Biology:Myosin binding protein C, cardiac
 Generic protein structure example |
The myosin-binding protein C, cardiac-type is a protein that in humans is encoded by the MYBPC3 gene.[1] This isoform is expressed exclusively in heart muscle during human and mouse development,[2] and is distinct from those expressed in slow skeletal muscle (MYBPC1) and fast skeletal muscle (MYBPC2).
Structure
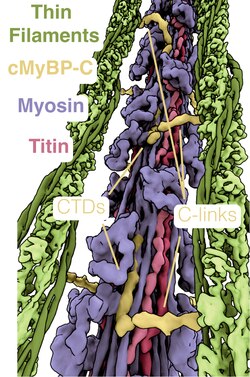
cMyBP-C is a 140.5 kDa protein composed of 1273 amino acids.[3][4][5] cMyBP-C is a myosin-associated protein that binds at 43 nm intervals along the myosin thick filament backbone, stretching for 200 nm on either side of the M-line within the crossbridge-bearing zone (C-region) of the A band in striated muscle.[6] The approximate stoichiometry of cMyBP-C along the thick filament is 1 per 9-10 myosin molecules, or 37 cMyBP-C molecules per thick filament.[7] In addition to myosin, cMyBP-C also binds titin and actin.[8][9] The cMyBP-C isoform expressed in cardiac muscle differs from those expressed in slow and fast skeletal muscle (MYBPC1 and MYBPC2, respectively) by three features: (1) an additional immunoglobulin (Ig)-like domain on the N-terminus, (2) a linker region between the second and third Ig domains, and (3) an additional loop in the sixth Ig domain.[10] cMyBP-C appears necessary for normal order, filament length and lattice spacing within the structure of the sarcomere.[11][12]
Function
cMyBP-C is not essential for sarcomere formation during embryogenesis, but is crucial for sarcomere organization and maintenance of normal cardiac function. Absence of cMyBP-C (Mybpc3-targeted knock-out mice) results in severe cardiac hypertrophy, increased heart-weight-to-body-weight-ratios, enlargement of ventricles, increased myofilament Ca2+ sensitivity and depressed diastolic and systolic function.[13][14][15] Histologically, Mybpc3-targeted knock-out hearts display structural rearrangements with cardiac myocyte disarray and increased interstitial fibrosis similar to patients with hypertrophic cardiomyopathy, without obvious alterations in shape or size of single cardiac myocytes. Ultrastructural examination revealed a loss of lateral alignment of adjacent myofibrils with their Z-lines misaligned.[13][14][16][17]
cMyBP-C appears to act as a brake on cardiac contraction, as loaded shortening, power and cycling kinetics all increase in cMyBP-C knockout mice.[18] Consistent with this notion, cMyBP-C knockout mice exhibit an abnormal systolic timecourse, with a shortened elastance timecourse and lower peak elastance in vivo,[19] and an accelerated force development in isolated, skinned cardiac fibers[20] suggesting that cMyBP-C is required to constrain the crossbridges in order to sustain a normal ejection.
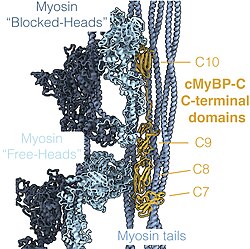
cMyBP-C regulates the positioning of myosin and actin for interaction and acts as a tether to the myosin S1 heads, limiting their mobility. This results in a decreased number of crossbridges formed, which hinders force generation, due to its N-terminal C1-M-C2 region interacting with the myosin-S2 domain.[21][22][23][24] Furthermore, cMyBP-C contributes to the regulation of cardiac contraction at short sarcomere length and is required for complete relaxation in diastole.[15][25]
Interactions of cMyBP-C with its binding partners vary with its posttranslational modification status. At least three extensively characterized phosphorylation sites (Ser273, 282 and 302; numbering refers to the mouse sequence) are localized in the M motif of cMyBP-C and are targeted by protein kinases in a hierarchical order of events. In its dephosphorylated state, cMyBP-C binds predominantly to myosin S2 and brakes crossbridge formation, however, when phosphorylated in response to β-adrenergic stimulation through activating cAMP-dependent protein kinase (PKA), it favours binding to actin, then accelerating crossbridge formation, enhancing force development and promoting relaxation.[26] Protein kinases identified thus far to phosphorylate cMyBP-C in the M motif are PKA,[27][28][29][30][31] Ca2+/calmodulin-dependent kinase II (CaMKII),[32] ribosomal s6 kinase (RSK),[33]protein kinase D (PKD),[34][35] and protein kinase C (PKC).[30] Furthermore, GSK3β was described as another protein kinase to phosphorylate cMyBP-C outside the M-domain in the proline-alanine-rich actin-binding site at Ser133 in human myocardium (mouse Ser131).[36] Phosphorylation is required for normal cardiac function and cMyBP-C stability,[37][38] and overall phosphorylation levels of cMyBP-C are reduced in human and experimental heart failure.[39][40] Other posttranslational modifications of cMyBP-C exist, which occur throughout the protein and are not thoroughly characterised yet, such as acetylation,[41] citrullination,[42] S-glutathiolation,[43][44][45][46] S-nitrosylation[47] and carbonylation.[48]
Genetics
The cloning of the human MYBPC3 cDNA and localization of the gene on human chromosome 11p11.2 has assisted the structure and function of cMyBP-C.[49] MYBPC3 became therefore the “best” candidate gene for the CMH4 locus for hypertrophic cardiomyopathy that was initially mapped by the group of Schwartz.[50] MYBPC3 mutations segregating in families with hypertrophic cardiomyopathy have been identified.[51][52] MYBPC3 was thus the fourth gene for hypertrophic cardiomyopathy, following MYH7, encoding β-myosin heavy chain, TNNT2 and TPM1, encoding cardiac troponin T and α-tropomyosin, respectively, earmarking hypertrophic cardiomyopathy (HCM) as a disease of the sarcomere. Truncation mutations in MYBPC3 stand as the primary cause of HCM.[53]
