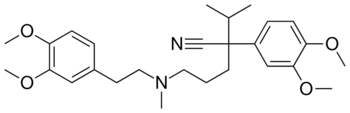
Chemistry:Verapamil
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /vɛˈræpəmɪl/ ve-RAP-ə-mil |
| Trade names | Isoptin, Calan, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a684030 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous |
| Drug class | Calcium channel blocker |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 35.1% |
| Metabolism | liver |
| Elimination half-life | 2.8–7.4 hours[6] |
| Excretion | kidney: 11% |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C27H38N2O4 |
| Molar mass | 454.611 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Verapamil, sold under various trade names,[1] is a calcium channel blocker medication used for the treatment of high blood pressure, angina (chest pain from not enough blood flow to the heart), and supraventricular tachycardia.[7] It may also be used for the prevention of migraines and cluster headaches.[8][9] It is given by mouth or by injection into a vein.[7]
Common side effects include headache, low blood pressure, nausea, and constipation.[7] Other side effects include allergic reactions and muscle pains.[10] It is not recommended in people with a slow heart rate or heart failure.[10] It is believed to cause problems for the fetus if used during pregnancy.[2] It is in the non–dihydropyridine calcium channel blocker family of medications.[7]
Verapamil was approved for medical use in the United States in 1981.[7][11] It is on the World Health Organization's List of Essential Medicines.[12][13] Verapamil is available as a generic medication.[7] Long acting formulations exist.[10] In 2020, it was the 151st most commonly prescribed medication in the United States, with more than 3 million prescriptions.[14][15]
Medical uses

Verapamil is used for controlling ventricular rate in supraventricular tachycardia (SVT) and migraine headache prevention.[16]
Verapamil is also used for the treatment of angina (chronic stable, vasospastic or Prinzmetal variant), unstable angina (crescendo, preinfarction), and for the prevention of paroxysmal supraventricular tachycardia (PSVT).[17]
Verapamil is a class-IV antiarrhythmic and more effective than digoxin in controlling ventricular rate.[18] Verapamil is not listed as a first line antihypertensive agent by the guidelines provided by JAMA in JNC-8.[19] However, it may be used to treat hypertension if patient has co-morbid atrial fibrillation or other types of arrhythmia.[20][21]
Verapamil is also used intra-arterially to treat cerebral vasospasm.[22] Verapamil is used to treat the condition cluster headache.[23] Tentative evidence supports the use of verapamil topically to treat plantar fibromatosis.[24]
Verapamil use in people with recent onset type-1 diabetes may improve pancreatic beta cell function. In a meta-analysis involving data from 2 randomized controlled trials (113 patients with recent onset type-1 diabetes), Dutta et al. demonstrated use of verapamil over 1 year was associated with significantly higher C-peptide area under the curve levels at 1-year [MD 0.27 nmol/L (95%CI: 0.19-0.35); P < 0.01]. Higher C-peptide levels means better pancreatic insulin production and beta cell function.[25]
Contraindications
Use of verapamil is generally avoided in people with severe left ventricular dysfunction, hypotension (systolic blood pressure less than 90 mm Hg), cardiogenic shock, and hypersensitivity to verapamil.[4] It is also contraindicated in people with atrial flutter or fibrillation and an existing accessory tract such as in Wolff-Parkinson-White syndrome.[26][4]
Side effects
The most common side effect of verapamil is constipation (7.3%). While the definite mechanism by which Verapamil causes constipation has not been studied, studies have been conducted to rule out mechanisms of actions that might yield this adverse effect. In a study conducted by The National Library of Medicine titled, "Effect of Verapamil on the Human Intestinal Transit", the study found that verapamil does not have an effect on upper GI transit but rather in the colon.[27]
Other side effects include dizziness (3.3%), nausea (2.7%), low blood pressure (2.5%), and headache 2.2%. Other side effects seen in less than 2% of the population include: edema, congestive heart failure, pulmonary edema, diarrhea, fatigue, elevated liver enzymes, shortness of breath, low heart rate, atrioventricular block, rash and flushing.[4] Along with other calcium channel blockers, verapamil is known to induce gingival enlargement.[28]
