 85.4% | Moore et al. (2006)[32] |
| Spain | Catalonia | 80 | 81.3% | Balaresque et al. (2009)[3] |
| Italy | Lombardy | 78 | 80.8% | Grugni et al.[20] |
| France | Ille-et-Vilaine | 82 | 80.5% | Balaresque et al. (2009)[3] |
| France | Haute-Garonne | 57 | 78.9% | Balaresque et al. (2009)[3] |
| England | Cornwall | 64 | 78.1% | Balaresque et al. (2009)[3] |
| France | Loire-Atlantique | 48 | 77.1% | Balaresque et al. (2009)[3] |
| Italy | Tuscany | 42 | 76.2% | Di Giacomo et al. (2003)[33] |
| France | Finistère | 75 | 76.0% | Balaresque et al. (2009)[3] |
| France | Basques | 61 | 75.4% | Balaresque et al. (2009)[3] |
| Italy | North East | 30 | 73.5% | Di Giacomo et al. (2003)[33] |
| Spain | East Andalucia | 95 | 72.0% | Balaresque et al. (2009)[3] |
| Spain | Castilla La Mancha | 63 | 72.0% | Balaresque et al. (2009)[3] |
| France | Vendée | 50 | 68.0% | Balaresque et al. (2009)[3] |
| Dominican Republic | National | 26 | 65.4% | Bryc et al. (2010)[34] |
| France | Baie de Somme | 43 | 62.8% | Balaresque et al. (2009)[3] |
| England | Leicestershire | 43 | 62.0% | Balaresque et al. (2009)[3] |
| Italy | North-East (Ladin) | 79 | 60.8% | Balaresque et al. (2009)[3] |
| Portugal | National | 657 | 59.9% | Beleza et al. (2006)[35] |
| Italy | Emilia | 29 | 58.5% | Boattini et al. (2013)[36] |
| Spain | Galicia | 88 | 58.0% | Balaresque et al. (2009)[3] |
| Spain | West Andalucia | 72 | 55.0% | Balaresque et al. (2009)[3] |
| Portugal | South | 78 | 46.2% | Balaresque et al. (2009)[3] |
| Italy | North-West | 99 | 45.0% | Balaresque et al. (2009)[3] |
| Denmark | National | 56 | 42.9% | Balaresque et al. (2009)[3] |
| Netherlands | National | 84 | 42.0% | Balaresque et al. (2009)[3] |
| Armenia | Ararat Valley | 41 | 37.3% | Herrera et al. (2012)[26] |
| Russia | Bashkirs | 471 | 34.40% | Lobov (2009)[18] |
| Italy | East Sicily | 246 | 34.14% | Tofanelli et al. (2015)[37] |
| Italy | West Sicily | 68 | 33.0% | Tofanelli et al. (2015)[37] |
| Germany | Bavaria | 80 | 32.3% | Balaresque et al. (2009)[3] |
| Turkey | Lake Van Armenians | 33 | 32.0% | Herrera et al. (2012) [26] |
| Armenia | Gardman | 30 | 31.3% | Herrera et al. (2012) [26] |
| Iran | Assyrians | 48 | 29.2% | Grugni,Viola et al. (2012)[24] |
| Poland | National | 110 | 22.7% | Myres et al. (2007)[38] |
| Slovenia | National | 75 | 21.3% | Battaglia et al. (2008)[39] |
| Kosovo Albanians | National | 114 | 21.1% | Pericic2005[40] |
| Slovenia | National | 70 | 20.6% | Balaresque et al. (2009)[3] |
| Turkey | Central | 152 | 19.1% | Cinnioğlu et al. (2004)[41] |
| Albanians in North Macedonia | National | 64 | 18.8% | Battaglia et al. (2008)[39] |
| Albanians | National | 55 | 18.2% | Battaglia et al. (2008)[39] |
| Crete | National | 193 | 17.0% | King et al. (2008)[42] |
| Italy | Sardinia | 930 | 17.0% | Contu et al. (2008)[43] |
| Turkey | Sasun Armenians | 16 | 15.4% | Herrera et al. (2012) [26] |
| Iran | North | 33 | 15.2% | Regueiro et al. (2006)[44] |
| Moldova | 268 | 14.6% | Varzari (2006)[45] | |