To date, roughly 350 mutations in MYBPC3 have been identified, and in large part, the mutations result in protein truncation, shifts in reading frames, and premature termination codons.[54][55] Genetic studies have revealed significant overlap between genotypes and phenotypes as MYBPC3 mutations can lead to various forms of cardiomyopathies, such as dilated cardiomyopathy[56] and left ventricular noncompaction cardiomyopathy.[57] In patients with isolated or familial cases of dilated cardiomyoathy, MYBPC3 mutations represented the second highest number of known mutations.[56] Furthermore, a 25-bp intronic MYBPC3 deletion leading to protein truncation is present in 4% of the population in South India and is associated with a higher risk to develop heart failure.[58] Founder MYBPC3 mutations have been reported in Iceland, Italy, The Netherlands, Japan, France and Finland, where they represent a large percentage of cases with hypertrophic cardiomyopathy. All of them are truncating mutations, resulting in a shorter protein, lacking the regulatory phosphorylatable M motif and/or major binding domains to other sarcomeric proteins.[59][60][61][62][63][64][65] A body of evidence indicates that patients with more than 1 mutation often develop a more severe phenotype,[66] and a significant fraction of childhood-onset hypertrophic cardiomyopathy (14%) is caused by compound genetic variants.[67] This suggests that a gene-dosage effect might be responsible for manifestations at a younger age. A total of 51 cases of homozygotes or compound heterozygotes have been reported, most of them with double truncating MYBPC3 mutations and associated with severe cardiomyopathy, leading to heart failure and death within the first year of life.[68]
Pathomechanisms
A great understanding of how MYBPC3 mutations lead to the development of inherited cardiomyopathy came from the analyses of human myocardial samples, gene transfer in different cell lines, naturally-occurring or transgenic animal models and more recently disease modeling using induced pluripotent stem cells (iPSC)-derived cardiac myocytes.[69][70] Although access to human myocardial samples is difficult, at least some studies provided evidence that truncated cMyBP-Cs, resulting from truncating MYBPC3 mutations are not detectable in human patient samples by Western-immunoblot analysis.[71][72][73][74] This was supported in heterozygous Mybpc3-targeted knock-in mice,[75] carrying the human c.772G>A transition (i.e. founder mutation in Tuscany[63] These data suggest haploinsufficiency as the main disease mechanism for heterozygous truncating mutations.[76][77] A body of evidence exists that the mechanisms regulating the expression of mutant allele involve the nonsense-mediated mRNA decay, the ubiquitin-proteasome system (UPS) and the autophagy-lysosomal pathway after gene transfer of mutant MYBPC3 in cardiac myocytes or in mice in vivo.[78][79][75][80][81][82] In contrast to truncating mutations, missense mutations lead, in most of the cases (although difficult to specifically detect), to stable mutant cMyBP-Cs that are, at least in part, incorporated into the sarcomere and could act as poison polypeptides on the structure and/or function of the sarcomere. Homozygous or compound heterozygous mutations are therefore likely subject to differential regulation depending on whether they are double missense, double truncating or mixed missense/truncating mutations. The homozygous Mybpc3-targeted knock-in mice, which genetically mimic the situation of severe neonatal cardiomyopathy are born without phenotype and soon after birth develop systolic dysfunction followed by (compensatory) cardiac hypertrophy.[83][84] The human c.772G>A transition results in low levels of three different mutant Mybpc3 mRNAs and cMyBP-Cs in homozygous mice, suggesting a combination of haploinsufficiency and polypeptide poisoning as disease mechanism in the homozygous state.[75] In addition, the combination of external stress (such as neurohumoral stress or aging) and Mybpc3 mutations have been shown to impair the UPS in mice,[85][86] and proteasomal activities were also depressed in patients with hypertrophic cardiomyopathy or dilated cardiomyopathy.[87]
Skinned trabeculae or cardiac myocytes obtained from human patients carrying a MYBPC3 mutation or from heterozygous and homozygous Mybpc3-targeted knock-in mice exhibited higher myofilament Ca2+ sensitivity than controls.[88][74][89][90][91] Disease-modeling by engineered heart tissue (EHT) technology with cardiac cells from heterozygous or homozygous Mybpc3-targeted knock-in mice reproduced observations made in human and mouse studies displaying abbreviated contractions, greater sensitivity to external Ca2+ and smaller inotropic responses to various drugs (isoprenaline, EMD 57033 and verapamil) compared to wild-type control EHTs.[92] Therefore, EHTs are suitable to model the disease phenotype and recapitulate functional alterations found in mice with hypertrophic cardiomyopathy. Another good system for modeling cardiomyopathies in the cell culture dish is the derivation of cardiac myocytes from iPSC. Reports of human iPSC models of sarcomeric cardiomyopathies showed cellular hypertrophy in most of the cases,[93][94][95][96] including one with the c.2995_3010del MYBPC3 mutation that exhibited in addition to hypertrophy contractile variability in the presence of endothelin-1.[96]
Therapy
Because of their tissue selectivity and persistent expression recombinant adeno-associated viruses (AAV) have therapeutic potential in the treatment of inherited cardiomyopathy resulting from MYBPC3 mutations-[97] Several targeting approaches have been developed.[98][99] The most recent is genome editing to correct a mutation by CRISPR/Cas9 technology.[100] Naturally existing as part of the prokaryotic immune system, the CRISPR/Cas9 system has been used for correction of mutations in the mammalian genome.[101] By inducing nicks in the double-stranded DNA and providing a template DNA sequence, it is possible to repair mutations by homologous recombination. This approach has not yet been evaluated for MYBPC3 mutations, but it could be used for each single or clustered mutation, and therefore applied preferentially for frequent founder MYBPC3 mutations.