Overdose
Acute overdose is often manifested by nausea, weakness, slow heart rate, dizziness, low blood pressure, and abnormal heart rhythms. Plasma, serum, or blood concentrations of verapamil and norverapamil, its major active metabolite, may be measured to confirm a diagnosis of poisoning in hospitalized patients or to aid in the medicolegal investigation of fatalities. Blood or plasma verapamil concentrations are usually in a range of 50–500 μg/L in persons on therapy with the drug, but may rise to 1–4 mg/L in acute overdose patients and are often at levels of 5–10 mg/L in fatal poisonings.[29][30]
Mechanism of action
Verapamil's mechanism in all cases is to block voltage-dependent calcium channels.[4] In cardiac pharmacology, calcium channel blockers are considered class-IV antiarrhythmic agents. Since calcium channels are especially concentrated in the sinoatrial and atrioventricular nodes, these agents can be used to decrease impulse conduction through the AV node, thus protecting the ventricles from atrial tachyarrhythmias. Verapamil is also a Kv voltage gated potassium channel blocker.[31]
Calcium channels are also present in the smooth muscle lining blood vessels. By relaxing the tone of this smooth muscle, calcium channel blockers dilate the blood vessels. This has led to their use in treating high blood pressure and angina pectoris. The pain of angina is caused by a deficit in oxygen supply to the heart.
Calcium channel blockers like verapamil dilate the coronary blood vessels, which increases the supply of blood and oxygen to the heart. They also cause dilatation of systemic peripheral vessels as well, causing a reduction in the workload of the heart. Thereby reducing myocardial oxygen consumption.[4]
Cluster headaches
Preventive therapy with verapamil is believed to work because it has an effect on the circadian rhythm and on CGRPs. As CGRP-release is controlled by voltage-gated calcium channels.[32]
Pharmacokinetic details
More than 90% of verapamil is absorbed when given orally,[4] but due to high first-pass metabolism, bioavailability is much lower (10–35%). It is 90% bound to plasma proteins and has a volume of distribution of 3–5 L/kg. It takes 1 to 2 hours to reach peak plasma concentration after oral administration.[4] It is metabolized in the liver to at least 12 inactive metabolites (though one metabolite, norverapamil, retains 20% of the vasodilatory activity of the parent drug). As its metabolites, 70% is excreted in the urine and 16% in feces; 3–4% is excreted unchanged in urine. This is a nonlinear dependence between plasma concentration and dosage. Onset of action is 1–2 hours after oral dosage. Half-life is 5–12 hours (with chronic dosages). It is not cleared by hemodialysis. It is excreted in human milk. Because of the potential for adverse reaction in nursing infants, nursing should be discontinued while verapamil is administered.
Verapamil has been reported to be effective in both short-term[33] and long-term treatment of mania and hypomania.[34] Addition of magnesium oxide to the verapamil treatment protocol enhances the antimanic effect.[35]
Veterinary use
Intra-abdominal adhesions are common in rabbits following surgery. Verapamil can be given postoperatively in rabbits which have suffered trauma to abdominal organs to prevent formation of these adhesions.[36][37][38] Such effect was not documented in another study with ponies.[39]
Uses in cell biology
Verapamil inhibits the ATP-binding cassette (ABC) transporter family of proteins found in stem cells and has been used to study cancer stem cells (CSC) within head and neck squamous cell carcinomas.[40]
Verapamil is also used in cell biology as an inhibitor of drug efflux pump proteins such as P-glycoprotein and other ABC transporter proteins.[41][40] This is useful, as many tumor cell lines overexpress drug efflux pumps, limiting the effectiveness of cytotoxic drugs or fluorescent tags. It is also used in fluorescent cell sorting for DNA content, as it blocks efflux of a variety of DNA-binding fluorophores such as Hoechst 33342. Radioactively labelled verapamil and positron emission tomography can be used with to measure P-glycoprotein function.
See also
References
- ↑ 1.0 1.1 "Verapamil". https://www.drugs.com/international/verapamil.html.