| Greece | National | 171 | 13.5% | King et al. (2008)[42] |
| Turkey | West | 163 | 13.5% | Cinnioğlu et al. (2004)[41] |
| Romania | National | 54 | 13.0% | Varzari (2006)[45] |
| Croatia | National | 89 | 12.4% | Battaglia et al. (2008)[39] |
| Turkey | East | 208 | 12.0% | Cinnioğlu et al. (2004)[41] |
| Algeria | Northwest (Oran area) | 102 | 11.8% | Robino et al. (2008)[46] |
| Russia | Roslavl (Smolensk Oblast) | 107 | 11.2% | Balanovsky et al. (2008)[47] |
| Iraq | National | 139 | 10.8% | Al-Zahery et al. (2003)[48] |
| Nepal | Newar | 66 | 10.6% | Gayden et al. (2007)[49] |
| Bulgaria | National | 808 | 10.5% | Karachanak et al. (2013)[50] |
| Serbia | National | 100 | 10.0% | Belaresque et al. (2009)[3] |
| Lebanon | National | 914 | 7.3% | Zalloua et al. (2008)[51] |
| Tunisia | National | 601 | 0.3% | Bekada et al. (2013)[52] |
| Tunisia | Tunis | 139 | 7.2% | Adams et al. (2008)[53] |
| Morocco | National | 760 | 3.5% | Bekada et al. (2013)[52] |
| Libya | National | 83 | 0.0% | Bekada et al. (2013)[52] |
| Egypt | National | 360 | 2.9% | Bekada et al. (2013)[52] |
| Algeria | National | 156 | 7.0% | Bekada et al. (2013)[52] |
| Algeria | Algiers, Tizi Ouzou | 46 | 6.5% | Adams et al. (2008)[53] |
| Bosnia-Herzegovina | Serbs | 81 | 6.2% | Marjanovic et al. (2005)[54] |
| Iran | South | 117 | 6.0% | Regueiro et al. (2006)[44] |
| Russia | Repyevka (Voronezh Oblast) | 96 | 5.2% | Balanovsky et al. (2008)[47] |
| UAE | 164 | 3.7% | Cadenas et al. (2007)[55] | |
| Bosnia-Herzegovina | Bosniaks | 85 | 3.5% | Marjanovic et al. (2005)[54] |
| Pakistan | 176 | 2.8% | Sengupta et al. (2006)[56] | |
| Russia | Belgorod | 143 | 2.8% | Balanovsky et al. (2008)[47] |
| Russia | Ostrov (Pskov Oblast) | 75 | 2.7% | Balanovsky et al. (2008)[47] |
| Russia | Pristen (Kursk Oblast) | 45 | 2.2% | Balanovsky et al. (2008)[47] |
| Bosnia-Herzegovina | Croats | 90 | 2.2% | Marjanovic et al. (2005)[54] |
| Qatar | 72 | 1.4% | Cadenas et al. (2007)[55] | |
| China | 128 | 0.8% | Sengupta et al. (2006)[56] | |
| India | various | 728 | 0.5% | Sengupta et al. (2006)[56] |
| Croatia | Osijek | 29 | 0.0% | Battaglia et al. (2008)[39] |
| Yemen | 62 | 0.0% | Cadenas et al. (2007)[55] | |
| Tibet | 156 | 0.0% | Gayden et al. (2007)[49] | |
| Nepal | Tamang | 45 | 0.0% | Gayden et al. (2007)[49] |
| Nepal | Kathmandu | 77 | 0.0% | Gayden et al. (2007)[49] |
| Japan | 23 | 0.0% | Sengupta et al. (2006)[56] |
Sub-clades
This article's factual accuracy is disputed. (August 2020) (Learn how and when to remove this template message) |
R1b1a1a2a (R-L23)
R-L23* (R1b1a1a2a*) is now most commonly found in Europe, Anatolia, the Caucasus.
R1b1a1a2a1 (R-L51)
R-L51* (R1b1a1a2a1*) is now concentrated in a geographical cluster centred on southern France and northern Italy.
R1b1a1a2a1a (R-L151)
R-L151 (L151/PF6542, CTS7650/FGC44/PF6544/S1164, L11, L52/PF6541, P310/PF6546/S129, P311/PF6545/S128) also known as R1b1a1a2a1, and its subclades, include most males with R1b in Western Europe.