Other strategies targeting the mutant pre-mRNA by exon skipping and/or spliceosome-mediated RNA trans-splicing (SMaRT) have been evaluated for MYBPC3. Exon skipping can be achieved using antisense oligonucleotide (AON) masking exonic splicing enhancer sequences and therefore preventing binding of the splicing machinery and therefore resulting in exclusion of the exon from the mRNA.[102][103] This approach can be applied when the resulting shorter, but in-frame translated protein maintains its function. Proof-of-concept of exon skipping was recently shown in homozygous Mybpc3-targeted knock-in mice.[83] Systemic administration of AAV-based AONs to Mybpc3-targeted knock-in newborn mice prevented both systolic dysfunction and left ventricular hypertrophy, at least for the duration of the investigated period.[83] For the human MYBPC3 gene, skipping of 6 single exons or 5 double exons with specific AONs would result in shortened in-frame cMyBP-Cs, allowing the preservation of the functionally important phosphorylation and protein interaction sites. With this approach, about half of missense or exonic/intronic truncating mutations could be removed, including 35 mutations in exon 25. The other strategy targeting the mutant pre-mRNA is SMaRT. Hereby, two independently transcribed molecules, the mutant pre-mRNA and the therapeutic pre-trans-splicing molecule carrying the wild-type sequence are spliced together to give rise to a repaired full-length mRNA.[104] Recently, the feasibility of this method was shown both in isolated cardiac myocytes and in vivo in the heart of homozygous Mybpc3-targeted knock-in mice, although the efficiency of the process was low and the amount of repaired protein was not sufficient to prevent the development of the cardiac disease phenotype.[84] In principle, however, this SmART strategy is superior to exon skipping or CRISPR/Cas9 genome editing and still attractive, because only two pre-trans-splicing molecules, targeting the 5’ and the 3’ of MYBPC3 pre-mRNA would be sufficient to bypass all MYBPC3 mutations associated with cardiomyopathies and therefore repair the mRNA.
AAV-mediated gene transfer of the full-length Mybpc3 (defined as “gene replacement”) dose-dependently prevents the development of cardiac hypertrophy and dysfunction in homozygous Mybpc3-targeted knock-in mice.[105] The dose-dependent expression of exogenous Mybpc3 was associated with the down-regulation of endogenous mutant Mybpc3. Additional expression of a sarcomeric protein is expected to replace partially or completely the endogenous protein level in the sarcomere, as it has been shown in transgenic mice expressing sarcomeric proteins.[69]
Notes
References
- ↑ "Phosphorylation switches specific for the cardiac isoform of myosin binding protein-C: a modulator of cardiac contraction?". The EMBO Journal 14 (9): 1952–60. May 1995. doi:10.1002/j.1460-2075.1995.tb07187.x. PMID 7744002.
- ↑ "Cardiac myosin binding protein C gene is specifically expressed in heart during murine and human development". Circulation Research 82 (1): 130–3. 1998. doi:10.1161/01.res.82.1.130. PMID 9440712.
- ↑ "Organization and sequence of human cardiac myosin binding protein C gene (MYBPC3) and identification of mutations predicted to produce truncated proteins in familial hypertrophic cardiomyopathy". Circulation Research 80 (3): 427–34. Mar 1997. doi:10.1161/01.res.0000435859.24609.b3. PMID 9048664.
- ↑ "Protein Information - Myosin-binding protein C, cardiac-type". Cardiac Organellar Protein Atlas Knowledgebase (COPaKB). NHLBI Proteomics Center at UCLA. http://www.heartproteome.org/copa/ProteinInfo.aspx?QType=Protein%20ID&QValue=Q14896.
- ↑ "Integration of cardiac proteome biology and medicine by a specialized knowledgebase". Circulation Research 113 (9): 1043–53. Oct 2013. doi:10.1161/CIRCRESAHA.113.301151. PMID 23965338.
- ↑ "The ultrastructural location of C-protein, X-protein and H-protein in rabbit muscle". Journal of Muscle Research and Cell Motility 7 (6): 550–67. Dec 1986. doi:10.1007/bf01753571. PMID 3543050.
- ↑ "A new protein of the thick filaments of vertebrate skeletal myofibrils. Extractions, purification and characterization". Journal of Molecular Biology 74 (4): 653–76. Mar 1973. doi:10.1016/0022-2836(73)90055-7. PMID 4269687.
- ↑ "A molecular map of the interactions between titin and myosin-binding protein C. Implications for sarcomeric assembly in familial hypertrophic cardiomyopathy". European Journal of Biochemistry 235 (1–2): 317–23. Jan 1996. doi:10.1111/j.1432-1033.1996.00317.x. PMID 8631348.
- ↑ "The myosin-binding protein C motif binds to F-actin in a phosphorylation-sensitive manner". The Journal of Biological Chemistry 284 (18): 12318–27. May 2009. doi:10.1074/jbc.M808850200. PMID 19269976.
- ↑ "Cardiac myosin binding protein C". Circulation Research 84 (10): 1117–26. May 1999. doi:10.1161/01.res.84.10.1117. PMID 10347086.
- ↑ "Effects of C-protein on synthetic myosin filament structure". Biophysical Journal 27 (3): 433–46. Sep 1979. doi:10.1016/S0006-3495(79)85227-3. PMID 263692. Bibcode: 1979BpJ....27..433K.
- ↑ "Radial displacement of myosin cross-bridges in mouse myocardium due to ablation of myosin binding protein-C". Journal of Molecular Biology 367 (1): 36–41. Mar 2007. doi:10.1016/j.jmb.2006.12.063. PMID 17254601.
- ↑ 13.0 13.1 "Hypertrophic cardiomyopathy in cardiac myosin binding protein-C knockout mice". Circulation Research 90 (5): 594–601. Mar 2002. doi:10.1161/01.res.0000012222.70819.64. PMID 11909824.
- ↑ 14.0 14.1 "Asymmetric septal hypertrophy in heterozygous cMyBP-C null mice". Cardiovascular Research 63 (2): 293–304. Aug 2004. doi:10.1016/j.cardiores.2004.04.009. PMID 15249187.
