Biology:Tylosaurus
Tylosaurus (/ˌtaɪˈloʊˈsɔːrəs/; "knob lizard") is a genus of russellosaurine mosasaur (an extinct group of predatory marine lizards) that lived about 92 to 66 million years ago during the Turonian to Maastrichtian stages of the Late Cretaceous. Its fossils have been found primarily around North Atlantic Ocean including in North America, Europe, and Africa.
Research history
Possible first finds
The earliest Tylosaurus fossils were likely discovered by various Native American peoples and may have been the source of much of their folklore, with the earliest known ones dating back to well before the arrival of European settlers, around the 1500s. More recent accounts from peoples living in the Great Plains even speak of an ancient era ruled by massive aquatic creatures that were in constant combat with thunderbirds and were petrified by them. The considerable presence of fossils of large mosasaurs such as Tylosaurus and pterosaurs such as Pteranodon in this region may have been the origins of these myths.[1]
In 1804, the Lewis and Clark Expedition discovered a now-lost fossil skeleton alongside the Missouri River, which was identified as a 45-foot (14 m) long fish.[2] In 2003, Richard Ellis speculated that the remains may have belonged to Mosasaurus missouriensis.[3] Alternatively, a 2007 study led by Robert W. Meredith and colleagues suggested that the fossils would possibly come from a tylosaurine mosasaur based on the measurements cited by Clark and Gass and the evidence of Tylosaurus fossils that have been found in the Missouri River. However, the authors also mentioned the possibility that the remains would also come from an elasmosaurid plesiosaur, which are also known from the river, although being rarer.[4]
First formal discoveries
Tylosaurus was the third new genus of mosasaur to be described from North America behind Clidastes and Platecarpus and the first in Kansas.[5] The early history of the genus as a taxon was subject to complications spurred by the infamous rivalry between American paleontologists Edward Drinker Cope and Othniel Charles Marsh during the Bone Wars.[5][6] The type specimen was described by Cope in 1869 based on a fragmentary skull measuring nearly 5 feet (1.5 m) in length and thirteen vertebrae lent to him by Louis Agassiz of the Harvard Museum of Comparative Zoology.[7] The fossil, which remains in the same museum under the catalog number MCZ 4374, was recovered from a deposit of the Niobrara Formation located in the vicinity of Monument Rocks[8] near the Union Pacific Railroad at Fort Hays, Kansas.[9] Cope's first publication of the fossil was very brief and was named Macrosaurus proriger, the genus being a preexisting European mosasaur taxon.[5][7] The specific epithet proriger means "prow-bearing", which is in reference to the specimen's unique prow-like elongated rostrum[10][11] and is derived from the Latin word prōra (prow) and suffix -gero (I bear).[12] In 1870, Cope published a more thorough description of MCZ 4374. Without explanation, he moved the species into another European genus Liodon and declared his original Macrosaurus proriger a synonym.[5][9]
In 1872, Marsh argued that Liodon proriger is taxonomically distinct from the European genus and must be assigned a new one. For this, he erected the genus Rhinosaurus, which means "nose lizard" and is a portmanteau derived from the Ancient Greek words ῥίς (rhī́s, meaning "nose") and σαῦρος (saûros, meaning "lizard").[13][14] Cope responded by arguing that Rhinosaurus was already a preoccupied synonym of Liodon. He disagreed with Marsh's arguments but proposed that in case Marsh was indeed correct, the genus name Rhamphosaurus should be used.[15] Marsh later discovered that the taxon Rhamphosaurus was preoccupied as a genus of lizard named in 1843. As a result, he suggested a move to a newly erected genus named Tylosaurus.[16] This name means "knob lizard" in another reference to the elongated rostrum characteristic of the genus. It is derived from the Latin tylos (knob) and Ancient Greek σαῦρος.[11] Despite coining the new genus, Marsh never formally transferred this Rhinosaurus species to Tylosaurus; this was first done in 1873 by Joseph Leidy.[17][18] Tylosaurus subsequently became the almost universally accepted genus to include this species, the exception to this adoption being Cope,[15] who refused to accept Marsh's new genus and continued to refer to its species as Liodon.[10] Cope's persistence can be seen in his 1874 description of another species of Tylosaurus, which he named Liodon nepaeolicus. The type specimen of this species was discovered by geologist Benjamin Franklin Mudge near the Solomon River, and consists of several cranial fragments and a dorsal vertebra[19] now catalogued as AMNH 1565. This species, whose specific epithet refers to Nepaholla, the Native American name for the Salomon River[17] is formally transferred to the genus Tylosaurus in 1894 by John Campbell Merriam.[20]
Later discoveries and other species

In his major work published in 1967, Dale A. Russell recognized only two valid species in Tylosaurus, namely T. proriger and T. nepaeolicus.[21] However, throughout the 19th and 20th centuries, many species of mosasaurs coming from around the world, originally described as being from separate genera, were now recognized as belonging to Tylosaurus.[22]
In 1885, Louis Dollo described the genus and species Hainosaurus bernardi from an almost complete but poorly preserved skeleton discovered in a phosphate quarry in the Ciply Basin in Belgium,[23] the specimen having since been catalogued as IRSNB R23.[24] The genus name means "lizard from the Haine", in reference to the eponymous river located nearby the Ciply Basin. The specific epithet is named in honor of Leopold Bernard, who made the excavation and exhumation of the specimen possible.[23] In 1988, a second species historically pertained to Hainosaurus was described by Elizabeth Nicholls based on a partial skeleton catalogued as MT 2 and having been discovered in Manitoba, Canada. The specific epithet refers to the type locality of the taxon, namely the Pembina Member of the Pierre Shale.[25] The attribution of H. pembinensis to Hainosaurus is first discussed by Johan Lindgren in 2005,[26] but it was in a revision published in 2010 that the species was moved to Tylosaurus by Timon Bullard and Michael Caldwell, being then renamed as T. pembinensis. In this same revision, the authors suggested that a redescription of the type species H. bernardi would be necessary in order to know if Hainosaurus should be maintained as a distinct genus.[27] This redescription was finally carried out by Paulina Jimenez-Huidobro and Caldwell in 2016, in which they transferred the species to Tylosaurus, being then renamed as T. bernardi.[28] Although this new combination has been widely recognized since, some authors nevertheless suggest continuing to maintain the genus Hainosaurus as distinct, justified in particular on the basis of dental traits not detailed in the 2016 revision.[29][30]