R1b1a1a2a1a1 (R-U106)
This subclade is defined by the presence of the SNP U106, also known as S21 and M405.[7][57] It appears to represent over 25% of R1b in Europe.[7] In terms of percentage of total population, its epicenter is Friesland, where it makes up 44% of the population.[58] In terms of total population numbers, its epicenter is Central Europe, where it comprises 60% of R1 combined.[58] See also Haplogroup R-Z18
| U106/S21/M405 |
| |||||||||||||||||||||||||||
While this sub-clade of R1b is frequently discussed amongst genetic genealogists, the following table represents the peer-reviewed findings published so far in the 2007 articles of Myres et al. and Sims et al.[38][57]
| Population | Sample size | R-M269 | R-U106 | R-U106-1 |
|---|---|---|---|---|
| Austria[38] | 22 | 27% | 23% | 0.0% |
| Central/South America[38] | 33 | 0.0% | 0.0% | 0.0% |
| Czech Republic[38] | 36 | 28% | 14% | 0.0% |
| Denmark[38] | 113 | 34% | 17% | 0.9% |
| Eastern Europe[38] | 44 | 5% | 0.0% | 0.0% |
| England[38] | 138 | 57% | 20% | 1.4% |
| France[38] | 56 | 52% | 7% | 0.0% |
| Germany[38] | 332 | 43% | 19% | 1.8% |
| Ireland[38] | 102 | 80% | 6% | 0.0% |
| Italy[19] | 34 | 53% | 6% | 0.0% |
| Jordan[38] | 76 | 0.0% | 0.0% | 0.0% |
| Middle-East[38] | 43 | 0.0% | 0.0% | 0.0% |
| Netherlands[38] | 94 | 54% | 35% | 2.1% |
| Oceania[38] | 43 | 0.0% | 0.0% | 0.0% |
| Oman[38] | 29 | 0.0% | 0.0% | 0.0% |
| Pakistan[38] | 177 | 3% | 0.0% | 0.0% |
| Palestine[38] | 47 | 0.0% | 0.0% | 0.0% |
| Poland[38] | 110 | 23% | 8% | 0.0% |
| Russia[38] | 56 | 21% | 5.4% | 1.8% |
| Slovenia[38] | 105 | 17% | 4% | 0.0% |
| Switzerland[38] | 90 | 58% | 13% | 0.0% |
| Turkey[38] | 523 | 14% | 0.4% | 0.0% |
| Ukraine[38] | 32 | 25% | 9% | 0.0% |
| United States (European)[57] | 125 | 46% | 15% | 0.8% |
| United States (Afroamerican)[57] | 118 | 14% | 2.5% | 0.8% |
R-P312
R1b1a1a2a1a2, better known as R-P312 (or R-S116) is one of the most common types of R-M269 in Europe, alongside R-U106. Myres et al. described it as originating in and spreading from the west of the Rhine basin.[19]
R-P312 has been the subject of significant, ongoing study concerning its complex internal structure.
| P312 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
R-DF27
R-M153
R-M153 is a subclade of R-DF27 that has been found mostly in Basques and Gascons, among whom it represents a sizeable fraction of the Y-DNA pool,[53][59] though is also found occasionally among Iberians in general. The first time it was located (Bosch 2001[60]) it was described as H102 and included seven Basques and one Andalusian.
R-M167
R-M167 is a subclade of R-DF27 defined by the presence of the marker M167. The first author to test for this marker (long before current haplogroup nomenclature existed) was Hurles in 1999, who tested 1158 men in various populations.[61] He found it relatively common among Basques (13/117: 11%) and Catalans (7/32: 22%). Other occurrences were found among other French, British, Spaniards, Béarnais, and Germany .
In 2000 Rosser et al., in a study which tested 3616 men in various populations[62] also tested for that same marker, naming the haplogroup Hg22, and again it was found mainly among Basques (19%), in lower frequencies among French (5%), Bavarians (3%), Spaniards (2%), Southern Portuguese (2%), and in single occurrences among Romanians, Slovenians, Dutch, Belgians and English.::In 2001 Bosch described this marker as H103, in 5 Basques and 5 Catalans.[60] Further regional studies have located it in significant amounts in Asturias, Cantabria and Galicia, as well as again among Basques.[60] Cases in the Azores have been reported.[citation needed] In 2008 two research papers by López-Parra[59] and Adams,[53] respectively, confirmed a strong association with all or most of the Pyrenees and Eastern Iberia.