- ↑ 15.0 15.1 "Length and protein kinase A modulations of myocytes in cardiac myosin binding protein C-deficient mice". Cardiovascular Research 69 (2): 370–80. Feb 2006. doi:10.1016/j.cardiores.2005.11.009. PMID 16380103.
- ↑ "In vivo left ventricular functional capacity is compromised in cMyBP-C null mice". American Journal of Physiology. Heart and Circulatory Physiology 292 (4): H1747–54. Apr 2007. doi:10.1152/ajpheart.01037.2006. PMID 17122190.
- ↑ "Understanding the organisation and role of myosin binding protein C in normal striated muscle by comparison with MyBP-C knockout cardiac muscle". Journal of Molecular Biology 384 (1): 60–72. Dec 2008. doi:10.1016/j.jmb.2008.09.013. PMID 18817784.
- ↑ "Loaded shortening, power output, and rate of force redevelopment are increased with knockout of cardiac myosin binding protein-C". Circulation Research 93 (8): 752–8. Oct 2003. doi:10.1161/01.RES.0000096363.85588.9A. PMID 14500336.
- ↑ "Role of cardiac myosin binding protein C in sustaining left ventricular systolic stiffening". Circulation Research 94 (9): 1249–55. May 2004. doi:10.1161/01.RES.0000126898.95550.31. PMID 15059932.
- ↑ "Ablation of myosin-binding protein-C accelerates force development in mouse myocardium". Biophysical Journal 90 (11): 4119–27. Jun 2006. doi:10.1529/biophysj.105.078147. PMID 16513777. Bibcode: 2006BpJ....90.4119S.
- ↑ "Mutations in beta-myosin S2 that cause familial hypertrophic cardiomyopathy (FHC) abolish the interaction with the regulatory domain of myosin-binding protein-C". Journal of Molecular Biology 286 (3): 933–49. Feb 1999. doi:10.1006/jmbi.1998.2522. PMID 10024460.
- ↑ "Myosin binding protein C, a phosphorylation-dependent force regulator in muscle that controls the attachment of myosin heads by its interaction with myosin S2". Circulation Research 86 (1): 51–8. 2000. doi:10.1161/01.res.86.1.51. PMID 10625305.
- ↑ "Binding of myosin binding protein-C to myosin subfragment S2 affects contractility independent of a tether mechanism". Circulation Research 95 (9): 930–6. Oct 2004. doi:10.1161/01.RES.0000147312.02673.56. PMID 15472117.
- ↑ "Dissecting the N-terminal myosin binding site of human cardiac myosin-binding protein C. Structure and myosin binding of domain C2". The Journal of Biological Chemistry 282 (12): 9204–15. Mar 2007. doi:10.1074/jbc.M610899200. PMID 17192269.
- ↑ "Cardiac myosin-binding protein C is required for complete relaxation in intact myocytes". Circulation Research 101 (9): 928–38. Oct 2007. doi:10.1161/CIRCRESAHA.107.158774. PMID 17823372.
- ↑ "Cardiac MyBP-C regulates the rate and force of contraction in mammalian myocardium". Circulation Research 116 (1): 183–92. Jan 2015. doi:10.1161/CIRCRESAHA.116.300561. PMID 25552695.
- ↑ "Effects of cholinergic and adrenergic agonists on phosphorylation of a 165,000-dalton myofibrillar protein in intact cardiac muscle". The Journal of Biological Chemistry 257 (4): 2111–20. Feb 1982. doi:10.1016/S0021-9258(19)68153-6. PMID 6276407.
- ↑ "Phosphorylation of purified cardiac muscle C-protein by purified cAMP-dependent and endogenous Ca2+-calmodulin-dependent protein kinases". The Journal of Biological Chemistry 259 (24): 15587–96. Dec 1984. doi:10.1016/S0021-9258(17)42588-9. PMID 6549009.
- ↑ "Phosphorylation switches specific for the cardiac isoform of myosin binding protein-C: a modulator of cardiac contraction?". The EMBO Journal 14 (9): 1952–60. May 1995. doi:10.1002/j.1460-2075.1995.tb07187.x. PMID 7744002.
- ↑ 30.0 30.1 "Cardiac myosin-binding protein C (MyBP-C): identification of protein kinase A and protein kinase C phosphorylation sites". Archives of Biochemistry and Biophysics 358 (2): 313–9. Oct 1998. doi:10.1006/abbi.1998.0857. PMID 9784245.
- ↑ "Changes in cardiac contractility related to calcium-mediated changes in phosphorylation of myosin-binding protein C". Biophysical Journal 81 (2): 1083–92. Aug 2001. doi:10.1016/S0006-3495(01)75765-7. PMID 11463649. Bibcode: 2001BpJ....81.1083M.
- ↑ "A critical function for Ser-282 in cardiac Myosin binding protein-C phosphorylation and cardiac function". Circulation Research 109 (2): 141–50. Jul 2011. doi:10.1161/CIRCRESAHA.111.242560. PMID 21597010.
- ↑ "Novel role for p90 ribosomal S6 kinase in the regulation of cardiac myofilament phosphorylation". The Journal of Biological Chemistry 286 (7): 5300–10. Feb 2011. doi:10.1074/jbc.M110.202713. PMID 21148481.
- ↑ "Distinct sarcomeric substrates are responsible for protein kinase D-mediated regulation of cardiac myofilament Ca2+ sensitivity and cross-bridge cycling". The Journal of Biological Chemistry 285 (8): 5674–82. Feb 2010. doi:10.1074/jbc.M109.066456. PMID 20018870.
- ↑ "Protein kinase D increases maximal Ca2+-activated tension of cardiomyocyte contraction by phosphorylation of cMyBP-C-Ser315". American Journal of Physiology. Heart and Circulatory Physiology 303 (3): H323–31. Aug 2012. doi:10.1152/ajpheart.00749.2011. PMID 22636676.