In 1896, Armand Thevenin described a new mosasaurid on the basis of a partial skull discovered at Éclusier-Vaux, in Somme, France.[31] In his description, Thevenin thinks that this specimen, since catalogued as MNHN 1896–15,[24][26] would represent a species of Mosasaurus, naming it Mosasaurus gaudryi. The specific epithet is named in honor of his mentor Jean Albert Gaudry, the latter having previously studied the skull and thinking that it would come from a species of Liodon.[31] In 1992, Theagarten Lingham-Soliar reassigned the species to Hainosaurus, the latter having previously interpreted the holotype of this taxon as an additional specimen of H. bernardi.[24] In 2005, Lindgren moved this species to Tylosaurus, notably due to its dental characteristics being closer to other lineages of the genus.[26]

In 1963, Per Ove Persson identified a new mosasaurid on the basis of isolated teeth discovered in a deposit located in an area called Ivö Klack, near Ivö Lake in the Kristianstad Basin in Scania, Sweden. Fossils from this same mosasaurid have been documented in this area since 1836, but it is from that year onwards that they are described as coming from a subspecies of Mosasaurus hoffmannii, being then named M. hoffmannii ivoensis, the second specific epithet referring to the type locality.[32] In 1967, Russell elevated the taxon to a separate species within the genus, and assigned to it fossils from the Niobrara Formation of Kansas, including a partial skull.[33] When the taxon was significantly revised in a in 2002 study, being reassigned to Tylosaurus, Lindgren and Mikael Siverson referred additional fossils to this latter that had been discovered at Ivö Klack, including cranial and vertebral remains. In their study, the authors also found that Russell's attributions of the Kansas fossils to this species were erroneous, the remains coming from a distinct taxon.[34] In a 2008 paper, Caldwell and colleagues suggested that T. ivoensis might belong in the related genus Taniwhasaurus based on its dental features and its fossils distribution, but the authors see this as a subject for another study.[35]
In 1964, Miguel Telles Antunes described the species Mosasaurus iembeensis from a partial skull excavated from the Itombe Formation near the town of Iembe (hence the name), Angola.[36] In 1992, Lingham-Soliar argued that the cranial features were not consistent with those of Mosasaurus and were more characteristic of Tylosaurus, the species being renamed as T. iembeensis. However, the author did not identify the holotype skull, which he considered to reside in the collections of the NOVA University Lisbon without a catalogue number,[24] and it is since 2006 reported as being destroyed in a fire.[37] In 2012, Octávio Mateus and colleagues reported that an additional specimen of T. iembeensis consisting of fragmentary cranial elements was recovered during an expedition to the locality of the since-destroyed holotype, although it was not figured or formally described.[38]

In 2005, Michael J. Everhart described the species T. kansasensis based on several specimens that had been discovered in Kansas, again in the fossil record of the Niobrara Formation. The holotype specimen consists of a well-preserved skull and six cervical vertebrae cataloged as FHSM VP-2295, which was discovered in 1968 in Ellis County.[39] The validity of this species was questioned as early as 2007 by Caldwell,[40] to the point that in a 2016 study led with Jiménez-Huidobro and other authors, the latter considers it a juvenile form of T. nepaeolicus, thus making the first name a junior synonym of the second.[41] This is disputed by Everhart himself in a 2017 book, but he only comments on the study as "poorly researched and written" without detailing how.[40] An ontogenetic review of Tylosaurus conducted by Robert F. Stewart and Jordan Mallon in 2018 favors maintaining T. kansasensis as valid,[42] while another conducted in 2020 by Amelia R. Zietlow prefers to follow the advice set out in the 2016 revision.[43]
In 2006, Bullard wrote a Master of Science thesis describing the species T. saskatchewanensis from a partial skeleton catalogued as RSM P2588.1, known from the Bearpaw Formation.[44] This specimen, nicknamed "Omācīw" (meaning "hunter" in Cree), was discovered in 1994 near Herbert Ferry, at the Lake Diefenbaker, Saskatchewan.[45][46] Although originally described informally and via incompletely prepared fossils, the proposed taxon was nevertheless recognized as valid in some subsequent studies.[28][35] In 2018, Bullard co-authored a multi-author study led by Jiménez-Huidobro which formally described Omācīw, which by then was more fully prepared, and confirmed its identity as belonging to a distinct species.[47]
In 2020, Samuel Garvey wrote a thesis on a partial skull of Tylosaurus catalogued as TMP 2014.011.0001, known from the Puskwaskau Formation. With visibly distinct features from other species and having been discovered approximately 55 kilometers (34 mi) northeast of Grande Prairie, Alberta, this makes the specimen the northernmost known occurrence of the genus, being then named T. 'borealis', in reference to its northernly presence.[48]
Depiction history

When Cope described the holotype specimen of T. proriger in 1870, he visualized it as an "excessively elongated reptile", due to the morphology of the caudal vertebrae which suggested this.[9] Taking into account his descriptions, this would result in a sea serpent-like reptile reaching lengths rivaling those of the largest cetaceans.[49][50][51] The following year, Cope added more details to his visualization of the animal. For him, the head of Tylosaurus would be conical in shape, with eyes on top, and having a jaw connected to a throat similar to that of a pelican, thus facilitating the entry of its prey. Still according to Cope, the animal would have had only the flippers located at the front of the body, those at the back being absent.[52][49] The tail is seen as long and flat, used in eel-like locomotion.[49] This depiction was followed in various works published during the late 19th century, although some depictions also depict the animal with a long neck.[53][51]

In a major revision of mosasaurs published in 1898, Samuel Wendell Williston provided a new anatomical description of Tylosaurus that corrected many of the misconceptions of earlier paleontologists about the genus. Specifically, his paper included a rigorous skeletal reconstruction of T. proriger based on three partial specimens from the collections of the University of Kansas Natural History Museum.[54][55] Thus, this reconstruction depicts the animal as very mobile marine predator with four flippers, a short neck and a much shorter tail than previous depictions, Williston also fixing a maximum body measurement close to those still cited today, i. e. 11 m (36 ft) long.[55] Despite the fact that the spinal column is drawn as straight and not as curved,[lower-alpha 1] this reconstruction is still recognized as valid by the scientific community.[56] The discovery of the first known substantially complete skeleton of Tylosaurus was revealed as early as 1899 by Henry Fairfield Osborn,[57] followed by other more or less similar finds which were made from the beginning of the 1900s.[58]
Description
Tylosaurus was a type of derived mosasaur, or a latecoming member with advanced evolutionary traits such as a fully aquatic lifestyle. As such, it had a streamlined body, an elongated tail ending with a downturn supporting a two-lobed fin, and two pairs of flippers. While in the past derived mosasaurs were depicted as akin to giant flippered sea snakes, it is now understood that they were more similar in build to other large marine vertebrates such as ichthyosaurs, marine crocodylomorphs, and archaeocete whales through convergent evolution.[59][60]