In a larger study of Portugal in 2006, with 657 men tested, Beleza et al. confirmed similar low levels in all the major regions, from 1.5%–3.5%.[35]
R-L165
This subclade is defined by the presence of the marker S68, also known as L165. It is found in England, Scandinavia, and Scotland (in this country it is mostly found in the Northern Isles and Outer Hebrides). It has been suggested, therefore, that it arrived in the British Isles with Vikings.[63]
R-U152
R-U152 is defined by the presence of the marker U152, also called S28.[7] Its existence was confirmed by Sims et al. (2007).[57] Myres et al. report this clade "is most frequent (20–44%) in Switzerland, Italy, France and Western Poland, with additional instances exceeding 15% in some regions of England and Germany."[38] Similarly Cruciani et al. (2010)[64] reported frequency peaks in Northern and Central Italy and France. Out of a sample of 135 men in Tyrol, Austria, 9 tested positive for U152/S28.[65] Far removed from this apparent core area, Myres et al. also mention a sub-population in north Bashkortostan, where 71% of 70 men tested belong to R-U152. They propose this to be the result of an isolated founder effect.[19] King et al. (2014) reported four living descendants of Henry Somerset, 5th Duke of Beaufort in the male line tested positive for U-152.[66] Ancient samples from the central European Bell Beaker, Hallstatt and Tumulus cultures belonged to this subclade.[13][67][68] Analyzed Iron Age Latins and Etruscans dating between 1000 and 100 BCE belonged primarily to haplogroup R1b-U152 (including the clades L2, Z56 and Z193).[69][70]
R-L21
R-L21 is also known as R-M529 and R-S145.[7] Myres et al. report it is most common in Ireland, Scotland and Wales (25–50% of the whole male population).[19]
R-L159.2 This subclade within R-L21 is defined by the presence of the marker L159 and is known as L159.2 because of a parallel mutation that exists inside haplogroup I2a1 (L159.1). L159.2 appears to be associated with the Kings of Leinster and Diarmait Mac Murchada; Irish Gaels belonging to the Laigin. It can be found in the coastal areas of the Irish Sea including the Isle of Man and the Hebrides, as well as Norway , western and southern Scotland, northern and southern England , northwest France , and northern Denmark .[71]
R-L193 This subclade within R-L21 is defined by the presence of the marker L193. Many surnames with this marker are associated geographically with the western "Border Region" of Scotland. A few other surnames have a Highland association. R-L193 is a relatively young subclade likely born within the last 2000 years.
R-L226 This subclade within R-L21 is defined by the presence of the marker L226, also known as S168. Commonly referred to as Irish Type III, it is concentrated in central western Ireland and associated with the Dál gCais kindred.[72]
R-DF21 This subclade within R-L21 is defined by the presence of the marker DF21 aka S192. It makes up about 10% of all L21 men and is c.3000 years old.[73]
R-L371 This subclade within R-L21 is defined by the presence of the marker L371, referred to as the Welsh modal and associated with ancient Welsh Kings and Princes.[74][75][76]
See also
- Y-DNA haplogroups in populations of Europe
- Haplogroup R1a
References
- ↑ Balaresque et al. (2010), figure 1B: "Geographical distribution of haplogroup frequency of hgR1b1b2, shown as an interpolated spatial frequency surface. Filled circles indicate populations for which microsatellite data and TMRCA estimates are available. Unfilled circles indicate populations included to illustrate R1b1b2 frequency only. Population codes are defined in Table 1."
- ↑ 2.0 2.1 "Mean estimates for individual populations vary (Table 2), but the oldest value is in Central Turkey (7,989 y [95% confidence interval (CI): 5,661–11,014]), and the youngest in Cornwall (5,460 y [3,764–7,777]). The mean estimate for the entire dataset is 6,512 y (95% CI: 4,577–9,063 years), with a growth rate of 1.95% (1.02%–3.30%). Thus, we see clear evidence of rapid expansion, which cannot have begun before the Neolithic period." Balaresque et al. (2010).
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 3.16 3.17 3.18 3.19 3.20 3.21 3.22 3.23 3.24 3.25 3.26 3.27 "A predominantly neolithic origin for European paternal lineages". PLOS Biology 8 (1): e1000285. January 2010. doi:10.1371/journal.pbio.1000285. PMID 20087410.
- ↑ 4.0 4.1 4.2 4.3 4.4 "Massive migration from the steppe was a source for Indo-European languages in Europe". Nature 522 (7555): 207–11. June 2015. doi:10.1038/nature14317. PMID 25731166. Bibcode: 2015Natur.522..207H.
- ↑ dates according to the ISOGG trees for each respective year.
- ↑ "The genetic legacy of Paleolithic Homo sapiens sapiens in extant Europeans: a Y chromosome perspective". Science 290 (5494): 1155–9. November 2000. doi:10.1126/science.290.5494.1155. PMID 11073453. Bibcode: 2000Sci...290.1155S.
- ↑ 7.0 7.1 7.2 7.3 7.4 International Society of Genetic Genealogy (ISOGG) – Y-DNA Haplogroup R and its Subclades
- ↑ Crawford, Michael H., ed (2007). "The peopling of Europe". Anthropological genetics: theory, methods and applications. Cambridge, UK: Cambridge University Press. p. 394. ISBN 978-0-521-54697-3. https://archive.org/details/anthropologicalg00craw.