- ↑ "GSK3β phosphorylates newly identified site in the proline-alanine-rich region of cardiac myosin-binding protein C and alters cross-bridge cycling kinetics in human: short communication". Circulation Research 112 (4): 633–9. Feb 2013. doi:10.1161/CIRCRESAHA.112.275602. PMID 23277198.
- ↑ "Pathogenic properties of the N-terminal region of cardiac myosin binding protein-C in vitro". Journal of Muscle Research and Cell Motility 33 (1): 17–30. May 2012. doi:10.1007/s10974-012-9292-y. PMID 22527638.
- ↑ "Myocardial infarction-induced N-terminal fragment of cardiac myosin-binding protein C (cMyBP-C) impairs myofilament function in human myocardium". The Journal of Biological Chemistry 289 (13): 8818–27. Mar 2014. doi:10.1074/jbc.M113.541128. PMID 24509847.
- ↑ "Decreased phosphorylation levels of cardiac myosin-binding protein-C in human and experimental heart failure". Journal of Molecular and Cellular Cardiology 43 (2): 223–9. Aug 2007. doi:10.1016/j.yjmcc.2007.05.003. PMID 17560599.
- ↑ "Analysis of cardiac myosin binding protein-C phosphorylation in human heart muscle". Journal of Molecular and Cellular Cardiology 49 (6): 1003–11. Dec 2010. doi:10.1016/j.yjmcc.2010.09.007. PMID 20850451. https://zenodo.org/record/3423484.
- ↑ "Top-down high-resolution mass spectrometry of cardiac myosin binding protein C revealed that truncation alters protein phosphorylation state". Proceedings of the National Academy of Sciences of the United States of America 106 (31): 12658–63. Aug 2009. doi:10.1073/pnas.0813369106. PMID 19541641. Bibcode: 2009PNAS..10612658G.
- ↑ "Proteomics of citrullination in cardiovascular disease". Proteomics: Clinical Applications 8 (7–8): 522–33. Aug 2014. doi:10.1002/prca.201400013. PMID 24946285.
- ↑ "The utility of N,N-biotinyl glutathione disulfide in the study of protein S-glutathiolation". Molecular & Cellular Proteomics 5 (2): 215–25. Feb 2006. doi:10.1074/mcp.M500212-MCP200. PMID 16223748.
- ↑ "Ranolazine improves cardiac diastolic dysfunction through modulation of myofilament calcium sensitivity". Circulation Research 110 (6): 841–50. Mar 2012. doi:10.1161/CIRCRESAHA.111.258251. PMID 22343711.
- ↑ "Tetrahydrobiopterin improves diastolic dysfunction by reversing changes in myofilament properties". Journal of Molecular and Cellular Cardiology 56: 44–54. Mar 2013. doi:10.1016/j.yjmcc.2012.12.003. PMID 23247392.
- ↑ "Novel control of cardiac myofilament response to calcium by S-glutathionylation at specific sites of myosin binding protein C". Frontiers in Physiology 4: 336. 2013. doi:10.3389/fphys.2013.00336. PMID 24312057.
- ↑ "Characterization of potential S-nitrosylation sites in the myocardium". American Journal of Physiology. Heart and Circulatory Physiology 300 (4): H1327–35. Apr 2011. doi:10.1152/ajpheart.00997.2010. PMID 21278135.
- ↑ "Doxorubicin-induced carbonylation and degradation of cardiac myosin binding protein C promote cardiotoxicity". Proceedings of the National Academy of Sciences of the United States of America 111 (5): 2011–6. Feb 2014. doi:10.1073/pnas.1321783111. PMID 24449919. Bibcode: 2014PNAS..111.2011A.
- ↑ "Phosphorylation switches specific for the cardiac isoform of myosin binding protein-C: a modulator of cardiac contraction?". The EMBO Journal 14 (9): 1952–60. May 1995. doi:10.1002/j.1460-2075.1995.tb07187.x. PMID 7744002.
- ↑ "Mapping of a novel gene for familial hypertrophic cardiomyopathy to chromosome 11". Nature Genetics 4 (3): 311–3. Jul 1993. doi:10.1038/ng0793-311. PMID 8358441.
- ↑ "Cardiac myosin binding protein-C gene splice acceptor site mutation is associated with familial hypertrophic cardiomyopathy". Nature Genetics 11 (4): 438–40. Dec 1995. doi:10.1038/ng1295-438. PMID 7493026.
- ↑ "Mutations in the cardiac myosin binding protein-C gene on chromosome 11 cause familial hypertrophic cardiomyopathy". Nature Genetics 11 (4): 434–7. Dec 1995. doi:10.1038/ng1295-434. PMID 7493025.
- ↑ O'Leary, Thomas S.; Snyder, Julia; Sadayappan, Sakthivel; Day, Sharlene M.; Previs, Michael J. (2019). "MYBPC3 truncation mutations enhance actomyosin contractile mechanics in human hypertrophic cardiomyopathy". Journal of Molecular and Cellular Cardiology 127: 165–173. doi:10.1016/j.yjmcc.2018.12.003. ISSN 0022-2828. PMID 30550750. PMC 6592272. http://dx.doi.org/10.1016/j.yjmcc.2018.12.003.
- ↑ "In the thick of it: HCM-causing mutations in myosin binding proteins of the thick filament". Circulation Research 108 (6): 751–64. Mar 2011. doi:10.1161/CIRCRESAHA.110.231670. PMID 21415409.
- ↑ "MYBPC3 in hypertrophic cardiomyopathy: from mutation identification to RNA-based correction". Pflügers Archiv 466 (2): 215–23. Feb 2014. doi:10.1007/s00424-013-1409-7. PMID 24337823.
- ↑ 56.0 56.1 "Atlas of the clinical genetics of human dilated cardiomyopathy". European Heart Journal 36 (18): 1123–35. May 2015. doi:10.1093/eurheartj/ehu301. PMID 25163546.
- ↑ "Sarcomere gene mutations in isolated left ventricular noncompaction cardiomyopathy do not predict clinical phenotype". Circulation: Cardiovascular Genetics 4 (4): 367–74. Aug 2011. doi:10.1161/CIRCGENETICS.110.959270. PMID 21551322.