Size

Some species of Tylosaurus are among the largest known mosasaurs. The largest well-known specimen, a skeleton of T. proriger from the University of Kansas Natural History Museum nicknamed "Bunker" (KUVP 5033), has been estimated to measure between 12–15.8 meters (39–52 ft) long.[17][61] A fragmentary skeleton of another T. proriger from the Sternberg Museum of Natural History (FHSM VP-2496) may be from an even larger individual; Everhart estimated the specimen to come from a 14 meters (46 ft) individual[62] compared to his 12 meters (39 ft) estimate for Bunker.[63] The genus exhibits Cope's rule, in which its body size has been observed to generally increase over geologic time.[17] In North America, the earliest representatives of Tylosaurus during the Turonian[64] and Coniacian (90-86 mya), which included early T. nepaeolicus and its precursors, typically measured 5–7 meters (16–23 ft) long[17] and weighed between 200–500 kilograms (440–1,100 lb).[65] During the Santonian (86-83 mya), T. nepaeolicus and newly-appearing T. proriger were 8–9 meters (26–30 ft) long[17] and weighed around 1,100 kilograms (2,400 lb).[66] By the Early Campanian, T. proriger attained lengths of 13–14 meters (43–46 ft).[67] Everhart speculated that because mosasaurs continuously grew throughout their lifetime, it would have been possible for some extremely old Tylosaurus individuals to reach 20 meters (66 ft) in absolute maximum length. However, he stressed the lack of fossil evidence suggesting such sizes and the odds against any being preserved.[68]
Other Campanian-Maastrichtian species were similarly large. The most recent maximum estimate for T. bernardi is 12.2 meters (40 ft) by Lindgren (2005); historically the species was erroneously estimated at even larger sizes of 15–17 meters (49–56 ft).[26] A reconstruction of T. saskatchewanensis by the Royal Saskatchewan Museum estimated a total length of over 9.75 meters (32.0 ft).[45] A mounted skeleton of T. pembinensis, nicknamed "Bruce," at the Canadian Fossil Discovery Centre measures at 13.05 meters (42.8 ft) long and was awarded a Guinness World Records for "Largest mosasaur on display" in 2014.[69] However, the skeleton was assembled for display prior[70] to Bullard and Caldwell (2010)'s reassessment that found the species' number of vertebrae to be exaggerated.[27] T. "borealis" is estimated at 6.5–8 meters (21–26 ft) in total length.[48]
Skull

The largest known skull of Tylosaurus is T. proriger KUVP 5033 (the "Bunker" specimen), estimated at 1.7 meters (5.6 ft) long.[43] Depending on age and individual variation,[43] Tylosaurus skulls were between 13 and 14% of the total skeleton length.[71] The head was strongly conical and the snout proportionally longer than most mosasaurs, with the exception of Ectenosaurus.[72]
Cranium
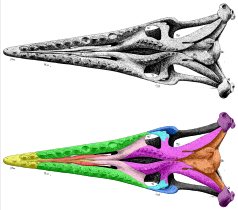
The most recognizable characteristic of Tylosaurus is the elongated edentulous rostrum that protrudes from its snout, for which the genus is named. This is formed by the elongation of the front end of the premaxilla[24] and dentary.[47] The rostrum was small and acutely angled at birth, but rapidly developed into a blunt, elongated "knob." The snout is heavily built, supported by a broad and robust internarial bar (comprising the posterodorsal process of the premaxilla, nasals, and anterior process of the frontal).[24] The snout holds the terminal branches for the trigeminal nerves through randomly scattered foramina[73] on the rostrum and along the ventral margin of the maxilla, above the gum line.[74]
The premaxilla, maxilla, and frontal bones border the external nares, or body nostril openings; unlike other mosasaurs, the prefrontal bones are excluded from the border of the nares by a long posterodorsal process of the maxilla.[22] The nares open above the fourth maxillary tooth anteriorly in T. proriger and T. pembinensis,[27][lower-alpha 2] between the third and fourth tooth in T. nepaeolicus,[27] and posterior to the fourth tooth in T. bernardi.[28] Nare length relative to skull length varied between species: it is proportionally short in T. proriger (20-27% skull length[75][34]), T. bernardi (24% skull length[27]), and T. gaudryi (25-27% skull length),[34] and long in T. pembinensis (28-31% skull length).[27] The nasal bones were either free-floating or lightly articulated to the internarial bar,[24] did not contact the frontal,[75] and were not fused to each other as they are in extant varanid lizards. The nasals' loose association with the rest of the skull in Tylosaurus and other mosasaurs may be why the bones are frequently lost and therefore exceedingly rare;[75] Tylosaurus is one of the only mosasaurs in which the nasal bones are clearly documented;[24] the other is the holotype of Plotosaurus, although one of the bones is missing.[76]
The external nares lead to the choanae (internal nares) in the palate, which provide passage from the nostrils to the throat.[77] In Tylosaurus, they are shaped like a compressed teardrop and bordered by the vomers, palatines, and the maxilla.[78] Anterior to the choanae, each vomer borders the fenestra for the Jacobson's organ, which is involved in the tongue-based sense of smell. It begins opposite of the fourth maxillary tooth in Tylosaurus,[79] and also ends immediately past the fifth maxillary tooth in T. bernardi.[24] The exit point for the veins leading to sinuses inside the palatine occur right in front of the Jacobson's organ between the vomers and maxilla. This differs from living varanids, where the exit occurs behind the organ.[78]
The frontal bone in Tylosaurus usually, but not always, possesses a low midline crest. It is most prominent in T. proriger,[22][41] and is moderately developed in T. saskatchewanensis[47] and T. bernardi, extending onto the premaxilla in the latter.[28] The frontal crest is present but poorly developed in most T. nepaeolicus skulls, and occasionally lost in some mature individuals.[43] The frontal overlaps the prefrontals and postorbitofrontals above the orbits (eye sockets), and the parietal posteriorly. The position of the pineal eye on the parietal is variable, either appearing close to the frontoparietal suture or contacting it.[28][41] The orbits are bordered by the prefrontal, lacrimal, postorbitofrontal, and jugal bones. A diagnostic feature of Tylosaurus is that the prefrontals and postorbitofrontals overlap above the orbits, preventing contribution of the frontal.[22] The jugal forms the bottom of the orbit; in Tylosaurus, it is L-shaped and has a distinctive serif-like extension at the lower back corner of the junction between the horizontal and vertical rami (arms) called the posteroventral process.[lower-alpha 3][41] The vertical ramus is overlapped by the postorbitofrontal in most species,[47][27][41] and the horizontal ramus overlaps the maxilla.[27] In T. bernardi, the vertical ramus is not overlapped but joins with the postorbitofrontal by a suture, and is much thicker than the horizontal ramus.[28]