- ↑ "Strong intra- and inter-continental differentiation revealed by Y chromosome SNPs M269, U106 and U152". Forensic Science International. Genetics 5 (3): e49–52. June 2011. doi:10.1016/j.fsigen.2010.07.006. PMID 20732840.
- ↑ "Population genomics of Bronze Age Eurasia". Nature 522 (7555): 167–172. June 2015. doi:10.1038/nature14507. PMID 26062507. Bibcode: 2015Natur.522..167A. https://depot.ceon.pl/handle/123456789/13155.
- ↑ "The peopling of Europe and the cautionary tale of Y chromosome lineage R-M269". Proceedings. Biological Sciences 279 (1730): 884–892. March 2012. doi:10.1098/rspb.2011.1044. PMID 21865258.
- ↑ "Genome-wide patterns of selection in 230 ancient Eurasians". Nature 528 (7583): 499–503. December 2015. doi:10.1038/nature16152. PMID 26595274. Bibcode: 2015Natur.528..499M.
- ↑ 13.0 13.1 13.2 "The Beaker phenomenon and the genomic transformation of northwest Europe". Nature 555 (7695): 190–196. March 2018. doi:10.1038/nature25738. PMID 29466337. Bibcode: 2018Natur.555..190O.
- ↑ "Ancient DNA, Mating Networks, and the Anatolian Split". Dispersals and Diversification. BRILL. 2019-12-06. pp. 21–53. doi:10.1163/9789004416192_003. ISBN 978-90-04-41619-2.
- ↑ "Origins of R-M269 Diversity in Europe". FamilyTreeDNA 9th Annual Conference. 2013.
- ↑ "Kinship and social organization in Copper Age Europe. A cross-disciplinary analysis of archaeology, DNA, isotopes, and anthropology from two Bell Beaker cemeteries". PLOS ONE 15 (11): e0241278. 2019-12-11. doi:10.1101/863944. PMID 33196640.
- ↑ "Y chromosome sequence variation and the history of human populations". Nature Genetics 26 (3): 358–61. November 2000. doi:10.1038/81685. PMID 11062480.
- ↑ 18.0 18.1 "Structure of the Gene Pool of Bashkir Subpopulations" (in ru). 2009. http://ftp.anrb.ru/molgen/Lobov_AS.PDF.
- ↑ 19.00 19.01 19.02 19.03 19.04 19.05 19.06 19.07 19.08 19.09 19.10 19.11 "A major Y-chromosome haplogroup R1b Holocene era founder effect in Central and Western Europe". European Journal of Human Genetics 19 (1): 95–101. January 2011. doi:10.1038/ejhg.2010.146. PMID 20736979.
- ↑ 20.0 20.1 "Reconstructing the genetic history of Italians: new insights from a male (Y-chromosome) perspective". Annals of Human Biology 45 (1): 44–56. February 2018. doi:10.1080/03014460.2017.1409801. PMID 29382284.
- ↑ "Analysis of Y-chromosomal SNP haplogroups and STR haplotypes in an Algerian population sample". International Journal of Legal Medicine 122 (3): 251–255. May 2008. doi:10.1007/s00414-007-0203-5. PMID 17909833.
- ↑ "Contrasting patterns of Y chromosome and mtDNA variation in Africa: evidence for sex-biased demographic processes". European Journal of Human Genetics 13 (7): 867–876. July 2005. doi:10.1038/sj.ejhg.5201408. PMID 15856073.
- ↑ "Genetic studies on the prehispanic population buried in Punta Azul cave (El Hierro, Canary Islands)". Journal of Archaeological Science 78: 20–28. 2017. doi:10.1016/j.jas.2016.11.004. Bibcode: 2017JArSc..78...20O.
- ↑ 24.0 24.1 "Ancient migratory events in the Middle East: new clues from the Y-chromosome variation of modern Iranians". PLOS ONE 7 (7): e41252. 2012. doi:10.1371/journal.pone.0041252. PMID 22815981. Bibcode: 2012PLoSO...741252G.
- ↑ Proceedings of the Russian Academy of DNA Genealogy, 3, 1676–1695 (in Russian).
- ↑ 26.0 26.1 26.2 26.3 26.4 "Neolithic patrilineal signals indicate that the Armenian plateau was repopulated by agriculturalists". European Journal of Human Genetics 20 (3): 313–20. March 2012. doi:10.1038/ejhg.2011.192. PMID 22085901.
- ↑ Трофимова Натал'я Вадимовна (Feb. 2015), "Изменчивость Митохондриальной ДНК и Y-Хромосомы в Популяциях Волго-Уральского Региона" ("Mitochondrial DNA variation and the Y-chromosome in the population of the Volga-Ural Region"). Автореферат. диссертации на соискание ученой степени кандидата биологических наук. Уфа – 2015.
