Biology:Minor capsid proteins VP2 and VP3
| Minor capsid protein VP2 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | VP2 | ||||||||
| Pfam | PF00761 | ||||||||
| InterPro | IPR001070 | ||||||||
| |||||||||
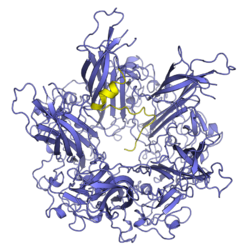
Minor capsid protein VP2 and minor capsid protein VP3 are viral proteins that are components of the polyomavirus capsid. Polyomavirus capsids are composed of three proteins; the major component is major capsid protein VP1, which self-assembles into pentamers that in turn self-assemble into enclosed icosahedral structures. The minor components are VP2 and VP3, which bind in the interior of the capsid.[1][2][3]
Gene expression

All three capsid proteins are expressed from alternative start sites on a single transcript of the "late region" of the circular viral chromosome (so named because it is transcribed late in the process of viral infection). The VP3 start site is in frame downstream from that of VP2; in consequence VP3's sequence is identical to the C-terminal portion of VP2, which has an additional N-terminal extension.[1][2] In at least some polyomaviruses, the VP2 N-terminus is myristoylated.[1][3] Some members of the polyomavirus family, such as Merkel cell polyomavirus, do not appear to encode or express VP3, though VP2 is present.[3][5]
Structure and interactions
Both VP2 and VP3 are primarily intrinsically unstructured proteins; they have DNA-binding domains[6] and a nuclear localization signal at their C-terminal ends.[7] Both VP2 and VP3 bind to the interior of VP1 pentamers in the assembled capsid.[1][2] It is generally believed that the stoichiometry of this interaction is one molecule of VP2 or VP3 to each VP1 pentamer,[2] though higher ratios have sometimes been reported, possibly indicating that pentamers can accommodate associations with two minor proteins.[3]
Function
VP2 and VP3 are thought to be involved in facilitating viral entry into the host cell, either by mediating associations with and exit from the endoplasmic reticulum or by facilitating the entry of the viral genome into the cell nucleus.[7][8][9][10][11] However, the precise mechanism of their involvement is unclear, and may vary among polyomaviruses. In most studies, viral propagation is either reduced or abrogated in the absence of one or both proteins, but the apparent mechanisms vary; for example, in JC virus both VP2 and VP3 seem to be essential for packaging the viral chromosome into the capsid,[12] while absence of these proteins in SV40 prevents successful entry into new host cells,[8][9] with variable effects on packaging reported.[8][13] In Merkel cell polyomavirus, the effect of VP2 appears to vary depending on the cell type of the infected cell.[3] In murine polyomavirus the minor proteins have been reported to induce apoptosis in the infected cell,[14] and in SV40 they have been identified as viroporins.[15]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "Interaction of polyomavirus internal protein VP2 with the major capsid protein VP1 and implications for participation of VP2 in viral entry". The EMBO Journal 17 (12): 3233–40. June 1998. doi:10.1093/emboj/17.12.3233. PMID 9628860.
- ↑ 2.0 2.1 2.2 2.3 "A cornucopia of human polyomaviruses". Nature Reviews. Microbiology 11 (4): 264–76. April 2013. doi:10.1038/nrmicro2992. PMID 23474680.
- ↑ 3.0 3.1 3.2 3.3 3.4 "The Merkel cell polyomavirus minor capsid protein". PLoS Pathogens 9 (8): e1003558. 2013-08-22. doi:10.1371/journal.ppat.1003558. PMID 23990782.
- ↑ "Identification of a novel polyomavirus from patients with acute respiratory tract infections". PLoS Pathogens 3 (5): e64. May 2007. doi:10.1371/journal.ppat.0030064. PMID 17480120.
- ↑ "The Ancient Evolutionary History of Polyomaviruses". PLoS Pathogens 12 (4): e1005574. April 2016. doi:10.1371/journal.ppat.1005574. PMID 27093155.
- ↑ "Identification of a DNA binding domain in simian virus 40 capsid proteins Vp2 and Vp3". The Journal of Biological Chemistry 268 (28): 20877–83. October 1993. PMID 8407920. http://www.jbc.org/content/268/28/20877.
- ↑ 7.0 7.1 "Role of a nuclear localization signal on the minor capsid proteins VP2 and VP3 in BKPyV nuclear entry". Virology 474: 110–6. January 2015. doi:10.1016/j.virol.2014.10.013. PMID 25463609.
- ↑ 8.0 8.1 8.2 "SV40 VP2 and VP3 insertion into ER membranes is controlled by the capsid protein VP1: implications for DNA translocation out of the ER" (in English). Molecular Cell 24 (6): 955–66. December 2006. doi:10.1016/j.molcel.2006.11.001. PMID 17189196.
- ↑ 9.0 9.1 "Minor capsid proteins of simian virus 40 are dispensable for nucleocapsid assembly and cell entry but are required for nuclear entry of the viral genome". Journal of Virology 81 (8): 3778–85. April 2007. doi:10.1128/jvi.02664-06. PMID 17267496.
- ↑ "A large and intact viral particle penetrates the endoplasmic reticulum membrane to reach the cytosol". PLoS Pathogens 7 (5): e1002037. May 2011. doi:10.1371/journal.ppat.1002037. PMID 21589906.
- ↑ "Hydrophobic domains of mouse polyomavirus minor capsid proteins promote membrane association and virus exit from the ER". The FEBS Journal 284 (6): 883–902. March 2017. doi:10.1111/febs.14033. PMID 28164464.
- ↑ "JC virus minor capsid proteins Vp2 and Vp3 are essential for virus propagation". Journal of Virology 80 (21): 10858–61. November 2006. doi:10.1128/jvi.01298-06. PMID 17041227.
- ↑ "The VP2/VP3 minor capsid protein of simian virus 40 promotes the in vitro assembly of the major capsid protein VP1 into particles". The Journal of Biological Chemistry 281 (15): 10164–73. April 2006. doi:10.1074/jbc.M511261200. PMID 16478732.
- ↑ "Minor capsid proteins of mouse polyomavirus are inducers of apoptosis when produced individually but are only moderate contributors to cell death during the late phase of viral infection". The FEBS Journal 277 (5): 1270–83. March 2010. doi:10.1111/j.1742-4658.2010.07558.x. PMID 20121946.
- ↑ "The viroporin activity of the minor structural proteins VP2 and VP3 is required for SV40 propagation". The Journal of Biological Chemistry 288 (4): 2510–20. January 2013. doi:10.1074/jbc.M112.428425. PMID 23223228.