The quadrate bones (homologous to the incus in mammals) are located at the back of the skull, articulating the lower jaw to the cranium[80] and holding the eardrums.[81] The complex anatomy of the bone[82] renders it extremely diagnostic, even to the species level.[22] In lateral view, the quadrate resembles a hook in immature T. nepaeolicus and T. proriger individuals, but in adult forms for both species[43] and in T. bernardi,[22][28] T. pembinensis,[27] and T. saskatchweanensis it takes on a robust oval-like shape.[47] The eardrum (tympanum) attached to the lateral surface of the bone within a bowl-like depression called the alar conch.[81] The conch is shallow in T. nepaeolicus,[82] T. proriger, and T. bernardi,[22] and deep in T. pembinensis[82] and T. saskatchewanensis.[22] The alar rim is thin in T. nepaeolicus, T. proriger,[43] and T. bernardi,[28][43] and thick in T. bernardi, T. pembinensis,[28] and T. saskatchewanensis.[47] The suprastapedial process is a hook-like extension of bone that curves posteroventrally from the apex of the shaft into an incomplete loop, and it likely served as the attachment point for the depressor mandibulae muscles that opened the lower jaw.[27][83][82] The process is slender and proportionally long in immature T. nepaeolicus and T. proriger, and thickened as the animals matured.[43] The process is of similar length to T. proriger in T. saskatchwanensis[47] and shorter in T. bernardi.[28] In T. pembinensis, it abruptly turns medially at a 45° downward angle.[27] A similar deflection appears in some juvenile T. nepaeolicus quadrates.[41] Emerging from the posteroventral margin of the alar conch is the infrastapedial process. Its shape appears to changes ontogenetically in T. nepaeolicus and T. proriger; in the former, the process is absent in juveniles but appears as a small bump in adults, while in T. proriger, it is present as a subtle point in juveniles of and becomes a distinctively broad semicircle in adults.[43] The process is small in T. bernardi,[28] and in T. pembinensis[27] and T. saskatchewanensis,[47] it is rounded. In T. saskatchewanensis, the suprastapedial process almost touches the infrastapedial process.[47] At the bottom of the shaft is the mandibular condyle, which forms the joint between the quadrate and the lower jaw. It is rounded in shape in adults.[82][28][47][43] On the medial surface of the bone, a thick, pillar-like vertical ridge often protrudes beyond the dorsal margin of the quadrate so that it is visible in lateral view.[82]
Jaws and teeth
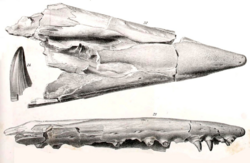
The upper jaws include the premaxilla and maxilla, and the lower jaws include the dentary, splenial, coronoid, angular, surangular, and prearticluar-articular (like other squamates, the prearticular is fused to the articular). The premaxilla, maxilla, and dentary house the marginal dentition, and the pterygoids house palatal dentition. On each side of the skull, Tylosaurus had 2 premaxillary teeth, 12 to 13 maxillary teeth, 13 dentary teeth, and 10 to 11 pterygoid teeth.[22] The dentition is homodont, meaning that all teeth are nearly identical in size and shape,[84][27][85] with the exception of the pterygoid teeth, which are smaller and more recurved than the marginal teeth.[34]
Tylosaurine dentaries were elongate; the dentary is between 56 and 60% of total length of the entire lower jaw in adult T. nepaeolicus and T. proriger,[43] about 55% in T. pembinensis,[25] and 62% in T. saskatchwanensis.[47] The dentary is robust, though not as strongly built as it is in Mosasaurus, Prognathodon, or Plesiotylosaurus.[86] The ventral margin of the dentary ranges from straight[22] to slightly concave.[27][24] A small dorsal ridge appears anterior to the first dentary tooth in mature individuals of T. proriger.[43]
The marginal dentition of most species is adapted for cutting large marine vertebrates,[48][87] while those in T. ivoensis and T. gaudryi appear more optimized for piercing or smashing prey,[88] and T. "borealis" in both piercing and cutting.[48] Marginal teeth are triangular with a slight recurve towards the back of the jaws so that the lingual (tongue-facing) side forms a U-shaped curve.[84] From top view, they are compressed at the lingual and labial (lip-facing) sides to form an oval-like shape.[41][47] Teeth of immature T. proriger are initially compressed, but become conical in adulthood.[34][42][89] Carinae (cutting edges) are finely serrated with small denticles[22][28][41] except in juvenile T. nepaeolicus.[41] In T. pembinensis, they are faint.[27] The teeth generally have both anterior and posterior carinae, but some anterior teeth may have only anterior carinae.[41][47] The placement of carinae, if paired, is not always equal; in at least T. proriger, T. ivoensis, T. gaudryi,[34] and T. pembinensis, they are positioned such that the surface area of the tooth's lingual side is greater than the labial side.[27] Both sides are always balanced in area in T. bernardi.[34] The enamel surface is lined with thin fine ridges called striations that run vertically from the tooth's base. The surface is also either smooth or faintly faceted, in which it is flattened into multiple sides to form a prism-like geometry.[88]
Bardet et al. (2006) classified Tylosaurus species into two morphological groups based on marginal dentition. The North American 'proriger group' includes T. proriger and T. nepaeolicus and is characterized by teeth with smooth or faint facets, less prominent carinae, and a vein-like network of primitive striations extending to near the tip.[90] The group was originally defined as having slender teeth,[90] but subsequent research has since recognized that slenderness is an ontogenetic trait in T. proriger with robust teeth appearing in adult forms.[89] Though not formally classified within a group, the marginal teeth of T. saskatchwanensis shares a comparable morphology with T. proriger.[47] The second is the Euro-American 'ivoensis group' and consists of T. ivoensis, T. gaudryi, and T. pembinensis. Their teeth are robust with prominent carinae with striations on the lingual and occasionally labial sides that do not reach the tooth's tip, and facets on the labial side.[90] The facets are gentle in T. pembinensis,[27] while in T. ivoensis they are slightly concave.[34] The latter feature is also known as fluting.[35] Marginal teeth in T. gaudryi are virtually indistinguishable from those in T. ivoensis.[34] T. iembeensis was not placed within either group; no further description is known of its teeth other than having striations and no facets.[90] The distinction of an 'ivoensis group' is contentious. Caldwell et al. (2008) argued that T. pembinensis cannot be compared with T. ivoensis as the former's teeth are not fluted, and that T. ivoensis is more allied with the distinctively fluted teeth of Taniwhasaurus.[35] Jiménez-Huidobro and Caldwell (2019) listed the absence of marginal fluting as a diagnostic (taxon-identifying) trait that differentiates Tylosaurus from Taniwhasaurus.[22]
The pterygoid teeth may have enabled ratchet feeding, in which the upper teeth held prey in place as the lower jaw slides back and forth via a streoptostylic jaw joint.[91] The bases of the pterygoid teeth are nearly circular, and each tooth is divided into front and back-facing sides of near-equal surface area via a pair of faint buccal and lingual carinae, except in T. gaudryi, in which the teeth are mediolaterally compressed.[34] Carinae are not serrated.[28][88] The anterior surface tends to be either smooth of faintly faceted, while the posterior surface is striated.