- ↑ "Massive migration from the steppe was a source for Indo-European languages in Europe". Nature 522 (7555): 207–11. June 2015. doi:10.1038/nature14317. PMID 25731166. Bibcode: 2015Natur.522..207H.
- ↑ "A comparison of Y-chromosome variation in Sardinia and Anatolia is more consistent with cultural rather than demic diffusion of agriculture". PLOS ONE 5 (4): e10419. April 2010. doi:10.1371/journal.pone.0010419. PMID 20454687. Bibcode: 2010PLoSO...510419M.
- ↑ ISOGG tree as of 2017 (isogg.org)
- ↑ "Kinship and Y-chromosome analysis of 7th century human remains: novel DNA extraction and typing procedure for ancient material". Croatian Medical Journal. 3 50 (3): 286–95. June 2009. doi:10.3325/cmj.2009.50.286. PMID 19480023.
- ↑ "A Y-chromosome signature of hegemony in Gaelic Ireland". American Journal of Human Genetics 78 (2): 334–8. February 2006. doi:10.1086/500055. PMID 16358217.
- ↑ 33.0 33.1 "Clinal patterns of human Y chromosomal diversity in continental Italy and Greece are dominated by drift and founder effects". Molecular Phylogenetics and Evolution 28 (3): 387–395. September 2003. doi:10.1016/s1055-7903(03)00016-2. PMID 12927125.
- ↑ "Colloquium paper: genome-wide patterns of population structure and admixture among Hispanic/Latino populations". Proceedings of the National Academy of Sciences of the United States of America 107 (Suppl 2): 8954–61. May 2010. doi:10.1073/pnas.0914618107. PMID 20445096. Bibcode: 2010PNAS..107.8954B.
- ↑ 35.0 35.1 "Micro-phylogeographic and demographic history of Portuguese male lineages". Annals of Human Genetics 70 (Pt 2): 181–94. March 2006. doi:10.1111/j.1529-8817.2005.00221.x. PMID 16626329. "395/657".
- ↑ "Uniparental markers in Italy reveal a sex-biased genetic structure and different historical strata". PLOS ONE 8 (5): e65441. 2013. doi:10.1371/journal.pone.0065441. PMID 23734255. Bibcode: 2013PLoSO...865441B.
- ↑ 37.0 37.1 "The Greeks in the West: genetic signatures of the Hellenic colonisation in southern Italy and Sicily". European Journal of Human Genetics 24 (3): 429–36. March 2016. doi:10.1038/ejhg.2015.124. PMID 26173964.
- ↑ 38.00 38.01 38.02 38.03 38.04 38.05 38.06 38.07 38.08 38.09 38.10 38.11 38.12 38.13 38.14 38.15 38.16 38.17 38.18 38.19 38.20 38.21 38.22 38.23 38.24 "Y-chromosome short tandem repeat DYS458.2 non-consensus alleles occur independently in both binary haplogroups J1-M267 and R1b3-M405". Croatian Medical Journal 48 (4): 450–9. August 2007. PMID 17696299. PMC 2080563. http://www.cmj.hr/2007/48/4/17696299.htm.
- ↑ 39.0 39.1 39.2 39.3 39.4 "Y-chromosomal evidence of the cultural diffusion of agriculture in Southeast Europe". European Journal of Human Genetics 17 (6): 820–30. June 2009. doi:10.1038/ejhg.2008.249. PMID 19107149.
- ↑ "High-resolution phylogenetic analysis of southeastern Europe traces major episodes of paternal gene flow among Slavic populations". Molecular Biology and Evolution 22 (10): 1964–75. October 2005. doi:10.1093/molbev/msi185. PMID 15944443.
- ↑ 41.0 41.1 41.2 "Excavating Y-chromosome haplotype strata in Anatolia". Human Genetics 114 (2): 127–48. January 2004. doi:10.1007/s00439-003-1031-4. PMID 14586639. http://hpgl.stanford.edu/publications/HG_2004_v114_p127-148.pdf.
- ↑ 42.0 42.1 "Differential Y-chromosome Anatolian influences on the Greek and Cretan Neolithic". Annals of Human Genetics 72 (Pt 2): 205–14. March 2008. doi:10.1111/j.1469-1809.2007.00414.x. PMID 18269686.
- ↑ "Y-chromosome based evidence for pre-neolithic origin of the genetically homogeneous but diverse Sardinian population: inference for association scans". PLOS ONE 3 (1): e1430. January 2008. doi:10.1371/journal.pone.0001430. PMID 18183308. Bibcode: 2008PLoSO...3.1430C. "174/930".