- ↑ "A common MYBPC3 (cardiac myosin binding protein C) variant associated with cardiomyopathies in South Asia". Nature Genetics 41 (2): 187–91. Feb 2009. doi:10.1038/ng.309. PMID 19151713.
- ↑ "Nationwide study on hypertrophic cardiomyopathy in Iceland: evidence of a MYBPC3 founder mutation". Circulation 130 (14): 1158–67. Sep 2014. doi:10.1161/CIRCULATIONAHA.114.011207. PMID 25078086.
- ↑ "A founder MYBPC3 mutation results in HCM with a high risk of sudden death after the fourth decade of life". Journal of Medical Genetics 52 (5): 338–47. May 2015. doi:10.1136/jmedgenet-2014-102923. PMID 25740977.
- ↑ "Founder mutations in hypertrophic cardiomyopathy patients in the Netherlands". Netherlands Heart Journal 18 (5): 248–54. May 2010. doi:10.1007/bf03091771. PMID 20505798.
- ↑ "Lifelong left ventricular remodeling of hypertrophic cardiomyopathy caused by a founder frameshift deletion mutation in the cardiac Myosin-binding protein C gene among Japanese". Journal of the American College of Cardiology 46 (9): 1737–43. Nov 2005. doi:10.1016/j.jacc.2005.05.087. PMID 16256878.
- ↑ 63.0 63.1 "A molecular screening strategy based on beta-myosin heavy chain, cardiac myosin binding protein C and troponin T genes in Italian patients with hypertrophic cardiomyopathy". Journal of Cardiovascular Medicine 7 (8): 601–7. Aug 2006. doi:10.2459/01.JCM.0000237908.26377.d6. PMID 16858239.
- ↑ "A human MYBPC3 mutation appearing about 10 centuries ago results in a hypertrophic cardiomyopathy with delayed onset, moderate evolution but with a risk of sudden death". BMC Medical Genetics 13: 105. 2012. doi:10.1186/1471-2350-13-105. PMID 23140321.
- ↑ "Genetics of hypertrophic cardiomyopathy in eastern Finland: few founder mutations with benign or intermediary phenotypes". Annals of Medicine 36 (1): 23–32. 2004. doi:10.1080/07853890310017161. PMID 15000344.
- ↑ "Hypertrophic cardiomyopathy: distribution of disease genes, spectrum of mutations, and implications for a molecular diagnosis strategy". Circulation 107 (17): 2227–32. May 2003. doi:10.1161/01.CIR.0000066323.15244.54. PMID 12707239.
- ↑ "Shared genetic causes of cardiac hypertrophy in children and adults". The New England Journal of Medicine 358 (18): 1899–908. May 2008. doi:10.1056/NEJMoa075463. PMID 18403758.
- ↑ "Compound heterozygous or homozygous truncating MYBPC3 mutations cause lethal cardiomyopathy with features of noncompaction and septal defects". European Journal of Human Genetics 23 (7): 922–8. Oct 2014. doi:10.1038/ejhg.2014.211. PMID 25335496.
- ↑ 69.0 69.1 "Animal and in silico models for the study of sarcomeric cardiomyopathies". Cardiovascular Research 105 (4): 439–48. Apr 2015. doi:10.1093/cvr/cvv006. PMID 25600962.
- ↑ "Modelling sarcomeric cardiomyopathies in the dish: from human heart samples to iPSC cardiomyocytes". Cardiovascular Research 105 (4): 424–38. Apr 2015. doi:10.1093/cvr/cvv017. PMID 25618410.
- ↑ "Novel splice donor site mutation in the cardiac myosin-binding protein-C gene in familial hypertrophic cardiomyopathy. Characterization Of cardiac transcript and protein". The Journal of Clinical Investigation 100 (2): 475–82. Jul 1997. doi:10.1172/JCI119555. PMID 9218526.
- ↑ "A newly created splice donor site in exon 25 of the MyBP-C gene is responsible for inherited hypertrophic cardiomyopathy with incomplete disease penetrance". Circulation 101 (12): 1396–402. Mar 2000. doi:10.1161/01.cir.101.12.1396. PMID 10736283.
- ↑ "Evidence from human myectomy samples that MYBPC3 mutations cause hypertrophic cardiomyopathy through haploinsufficiency". Circulation Research 105 (3): 219–22. Jul 2009. doi:10.1161/CIRCRESAHA.109.202440. PMID 19574547.
- ↑ 74.0 74.1 "Cardiac myosin-binding protein C mutations and hypertrophic cardiomyopathy: haploinsufficiency, deranged phosphorylation, and cardiomyocyte dysfunction". Circulation 119 (11): 1473–83. Mar 2009. doi:10.1161/CIRCULATIONAHA.108.838672. PMID 19273718.
- ↑ 75.0 75.1 75.2 "Nonsense-mediated mRNA decay and ubiquitin-proteasome system regulate cardiac myosin-binding protein C mutant levels in cardiomyopathic mice". Circulation Research 105 (3): 239–48. Jul 2009. doi:10.1161/CIRCRESAHA.109.201251. PMID 19590044.
- ↑ "How do MYBPC3 mutations cause hypertrophic cardiomyopathy?". Journal of Muscle Research and Cell Motility 33 (1): 75–80. May 2012. doi:10.1007/s10974-011-9268-3. PMID 22057632.
- ↑ "Research priorities in sarcomeric cardiomyopathies". Cardiovascular Research 105 (4): 449–56. Apr 2015. doi:10.1093/cvr/cvv019. PMID 25631582.
- ↑ "Impairment of the ubiquitin-proteasome system by truncated cardiac myosin binding protein C mutants". Cardiovascular Research 66 (1): 33–44. Apr 2005. doi:10.1016/j.cardiores.2005.01.004. PMID 15769446.