- ↑ 2.0 2.1 "Verapamil Use During Pregnancy". 18 November 2019. https://www.drugs.com/pregnancy/verapamil.html.
- ↑ "Securon SR - Summary of Product Characteristics (SmPC)". 17 May 2017. https://www.medicines.org.uk/emc/product/6243/smpc.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 "Calan- verapamil hydrochloride tablet, film coated". 17 December 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=55d5f933-42ff-4c80-a102-0ccb7f76b082.
- ↑ Human Medicines Evaluation Division (14 October 2020). "Active substance(s): verapamil". List of nationally authorised medicinal products. European Medicines Agency. https://www.ema.europa.eu/documents/psusa/verapamil-list-nationally-authorised-medicinal-products-psusa/00003105/202001_en.pdf.
- ↑ "Pharmacologic Options for Treatment of Ischemic Disease". Cardiovascular Therapeutics: A Companion to Braunwald's Heart Disease. Elsevier. 2013. pp. 83–130. doi:10.1016/b978-1-4557-0101-8.00007-2. ISBN 978-1-4557-0101-8. "The elimination half-life of standard verapamil tablets is usually 3 to 7 hours,..."
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 "Verapamil Hydrochloride". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/verapamil-hydrochloride.html.
- ↑ "Management of cluster headache". CNS Drugs 26 (7): 571–580. July 2012. doi:10.2165/11632850-000000000-00000. PMID 22650381.
- ↑ "Diagnosis and Treatment of Childhood Migraine". Current Treatment Options in Neurology 18 (11): 48. November 2016. doi:10.1007/s11940-016-0431-4. PMID 27704257.
- ↑ 10.0 10.1 10.2 WHO Model Formulary 2008. World Health Organization. 2009. ISBN 9789241547659.
- ↑ "Isoptin: FDA-Approved Drugs". https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=018485.
- ↑ World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. 2021. WHO/MHP/HPS/EML/2021.02.
- ↑ "The Top 300 of 2020". https://clincalc.com/DrugStats/Top300Drugs.aspx.
- ↑ "Verapamil - Drug Usage Statistics". https://clincalc.com/DrugStats/Drugs/Verapamil.
- ↑ Koda-Kimble and Young's Applied Therapeutics: The Clinical Use of Drugs (10th ed.). USA: LWW. 2012. pp. 497, 1349. ISBN 978-1609137137.
- ↑ "Verapamil". StatPearls [Internet].. Treasure Island (FL): StatPearls Publishing. 2023. https://www.ncbi.nlm.nih.gov/books/NBK538495/.
- ↑ "Detection, isolation and characterization of principal synthetic route indicative impurities in verapamil hydrochloride". Scientia Pharmaceutica 79 (3): 555–568. 2011. doi:10.3797/scipharm.1101-19. PMID 21886903.
- ↑ "2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8)". JAMA 311 (5): 507–520. February 2014. doi:10.1001/jama.2013.284427. PMID 24352797.
- ↑ Koda-Kimble and Young's Applied Therapeutics: The Clinical Use of Drugs (Tenth, North American ed.). LWW. 2012. pp. 320–22. ISBN 978-1609137137.
- ↑ "Management of cluster headache". American Family Physician 71 (4): 717–724. February 2005. PMID 15742909. http://www.aafp.org/afp/2005/0215/p717.html. Retrieved 2015-11-04.
- ↑ "Endovascular treatment of medically refractory cerebral vasospasm following aneurysmal subarachnoid hemorrhage". AJNR. American Journal of Neuroradiology 31 (10): 1911–1916. November 2010. doi:10.3174/ajnr.A2183. PMID 20616179.
- ↑ Blueprints Neurology. Lippincott Williams & Wilkins. 1 January 2009. pp. 71–. ISBN 978-0-7817-9685-9. https://books.google.com/books?id=vj3td6GY0gcC&pg=PA71. Retrieved 14 November 2010.
- ↑ "The etiology, evaluation, and management of plantar fibromatosis". Orthopedic Research and Reviews 11: 1–7. 2018-12-17. doi:10.2147/ORR.S154289. PMID 30774465.