- ↑ 44.0 44.1 "Iran: tricontinental nexus for Y-chromosome driven migration". Human Heredity 61 (3): 132–43. 2006. doi:10.1159/000093774. PMID 16770078.
- ↑ 45.0 45.1 Varzari A (2006). Population History of the Dniester-Carpathians: Evidence from Alu Insertion and Y-Chromosome Polymorphisms (PDF) (Dissertation). Ludwig-Maximilians-Universität München.
- ↑ "Analysis of Y-chromosomal SNP haplogroups and STR haplotypes in an Algerian population sample". International Journal of Legal Medicine 122 (3): 251–5. May 2008. doi:10.1007/s00414-007-0203-5. PMID 17909833.
- ↑ 47.0 47.1 47.2 47.3 47.4 "Two sources of the Russian patrilineal heritage in their Eurasian context". American Journal of Human Genetics 82 (1): 236–50. January 2008. doi:10.1016/j.ajhg.2007.09.019. PMID 18179905.
- ↑ "Y-chromosome and mtDNA polymorphisms in Iraq, a crossroad of the early human dispersal and of post-Neolithic migrations". Molecular Phylogenetics and Evolution 28 (3): 458–72. September 2003. doi:10.1016/S1055-7903(03)00039-3. PMID 12927131. https://www.familytreedna.com/pdf/Al_Zahery.pdf. Retrieved 2017-03-17. "16/139".
- ↑ 49.0 49.1 49.2 49.3 "The Himalayas as a directional barrier to gene flow". American Journal of Human Genetics 80 (5): 884–94. May 2007. doi:10.1086/516757. PMID 17436243.
- ↑ "Y-chromosome diversity in modern Bulgarians: new clues about their ancestry". PLOS ONE 8 (3): e56779. 2013. doi:10.1371/journal.pone.0056779. PMID 23483890. Bibcode: 2013PLoSO...856779K.
- ↑ "Y-chromosomal diversity in Lebanon is structured by recent historical events". American Journal of Human Genetics 82 (4): 873–82. April 2008. doi:10.1016/j.ajhg.2008.01.020. PMID 18374297.
- ↑ 52.0 52.1 52.2 52.3 52.4 "Introducing the Algerian mitochondrial DNA and Y-chromosome profiles into the North African landscape". PLOS ONE 8 (2): e56775. 2013-02-19. doi:10.1371/journal.pone.0056775. PMID 23431392. Bibcode: 2013PLoSO...856775B.
- ↑ 53.0 53.1 53.2 53.3 "The genetic legacy of religious diversity and intolerance: paternal lineages of Christians, Jews, and Muslims in the Iberian Peninsula". American Journal of Human Genetics 83 (6): 725–36. December 2008. doi:10.1016/j.ajhg.2008.11.007. PMID 19061982.
- ↑ 54.0 54.1 54.2 "The peopling of modern Bosnia-Herzegovina: Y-chromosome haplogroups in the three main ethnic groups". Annals of Human Genetics 69 (Pt 6): 757–63. November 2005. doi:10.1111/j.1529-8817.2005.00190.x. PMID 16266413.
- ↑ 55.0 55.1 55.2 "Y-chromosome diversity characterizes the Gulf of Oman". European Journal of Human Genetics 16 (3): 374–86. March 2008. doi:10.1038/sj.ejhg.5201934. PMID 17928816.
- ↑ 56.0 56.1 56.2 56.3 "Polarity and temporality of high-resolution y-chromosome distributions in India identify both indigenous and exogenous expansions and reveal minor genetic influence of Central Asian pastoralists". American Journal of Human Genetics 78 (2): 202–21. February 2006. doi:10.1086/499411. PMID 16400607. "8/176 R-M73 and 5/176 R-M269 for a total of 13/176 R1b in Pakistan and 4/728 R-M269 in India".
- ↑ 57.0 57.1 57.2 57.3 57.4 "Sub-populations within the major European and African derived haplogroups R1b3 and E3a are differentiated by previously phylogenetically undefined Y-SNPs". Human Mutation 28 (1): 97. January 2007. doi:10.1002/humu.9469. PMID 17154278.
- ↑ 58.0 58.1 "Origins of R-M269 Diversity in Europe". https://gap.familytreedna.com/media/docs/2013/Hammer_M269_Diversity_in_Europe.pdf.
- ↑ 59.0 59.1 "In search of the pre- and post-neolithic genetic substrates in Iberia: evidence from Y-chromosome in Pyrenean populations". Annals of Human Genetics 73 (1): 42–53. January 2009. doi:10.1111/j.1469-1809.2008.00478.x. PMID 18803634.