Postcranial skeleton
Both pectoral and pelvic girdles are unfused in adult Tylosaurus, in contrast to other taxa (e.g., Prognathodon overtoni).[92][page needed][93] Tylosaurus is also distinguished from other mosasaurs by a scapula that is significantly smaller than the coracoid and the absence of the anterior emargination of the coracoid, as well as the absence of a well-developed pubic tubercle.[92][page needed]
Tylosaurus limbs are primitive relative to other mosasaurs; their stylopodia (humeri and femora) lack both the complex muscle attachment sites and extreme proximodistal shortening present in other derived taxa. Both carpals and tarsals in tylosaurines are mostly unossified; while other mosasaurs typically have between three and five carpals and tarsals, adult Tylosaurus never possess more than two ossified carpal bones (usually only the ulnare, sometimes the ulnare and distal carpal four) and two ossified tarsal bones (usually only the astragalus, sometimes the astragalus and distal tarsal four).[92][page needed][94] Hyperphalangy (increased number of phalanges relative to the ancestral condition) is present in both fore- and hindlimbs, and the phalanges are spindle-shaped, unlike the short, blocky hourglass-shaped phalanges possessed by mosasaurines.[92][page needed] The pisiform appears to be either unossified or absent in tylosaurines. The functional consequences of differences in limb anatomy across different mosasaur clades is unclear.
Tylosaurus had 29 to 30 presacral vertebrae, 6 to 7 pygal vertebrae, and 89 to 112 caudal vertebrae; due to the lack of a bony articulation between the ilium and vertebral column, it is unclear whether any mosasaurs possessed true sacral vertebrae.[28][92][page needed] In all tylosaurines, like in plioplatecarpines, the chevrons articulate to the caudal vertebrae, and are not fused to them, as they are in mosasaurines. The tail possesses a distinct downward curve, suggesting the presence of a tail fluke.[57][95]
Soft tissue
Skin and coloration
Fossil evidence of the skin of Tylosaurus in the form of scales has been described since the late 1870s. These scales were small and diamond-shaped and were arranged in oblique rows, comparable to that found in modern rattlesnakes and other related reptiles. However, the scales in the mosasaur were much smaller in proportion to the whole body.[96][97] An individual measuring 5 meters (16 ft) in total body length had dermal scales measuring 3.3 by 2.5 millimeters (0.130 in × 0.098 in),[98] and in each square inch (2.54 cm) of the mosasaur's underside an average of ninety scales were present.[96] Each scale was keeled in a form resembling that of a shark's denticles.[97] This probably helped reduce underwater drag[97] and reflection on the skin.[99]
Microscopic analysis of scales in a T. nepaeolicus specimen by Lindgren et al. (2014) detected high traces of the pigment eumelanin indicative of a dark coloration similar to the leatherback sea turtle in life. This may have been complemented with countershading, present in many aquatic animals, though the distribution of dark and light pigments in the species remains unknown. A dark-colored form would have provided several evolutionary advantages. Dark coloration increases absorption of heat, allowing the animal to maintain elevated body temperatures in colder environments. Possession of this trait during infancy would in turn facilitate fast growth rates. Unreflective dark coloring and countershading would have provided the mosasaur with increased camouflage. Additional speculative functions includes increased tolerance to solar ultraviolet radiation, strengthened integuments. The study remarked that certain melanism-coding genes are pleiotropic for increased aggression.[99]
Respiratory system
AMNH FR 221 preserves parts of the cartilaginous respiratory system. This includes parts of the larynx (voice box), trachea (windpipe), and bronchi (lung airways). They were however only briefly described in the preserved position by Osborn (1899). The larynx is poorly preserved; a piece of its cartilage first appears below just between the pterygoid and quadrate and extends to behind the latter. This connects to the trachea, which appears below the atlas vertebra but is not preserved afterwards. The respiratory tract reappears below the fifth rib as a pair of bronchi and extends to just behind the as-preserved coracoids where preservation is lost.[57] The pairing is suggestive of two functional lungs like modern limbed lizards but unlike snakes.[59] Similar branching is also found in Platecarpus[59] and putatively Mosasaurus, the only two other derived mosasaurs with their respiratory systems documented.[100] The bifurcation point for the Tylosaurus specimen is anywhere between the first and sixth cervical vertebrae.[lower-alpha 4][57] In Platecarpus, the bronchi probably diverged below the sixth cervical into near-parallel pairs,[101] while in Mosasaurus the organ is dislocated.[100] A bifurcation point's position ahead of the forelimbs would be unlike terrestrial lizards, whose point is within the chest region, but similar to the short trachea and parallel bronchi of whales.[59]
Classification
Taxonomy

Tylosaurus is classified within the family Mosasauridae in the superfamily Mosasauroidea. The genus is the type genus of its own subfamily, the Tylosaurinae. Other members of this group include Taniwhasaurus and possibly Kaikaifilu, and the subfamily is defined by a shared feature of an elongated premaxillary rostrum that does not bear teeth.[22] The closest relatives of the Tylosaurinae include the Plioplatecarpinae and the primitive subfamilies Tethysaurinae and Yaguarasaurinae; together they are members of one of three possible major lineages of mosasaurs (the others being the Mosasaurinae subfamily and Halisauromorpha group) that was first recognized in 1993. This clade was named the Russellosaurina by Polcyn and Bell in 2005.[102][60][103]

Tylosaurus was among the earliest derived mosasaurs. The oldest fossil attributable to the genus is a premaxilla (TMM 40092-27) recovered from Middle Turonian deposits of the Arcadia Park Shale in Texas,[104] which is dated between 92.1 and 91.4 million years old based on correlations with index fossils.[105] Although formally referred to as Tylosaurinae incertae sedis during its first description, it was remarked to probably belong to T. kansasensis.[104] The specimen was later listed within said species in a 2020 reexamination.[43] A slightly younger specimen is of a skull (SGM-M1) of an indeterminate Tylosaurus species similar to T. kansasensis from the Ojinaga Formation in Chihuahua, Mexico,[85] dated around ~90 million years old at earliest.[105] A tooth from a Late Maastrichtian deposit in Nasiłów, Poland dating close to the Cretaceous–Paleogene boundary has been attributed to Hainosaurus sp.[106][107] With the incorporation of Hainosaurus as a synonym of Tylosaurus, this also makes the genus one of the last mosasaurs.[28][106] Currently, eight species of Tylosaurus are recognized by scientists as taxonomically valid. They are as follow: T. proriger, T. nepaeolicus, T. bernardi, T. gaudryi, T. ivoensis, T. iembeensis, T. pembinensis, and T. saskatchewanensis. The validity of two additional taxa remain unsettled; there is still debate whether T. kansasensis is synonymous with T. nepaeolicus, and T. "borealis" has yet to be described in a formal publication.[22][48]