- ↑ "Ubiquitin-proteasome system impairment caused by a missense cardiac myosin-binding protein C mutation and associated with cardiac dysfunction in hypertrophic cardiomyopathy". Journal of Molecular Biology 384 (4): 896–907. Dec 2008. doi:10.1016/j.jmb.2008.09.070. PMID 18929575. http://repository.lib.tottori-u.ac.jp/files/public/0/4136/20180622151113992329/G14_15101A00024R.pdf.
- ↑ "The ubiquitin-proteasome system in cardiac dysfunction". Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 1782 (12): 749–63. Dec 2008. doi:10.1016/j.bbadis.2008.06.009. PMID 18634872. https://hal.archives-ouvertes.fr/hal-00501585/file/PEER_stage2_10.1016%252Fj.bbadis.2008.06.009.pdf.
- ↑ "The ubiquitin-proteasome system and nonsense-mediated mRNA decay in hypertrophic cardiomyopathy". Cardiovascular Research 85 (2): 330–8. Jan 2010. doi:10.1093/cvr/cvp247. PMID 19617224.
- ↑ "Ubiquitin-proteasome system and hereditary cardiomyopathies". Journal of Molecular and Cellular Cardiology 71: 25–31. Jun 2014. doi:10.1016/j.yjmcc.2013.12.016. PMID 24380728.
- ↑ 83.0 83.1 83.2 "Rescue of cardiomyopathy through U7snRNA-mediated exon skipping in Mybpc3-targeted knock-in mice". EMBO Molecular Medicine 5 (7): 1128–1145. Jul 2013. doi:10.1002/emmm.201202168. PMID 23716398.
- ↑ 84.0 84.1 "Repair of Mybpc3 mRNA by 5'-trans-splicing in a Mouse Model of Hypertrophic Cardiomyopathy". Molecular Therapy: Nucleic Acids 2 (7): e102. 2013. doi:10.1038/mtna.2013.31. PMID 23820890.
- ↑ "Defective proteolytic systems in Mybpc3-targeted mice with cardiac hypertrophy". Basic Research in Cardiology 107 (1): 235. Jan 2012. doi:10.1007/s00395-011-0235-3. PMID 22189562. https://zenodo.org/record/3411902.
- ↑ "Adrenergic stress reveals septal hypertrophy and proteasome impairment in heterozygous Mybpc3-targeted knock-in mice". Journal of Muscle Research and Cell Motility 33 (1): 5–15. May 2012. doi:10.1007/s10974-011-9273-6. PMID 22076249. https://zenodo.org/record/3413136.
- ↑ "Ubiquitin proteasome dysfunction in human hypertrophic and dilated cardiomyopathies". Circulation 121 (8): 997–1004. Mar 2010. doi:10.1161/CIRCULATIONAHA.109.904557. PMID 20159828.
- ↑ "Hypercontractile properties of cardiac muscle fibers in a knock-in mouse model of cardiac myosin-binding protein-C". The Journal of Biological Chemistry 276 (7): 5353–9. Feb 2001. doi:10.1074/jbc.M008691200. PMID 11096095.
- ↑ "Increased myofilament Ca2+ sensitivity and diastolic dysfunction as early consequences of Mybpc3 mutation in heterozygous knock-in mice". Journal of Molecular and Cellular Cardiology 52 (6): 1299–307. Jun 2012. doi:10.1016/j.yjmcc.2012.03.009. PMID 22465693.
- ↑ "Contractile dysfunction irrespective of the mutant protein in human hypertrophic cardiomyopathy with normal systolic function". Circulation: Heart Failure 5 (1): 36–46. Jan 2012. doi:10.1161/CIRCHEARTFAILURE.111.963702. PMID 22178992.
- ↑ "Perturbed length-dependent activation in human hypertrophic cardiomyopathy with missense sarcomeric gene mutations". Circulation Research 112 (11): 1491–505. May 2013. doi:10.1161/CIRCRESAHA.111.300436. PMID 23508784.
- ↑ "Contractile abnormalities and altered drug response in engineered heart tissue from Mybpc3-targeted knock-in mice". Journal of Molecular and Cellular Cardiology 63: 189–98. Oct 2013. doi:10.1016/j.yjmcc.2013.07.011. PMID 23896226.
- ↑ "hiPSC Modeling of Inherited Cardiomyopathies". Current Treatment Options in Cardiovascular Medicine 16 (7): 320. Jul 2014. doi:10.1007/s11936-014-0320-7. PMID 24838688.
- ↑ "Abnormal calcium handling properties underlie familial hypertrophic cardiomyopathy pathology in patient-specific induced pluripotent stem cells". Cell Stem Cell 12 (1): 101–13. Jan 2013. doi:10.1016/j.stem.2012.10.010. PMID 23290139.
- ↑ "Study familial hypertrophic cardiomyopathy using patient-specific induced pluripotent stem cells". Cardiovascular Research 104 (2): 258–69. Nov 2014. doi:10.1093/cvr/cvu205. PMID 25209314.
- ↑ 96.0 96.1 "Endothelin-1 induces myofibrillar disarray and contractile vector variability in hypertrophic cardiomyopathy-induced pluripotent stem cell-derived cardiomyocytes". Journal of the American Heart Association 3 (6): e001263. Dec 2014. doi:10.1161/JAHA.114.001263. PMID 25389285.
- ↑ "Adeno-associated virus vectors as therapeutic and investigational tools in the cardiovascular system". Circulation Research 114 (11): 1827–46. May 2014. doi:10.1161/CIRCRESAHA.114.302331. PMID 24855205.
- ↑ "Genetic therapies for RNA mis-splicing diseases". Trends in Genetics 27 (5): 196–205. May 2011. doi:10.1016/j.tig.2011.02.004. PMID 21497936.
- ↑ "Genome editing. The new frontier of genome engineering with CRISPR-Cas9". Science 346 (6213): 1258096. Nov 2014. doi:10.1126/science.1258096. PMID 25430774.