- ↑ 60.0 60.1 60.2 "High-resolution analysis of human Y-chromosome variation shows a sharp discontinuity and limited gene flow between northwestern Africa and the Iberian Peninsula". American Journal of Human Genetics 68 (4): 1019–29. April 2001. doi:10.1086/319521. PMID 11254456.
- ↑ "Recent male-mediated gene flow over a linguistic barrier in Iberia, suggested by analysis of a Y-chromosomal DNA polymorphism". American Journal of Human Genetics 65 (5): 1437–48. November 1999. doi:10.1086/302617. PMID 10521311.
- ↑ "Y-chromosomal diversity in Europe is clinal and influenced primarily by geography, rather than by language". American Journal of Human Genetics 67 (6): 1526–43. December 2000. doi:10.1086/316890. PMID 11078479.
- ↑ The Scots: a genetic journey. Birlinn. 2011. pp. 181–182, 192. ISBN 978-0-85790-020-3. https://archive.org/details/scotsgeneticjour0000moff.
- ↑ "Strong intra- and inter-continental differentiation revealed by Y chromosome SNPs M269, U106 and U152". Forensic Science International. Genetics 5 (3): e49–52. June 2011. doi:10.1016/j.fsigen.2010.07.006. PMID 20732840.
- ↑ "Recently introduced Y-SNPs improve the resolution within Y-chromosome haplogroup R1b in a central European population sample (Tyrol, Austria)". Forensic Science International: Genetics Supplement Series 1: 226–227. August 2008. doi:10.1016/j.fsigss.2007.10.158.
- ↑ Henry Somerset was in turn descended in the patrilineal line from John of Gaunt (1340–1399), a son of King Edward III (1312–1377). In the context of the analysis of the remains of Richard III, which proved to belong to haplogroup G2, the possibility of a false-paternity event, most likely between Edward III and Henry Somerset, was discussed; possibly confirming rumors to the effect that John of Gaunt was illegitimate (Jonathan Sumption, Divided Houses: The Hundred Years War III, 2009, p. 274). "Identification of the remains of King Richard III". Nature Communications 5 (5631): 5631. December 2014. doi:10.1038/ncomms6631. PMID 25463651. Bibcode: 2014NatCo...5.5631K. "Y-chromosome haplotypes from male-line relatives and the remains do not match, which could be attributed to a false-paternity event occurring in any of the intervening generations."
- ↑ "Population genomics of Bronze Age Eurasia". Nature 522 (7555): 167–72. June 2015. doi:10.1038/nature14507. PMID 26062507. Bibcode: 2015Natur.522..167A. https://depot.ceon.pl/handle/123456789/13155.
- ↑ "137 ancient human genomes from across the Eurasian steppes". Nature 557 (7705): 369–374. May 2018. doi:10.1038/s41586-018-0094-2. PMID 29743675. Bibcode: 2018Natur.557..369D.
- ↑ "Ancient Rome: A genetic crossroads of Europe and the Mediterranean". Science 366 (6466): 708–714. November 2019. doi:10.1126/science.aay6826. PMID 31699931. Bibcode: 2019Sci...366..708A.
- ↑ "The origin and legacy of the Etruscans through a 2000-year archeogenomic time transect" (in English). Science Advances (American Association for the Advancement of Science) 7 (39): eabi7673. September 2021. doi:10.1126/sciadv.abi7673. PMID 34559560. Bibcode: 2021SciA....7.7673P.
- ↑ "R-L159 Project Goals". http://www.familytreedna.com/public/R1b-L159.2/default.aspx?section=goals.
- ↑ "A Set of Distinctive Marker Values Defines a Y-STR Signature for Gaelic Dalcassian Families". Journal of Genetic Genealogy. 2009. http://www.jogg.info/51/files/Wright.htm. Retrieved 2017-03-17.
- ↑ "R-DF21 and Subclades Project". http://www.familytreedna.com/public/R-DF21/default.aspx.
- ↑ "Dafydd Iwan's rare genetic roots unveiled in new project". walesonline. 2014-09-25. https://www.walesonline.co.uk/lifestyle/showbiz/dafydd-iwan-discovers-impressive-celtic-7829517.
- ↑ "Dafydd Iwan 'descended from Welsh kings' who ruled in England". northwales. 2014-09-26. https://www.dailypost.co.uk/news/north-wales-news/dafydd-iwan-descended-welsh-kings-7838225.
- ↑ "Family Tree DNA - My FamilyTree DNA Project Website Title". https://www.familytreedna.com/public/R-17-14-10/.
 |