Phylogeny and evolution
In 2020, Madzia and Cau performed a Bayesian analysis to better understand the evolutionary influence on early mosasaurs by contemporaneous pliosaurs and polycotylids by examining the rates of evolution in mosasauroids like Tylosaurus (specifically T. proriger, T. nepaeolicus, and T. bernardi). A Bayesian analysis in the study's implementation can approximate numerically defined rates of morphological evolution and ages of divergence of clades. The Tylosaurinae was approximated to have diverged from the Plioplatecarpinae around 93 million years ago; the divergence was characterized by the highest rate of evolution among all mosasaurid lineages. This trend of rapid evolution coincided with the extinction of the pliosaurs and a decrease in polycotylid diversity. The study noted converging traits between Tylosaurus, pliosaurs, and some polycotylids in tooth morphology and body size. However, there was no evidence to suggest that Tylosaurus or its precursors evolved as a result of out-competing and/or driving to extinction the pliosaurs and polycotylids. Instead, Madiza and Cau proposed that Tylosaurus may have taken advantage of the extinction of the pliosaurs and decline of polycotylids to quickly fill the ecological void they left behind. The Bayesian analysis also approximated a divergence of T. nepaeolicus from the rest of the genus around 86.88 million years ago and a divergence between T. proriger and T. bernardi around 83.16 million years ago. The analysis also generated a paraphyletic status of the genus, approximating Taniwhasaurus to have diverged from Tylosaurus around 84.65 million years ago, but this result is not consistent with previous phylogenetic analyses.[108]
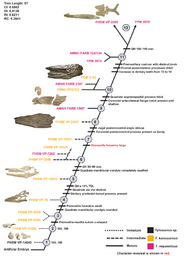
In the Western Interior Seaway, two species—T. nepaeolicus and T. proriger—may represent a chronospecies, in which they make up a single lineage that continuously evolves without branching in a process known as anagenesis. This is evident by how the two species do not stratigraphically overlap, are sister species, share minor and intermediate morphological differences such as a gradual change in the development of the quadrate bone, and lived in the same locations.[39][43] The means by which this lineage evolved has been hypothesized to be through one of two evolutionary mechanisms related to changes in ontogeny. First, Jiménez-Huidobro, Simões, and Caldwell proposed in 2016 that T. proriger evolved as a paedomorph of T. nepaeolicus, in which the descendant arose as a result of morphological changes through the retention of juvenile features of the ancestor in adulthood. This was based on the presence of a frontal crest and convex borders of the parietal bone of the skull shared in both juvenile T. nepaeolicus and all T. proriger but lost in adult T. nepaeolicus.[41][43] However, an ontogenetic study by Zietlow (2020) found that it was unclear whether this observation was a result of paedomorphosis, although this uncertainty may have been due that the sample size of mature T. nepaeolicus was too low to determine statistical significance. Second, the same study proposed an alternative hypothesis of peramorphosis, in which T. proriger evolved by developing traits found in mature T. nepaeolicus during immaturity. Based on results from a cladistical ontogram developed using data from 74 Tylosaurus specimens, the study identified a multitude of traits that were present in all T. proriger and mature T. nepaeolicus but absent in juvenile T. nepaeolicus: the skull size and depth are large, the length of the elongated rostrum exceeds 5% of the total skull length, the quadrate suprastapedial processes are thick, the overall quadrate shape converges, and the posteroventral process is fan-like.[43]
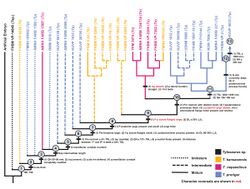
The following cladogram is modified from a phylogenetic analysis by Jiménez-Huidobro & Caldwell (2019) using Tylosaurus species with sufficiently known material to model accurate relationships; T. gaudryi, T. ivoensis, and T. iembeensis were excluded from the analysis due to extensive missing data (i.e., lack of material with scoreable phylogenetic characters).[22]
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Paleobiology
Function of edentulous rostrum
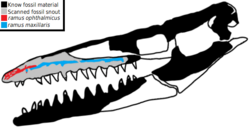
Russell (1967) first speculated the elongated rostrum was used to "stun" prey or defend against sharks, though provided no further explanation.[109] This combat hypothesis was further elaborated in Lingham-Soliar's (1992) review of Tylosaurus biomechanics. He observed that the internarial bar was unusually robust for a mosasaur, and sutured to the frontal with deep interdigitations such that there is a large interfacial shear area. This probably allowed for greater resistance to bending, shearing, and breaking forces though more effective shock absorption and stress transfer from a blunt force to the rostrum.[24] In T. bernardi, T. gaudryi,[24] and some individuals of other species as early-evolving as T. nepaeolicus,[34] the suture between the premaxilla and maxilla is also secured with a double buttress, which would allow the rostrum to lock in place upon a frontal force, with the elongation of the rostrum serving as a strut between the two bones. Lingham-Soliar reasoned that such biomechanical adaptations allowed Tylosaurus to use its rostrum as a battering ram.[24] This possible function was analogized to ramming behaviors seen in modern crocodiles during male-male combat, bottlenose dolphins to kill or deter sharks,[110] and orcas to attack prey.[111] Lingham-Soliar (1998) would later suggest possible evidence of a ram-attack in action in the skull of a subadult Mosasaurus whose braincase was fractured in life by a concentrated force, which he argued was rammed by a T. bernardi due to lack of damage elsewhere in the skull that would indicate other causes of injury.[110] Lingham-Soliar doubted that the elongated rostrum was involved in sexual selection, such as male-male combat, as previous surveys of Tylosaurus skulls found no evidence of sexual dimorphism in the length of the rostrum.[24] This was supported by Konishi et al.'s (2018) finding that the edentulous rostrum began to grow exceptionally early in development, which should not occur if the trait was sexual in nature but reasonable for general-purpose use like ramming.[111] Likewise, Zietlow (2020) found no evidence of sexual dimorphism relating to rostrum growth.[43]