- ↑ "Development and applications of CRISPR-Cas9 for genome engineering". Cell 157 (6): 1262–78. Jun 2014. doi:10.1016/j.cell.2014.05.010. PMID 24906146.
- ↑ "Genome engineering using the CRISPR-Cas9 system". Nature Protocols 8 (11): 2281–308. Nov 2013. doi:10.1038/nprot.2013.143. PMID 24157548.
- ↑ "Regulation of alternative pre-mRNA splicing". Briefings in Functional Genomics & Proteomics 1 (3): 266–77. Oct 2002. doi:10.1093/bfgp/1.3.266. PMID 15239893.
- ↑ "Rescue of dystrophic muscle through U7 snRNA-mediated exon skipping". Science 306 (5702): 1796–9. Dec 2004. doi:10.1126/science.1104297. PMID 15528407. Bibcode: 2004Sci...306.1796G.
- ↑ "Spliceosome-mediated trans-splicing: the therapeutic cut and paste". The Journal of Investigative Dermatology 132 (8): 1959–66. Aug 2012. doi:10.1038/jid.2012.101. PMID 22495179.
- ↑ "Mybpc3 gene therapy for neonatal cardiomyopathy enables long-term disease prevention in mice". Nature Communications 5: 5515. 2014. doi:10.1038/ncomms6515. PMID 25463264. Bibcode: 2014NatCo...5.5515M.
Further reading
- "Contractile protein mutations and heart disease". Current Opinion in Cell Biology 8 (1): 97–105. Feb 1996. doi:10.1016/S0955-0674(96)80053-6. PMID 8791411.
- "Modulation of contractility in human cardiac hypertrophy by myosin essential light chain isoforms". Cardiovascular Research 37 (2): 381–404. Feb 1998. doi:10.1016/S0008-6363(97)00258-7. PMID 9614495. http://doc.rero.ch/record/301870/files/37-2-381.pdf.
- "Familial hypertrophic cardiomyopathy: from mutations to functional defects". Circulation Research 83 (6): 580–93. Sep 1998. doi:10.1161/01.res.83.6.580. PMID 9742053.
- "Genetics of hypertrophic cardiomyopathy in eastern Finland: few founder mutations with benign or intermediary phenotypes". Annals of Medicine 36 (1): 23–32. 2004. doi:10.1080/07853890310017161. PMID 15000344.
- "The interaction of C-protein with heavy meromyosin and subfragment-2". The Biochemical Journal 171 (3): 813–6. Jun 1978. doi:10.1042/bj1710813. PMID 352343.
- "Effect of C-protein on actomyosin ATPase". Biochimica et Biophysica Acta (BBA) - General Subjects 632 (2): 141–9. Oct 1980. doi:10.1016/0304-4165(80)90071-9. PMID 6448079.
- "Mutations in the cardiac myosin binding protein-C gene on chromosome 11 cause familial hypertrophic cardiomyopathy". Nature Genetics 11 (4): 434–7. Dec 1995. doi:10.1038/ng1295-434. PMID 7493025.
- "Cardiac myosin binding protein-C gene splice acceptor site mutation is associated with familial hypertrophic cardiomyopathy". Nature Genetics 11 (4): 438–40. Dec 1995. doi:10.1038/ng1295-438. PMID 7493026.
- "Phosphorylation switches specific for the cardiac isoform of myosin binding protein-C: a modulator of cardiac contraction?". The EMBO Journal 14 (9): 1952–60. May 1995. doi:10.1002/j.1460-2075.1995.tb07187.x. PMID 7744002.
- "Mapping of a novel gene for familial hypertrophic cardiomyopathy to chromosome 11". Nature Genetics 4 (3): 311–3. Jul 1993. doi:10.1038/ng0793-311. PMID 8358441.
- "A molecular map of the interactions between titin and myosin-binding protein C. Implications for sarcomeric assembly in familial hypertrophic cardiomyopathy". European Journal of Biochemistry 235 (1–2): 317–23. Jan 1996. doi:10.1111/j.1432-1033.1996.00317.x. PMID 8631348.
- "Organization and sequence of human cardiac myosin binding protein C gene (MYBPC3) and identification of mutations predicted to produce truncated proteins in familial hypertrophic cardiomyopathy". Circulation Research 80 (3): 427–34. Mar 1997. doi:10.1161/01.res.0000435859.24609.b3. PMID 9048664.
- "Novel splice donor site mutation in the cardiac myosin-binding protein-C gene in familial hypertrophic cardiomyopathy. Characterization Of cardiac transcript and protein". The Journal of Clinical Investigation 100 (2): 475–82. Jul 1997. doi:10.1172/JCI119555. PMID 9218526.
- "Molecular pathology of familial hypertrophic cardiomyopathy caused by mutations in the cardiac myosin binding protein C gene". Journal of Medical Genetics 35 (3): 205–10. Mar 1998. doi:10.1136/jmg.35.3.205. PMID 9541104.
- "Identification of a new missense mutation in MyBP-C associated with hypertrophic cardiomyopathy". Journal of Medical Genetics 35 (3): 253–4. Mar 1998. doi:10.1136/jmg.35.3.253. PMID 9541115.
- "Mutations in the gene for cardiac myosin-binding protein C and late-onset familial hypertrophic cardiomyopathy". The New England Journal of Medicine 338 (18): 1248–57. Apr 1998. doi:10.1056/NEJM199804303381802. PMID 9562578.
- "Double heterozygosity for mutations in the beta-myosin heavy chain and in the cardiac myosin binding protein C genes in a family with hypertrophic cardiomyopathy". Journal of Medical Genetics 36 (7): 542–5. Jul 1999. doi:10.1136/jmg.36.7.542. PMID 10424815.
External links
- Mass spectrometry characterization of MYBPC3 at COPaKB
- GeneReviews/NIH/NCBI/UW entry on Familial Hypertrophic Cardiomyopathy Overview
- Overview of all the structural information available in the PDB for UniProt: Q14896 (Myosin-binding protein C, cardiac-type) at the PDBe-KB.
 |