- ↑ "Verapamil improves One-Year C-Peptide Levels in Recent Onset Type-1 Diabetes: A Meta-Analysis.". Indian J Endocrinol Metab 27 (3): 192–200. May–Jun 2023. doi:10.4103/ijem.ijem_122_23. PMID 37583402.
- ↑ "Securon 2.5 mg/ml IV Intravenous Injection - Summary of Product Characteristics (SmPC)". 24 November 2016. https://www.medicines.org.uk/emc/product/979/smpc.
- ↑ "Effect of verapamil on human intestinal transit". Digestive Diseases and Sciences 37 (6): 919–924. June 1992. doi:10.1007/BF01300391. PMID 1587197.
- ↑ "Calcium antagonist-induced gingival hyperplasia". Annals of Internal Medicine 120 (8): 663–664. April 1994. doi:10.7326/0003-4819-120-8-199404150-00006. PMID 8135450.
- ↑ "Monitoring of verapamil enantiomers concentration in overdose". Clinical Toxicology 44 (2): 169–171. 2006. doi:10.1080/15563650500514541. PMID 16615674.
- ↑ Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, California: Biomedical Publications. 2008. pp. 1637–39.
- ↑ "Potassium channel currents in rat mesenchymal stem cells and their possible roles in cell proliferation". Clinical and Experimental Pharmacology & Physiology 35 (9): 1077–1084. September 2008. doi:10.1111/j.1440-1681.2008.04964.x. PMID 18505444.
- ↑ "Verapamil and Cluster Headache: Still a Mystery. A Narrative Review of Efficacy, Mechanisms and Perspectives". Headache 59 (8): 1198–1211. September 2019. doi:10.1111/head.13603. PMID 31339562.
- ↑ "Antimanic effects of verapamil". The American Journal of Psychiatry 141 (12): 1602–1603. December 1984. doi:10.1176/ajp.141.12.1602. PMID 6439057.
- ↑ "Verapamil and lithium in maintenance therapy of manic patients". Journal of Clinical Pharmacology 27 (12): 980–982. December 1987. doi:10.1002/j.1552-4604.1987.tb05600.x. PMID 3325531.
- ↑ "Magnesium oxide augmentation of verapamil maintenance therapy in mania". Psychiatry Research 93 (1): 83–87. February 2000. doi:10.1016/S0165-1781(99)00116-X. PMID 10699232.
- ↑ "The effect of verapamil and other calcium antagonists on chemotaxis of polymorphonuclear leukocytes". Biochemical Pharmacology 33 (1): 35–39. January 1984. doi:10.1016/0006-2952(84)90367-8. PMID 6704142.
- ↑ "Modulation of fibroblast-induced clot retraction by calcium channel blocking drugs and the monoclonal antibody ALB6". Journal of Cellular Physiology 125 (3): 420–426. December 1985. doi:10.1002/jcp.1041250309. PMID 3864783.
- ↑ "Reduction of primary postoperative adhesion formation under calcium channel blockade in the rabbit". The Journal of Surgical Research 48 (1): 42–45. January 1990. doi:10.1016/0022-4804(90)90143-P. PMID 2296179.
- ↑ "Failure of calcium channel blockade to prevent intra-abdominal adhesions in ponies". Veterinary Surgery 22 (6): 496–500. 1993. doi:10.1111/j.1532-950X.1993.tb00427.x. PMID 8116206.
- ↑ 40.0 40.1 "Characterization of side populations in HNSCC: highly invasive, chemoresistant and abnormal Wnt signaling". PLOS ONE 5 (7): e11456. July 2010. doi:10.1371/journal.pone.0011456. PMID 20625515. Bibcode: 2010PLoSO...511456S.
- ↑ "P-glycoproteins and multidrug resistance". Annual Review of Pharmacology and Toxicology 36: 161–183. 1996. doi:10.1146/annurev.pa.36.040196.001113. PMID 8725386.
External links
- "Verapamil". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/verapamil.
- "Verapamil hydrochloride". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/verapamil%20hydrochloride.
 |