An alternate hypothesis holds that the rostrum was used as a sensory tool. It was long known since Russell (1967) that the edentulous region of the rostrum contains numerous foramen that housed exits for the ophthalmic nerve, which transmits sensory information from the skin to the brain.[73] But that the elongated rostrum specifically functioned for sensation was first advanced by Álvarez–Herrera et al. (2020) based on CT scans of a Taniwhasaurus skull. They discovered a large network of internal canals of the ophthalmic nerve along the rostrum with up to 16 branching terminals inside the front end of the premaxilla, half of which exit into external foramina that dot the front and dorsal surfaces of the bone's edentulous region.[112] This pattern was later found in preliminary CT scans of a T. nepaeolicus skull by Paulina-Carabajal et al. (2023), which similarly observed profuse branching of the ophthalmic nerve inside the premaxilla exiting through many foramina on the dorsal tip of the rostrum.[113] The high concentration of sensory nerves within the edentulous portion of the rostrum suggests that it housed a mechanosensory or electrosensory organ. Similar adaptations were previously found in plesiosaurs and ichthyosaurs.[112] It was also suggested that the heavy branching inside the rostrum is associated with pathways for blood supply, and therefore a thermoregulation mechanism.[113] Paleontologist Gordon Bell remarked in 2002 that any injuries to the snout, such as if inflicted via ramming, would have been extremely painful due to the nerve endings.[114]
Growth and development
The ontogenetic history of Tylosaurus remains poorly researched despite the wealth of specimens including of juveniles.[43] Scientists who have attempted to study the topic found obtaining the samples needed for an ideal analysis to be difficult as many Tylosaurus specimens are scattered across the globe due to early cross-museum trading[42] and because museums are hesitant to provide many specimens for methods that require examination of internal bone anatomy which necessarily damages the fossil.[115] Several insights have been made nonetheless. Mosasaurs like Tylosaurus likely gave birth to live young.[43] While no evidence has been attributed to Tylosaurus specifically, live birth has been documented in other mosasauroids, including pregnant fossils of the fellow russellosaurine Plioplatecarpus and aigialosaur Carsosaurus, and fossils of newborn Clidastes in open water deposits.[116] The youngest known Tylosaurus individual (FHSM VP-14845) is a newborn (neonate) estimated to have measured 30 cm (12 in) in skull length and 2.23 m (7.3 ft) in total length, about 17.2% the size of Bunker and 24.8–27.9% of the estimated maximum lengths for T. nepaeolicus. The premaxilla of this specimen lacked an edentulous elongation of the rostrum; this trait already appears in juvenile specimens only slightly larger than FHSM VP-14845, including T. nepaeolicus and T. proriger specimens with estimated skull lengths of 40 cm (16 in) and 60 cm (24 in) respectively.[111]
A 2007 study by Pellegrini applied skeletochronology on the humeri of two juvenile Tylosaurus sp. specimens to measure the growth rates based on lines of arrested growth (LAGs). In poikilothermic sauropsids, bone growth tends to slow or freeze seasonally, either due to physiological cycles or in response to seasonal change in their environment. This creates a thin layer of avascular tissue called an annulus that, when deposited as a single line due to near-complete cease of growth, is called an LAG. The seasonal nature of LAG deposition can therefore proxy as annual growth rings, and measuring the amount of bone deposition during growing seasons via the distance of each LAG from the center of the bone can infer the animal's growth rate each year starting from the first year of life.[lower-alpha 5] The older humerus in Pellegrini's sample indicated the individual was 14 years old at death while the younger was aged 7 years. Both showed similar logarithmic growth curves that grew rapidly in the first few years but reduced to a steadier rate starting year 5 in the older specimen and year 6 in the younger. This may represent the ages when the individuals reached sexual maturity. The distance of first-year LAGs to the bone center in both specimens were longer than those in Platecarpus and Clidastes humeri, which suggests that Tylosaurus was larger at birth rather than being born at a similar size and then growing more rapidly. Like the other two mosasaurs, the overall growth rate was similar to modern monitor lizards but accelerated, which Pellegrini attributed to the need for increased metabolic rates to adapt to an aquatic environment.[115]
A 2018 study by Stewart and Mallon attempted to assess the degree of allometry in the growth of the T. proriger skull based on fifteen specimens. Their analysis of covariance test failed to reject isometry from five of the seven skull variables tested. The two definitively shown to be allometric were length of the edentulous rostrum (grew proportionally smaller with age) and height of the quadrate (grew proportionally larger with age). The isometric results were interpreted as most likely an artifact of low sample size, also known as "soft isometry", and the authors maintained that a larger sample size could reveal allometry in the five other variables. The negative allometry found for the edentulous rostrum was also unexpected, as it contradicted prior observations that juvenile Tylosaurus tended to have shorter rostrums than adults.[42]
A 2020 study by Zietlow used cladistic analysis to reconstruct ontograms (growth series charts) of the skull in T. proriger, T. nepaeolicus, and T. kansasensis based on 79 specimens. This approach tracks morphological changes with maturity although, it does not correlate them to particular ages. The resulting ontogram for T. proriger yielded 17 growth series. The most notable changes included the appearance of bony processes on the several bones of the skull, shrinking of the premaxillary foramina, ossification of the basioccipital and quadrate, reshaping of the premaxilla-maxilla suture to an M-shape, and increase in tooth size relative to the skull. T. nepaeolicus and T. kansasensis was coalesced into a single ontogram to test whether they were a single species. The result showed no evidence of separation; instead, the T. kansasensis specimens were recovered at the earlier stages and T. nepaeolicus at the later stages over a total of 12 growth series. The most notable changes were the enlargement of bony processes in several bones, enlargement of the premaxillary foramina, change in pterygoid shape, ossification of the quadrate and change from a hook-like to elongate shape, development of the M-shape in the premaxilla-maxilla suture, development of an extra dentary tooth, and increase in tooth size relative to the skull. Skull size in both ontograms as well as skull depth in T. proriger progressively increased through all growth stages. Statistical tests showed that both total skull length and quadrate height are reliably correlated with maturity in T. proriger and T. nepaeolicus/kansasensis. Neither ontogram showed evidence of sexual dimorphism, although Zietlow held that this is either an artifact of small sample size or because sex-differentiation does not manifest in the skull.[43]
Zietlow subsequently tested whether the identified ontogenetic traits remains supported under a new paradigm that assumes Tylosaurus developmental sequences should be consistent with close relatives, namely monitor lizards, and amniotes broadly. In a 2024 conference, she reported that ontogenetic change in quadrate shape is inconsistent with monitor lizard development, and that one previously proposed ontogenetic change, the migration of the parietal foramina, is biologically impossible under standard amniote embryogenesis. Zietlow also found that several traits she previously interpreted as part of growth stages in T. proriger and T. nepaeolicus in her 2020 study were actually interspecific variation, suggesting that T. kansasensis was not an immature form of T. nepaeolicus, that their associated ontogram also contained a new species, and that her T. proriger ontogram was an amalgamation of T. proriger and another new species.[117]
Metabolism
Nearly all squamates are characterized by their cold-blooded ectothermic metabolism, but mosasaurs like Tylosaurus are unique in that they were likely endothermic, or warm-blooded.[118] The only other known lizard with such a trait is the Argentine black and white tegu, though only partially.[119] Endothermy in Tylosaurus was demonstrated in a 2016 study by Harrell, Pérez‐Huerta, and Suarez by examining δ18O isotopes in Tylosaurus bones. δ18O levels can be used to calculate the internal body temperature of animals, and by comparing such calculated temperatures between coexisting cold-blooded and warm-blooded animals, the type of metabolism can be inferred. The study used the body temperatures of the cold-blooded fish Enchodus and sea turtle Toxochelys (correlated with ocean temperatures) and warm-blooded seabird Ichthyornis from the Mooreville Chalk as a proxy. Analyzing the isotope levels of eleven Tylosaurus specimens an average internal body temperature of 34.3 °C (93.7 °F) was calculated. This was much higher than the body temperature of Enchodus and Toxochelys (28.3 °C (82.9 °F) and 27.2 °C (81.0 °F) respectively) and similar to that of Ichthyornis (38.6 °C (101.5 °F)). Harrell, Pérez‐Huerta, and Suarez also calculated the body temperatures of Platecarpus and Clidastes with similar numbers, 36.3 °C (97.3 °F) and 33.1 °C (91.6 °F) respectively. The fact that the other mosasaurs were much smaller in size than Tylosaurus and yet maintained similar body temperatures made it unlikely that Tylosaurus's body temperature was the result of another metabolic type like gigantothermy.[lower-alpha 6] Endothermy would have provided several advantages to Tylosaurus such as increased stamina for foraging larger areas and pursuing prey, the ability to access colder waters, and better adaptation to withstand the gradual cooling of global temperatures during the Late Cretaceous.[118]
Mobility
Scientists previously interpreted Tylosaurus as an anguilliform swimmer that moved by undulating its entire body like a snake due to its close relationship with the animal. However, it is now understood that Tylosaurus actually used carangiform locomotion, meaning that the upper body was less flexible and movement was largely concentrated at the tail like in mackerels.[121] This is corroborated by a statistical reconstruction of the tail fin by Song and Lindgren (2025), which predicted an outline resembling those in carangiform sharks.[122] A BS thesis by Jesse Carpenter published in 2017 examined the vertebral mobility of T. proriger spinal columns and found that the dorsal vertebrae were relatively rigid but the cervical, pygal, and caudal vertebrae were more liberal in movement, indicating flexibility in the neck, hip, and tail regions. This contrasted with more derived mosasaurs like Plotosaurus, whose vertebral column was stiff up to the hip. Interestingly, an examination of a juvenile T. proriger found that its cervical and dorsal vertebrae were much stiffer than those in adult specimens. This may have been an evolutionary adaptation among young individuals; a more rigid tail-based locomotion is associated with faster speed, and this would allow vulnerable juveniles to better escape predators or catch prey. Older individuals would see their spine grow in flexibility as predator evasion becomes less important for survival.[121]
Tylosaurus likely specialized as an ambush predator. It was lightweight for a mosasaur of its size, having a morphological build designed to vastly reduce body mass and density. Its pectoral and pelvic girdles and paddles, which are associated with weight, are proportionally small. Its bones were highly cancellous and were likely filled with fat cells in life, which also increased buoyancy. It is unlikely that the latter trait was evolved in response to increasing body size as the similarly sized Mosasaurus hoffmannii lacked highly cancellous bone. These traits allowed Tylosaurus to be more conservative in its energy requirements, which is useful when traveling between ambush sites over large distances or through stealth. In addition, a reduced body density likely helped Tylosaurus to rapidly accelerate during an attack, assisted with the long and powerful tail of the mosasaur.[24]
A 1988 study by Judith Massare attempted to calculate the sustained swimming speed, the speed at which the animal moves without tiring, of Tylosaurus through a series of mathematical models incorporating hydrodynamic characteristics and estimations of locomotive efficiency and metabolic costs. Using two T. proriger specimens, one 6.46 meters (21.2 ft) long and the other 6.32 meters (20.7 ft), she calculated a consistent average maximum sustained swimming speed of 2.32 m/s (5.2 mph). However, when testing whether the models represented an accurate framework, they were found to exaggerated. This was primarily because the variables accounting for drag may have been underestimated; estimation of drag coefficients for an extinct species can be difficult as it requires a hypothetical reconstruction of the morphological dimensions of the animal. Massare predicted that the actual sustained swimming speed of Tylosaurus was somewhere near half the calculated speed.[123]
Feeding
One of the largest marine carnivores of its time, Tylosaurus was an apex predator that exploited the wide variety of species in the marine fauna of its ecosystem. Stomach contents are well documented in the genus, which includes other mosasaurs, plesiosaurs, turtles, birds, bony fish, and sharks.[100] Additional evidence from bite marks suggests the animal also preyed on giant squid[124] and ammonites.[125]
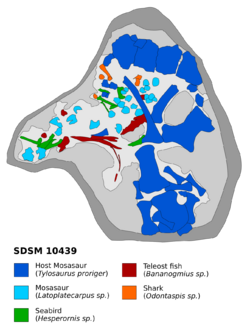
The enormous and varied appetite of Tylosaurus can be demonstrated in a 1987 find that identified fossils of a mosasaur measuring 2 meters (6.6 ft) or longer, the diving bird Hesperornis, a Bananogmius fish, and possibly a shark all within the stomach of a single T. proriger skeleton (SDSM 10439) recovered from the Pierre Shale of South Dakota.[lower-alpha 7][24][100][128] Other records of stomach contents include a sea turtle in a T. bernardi-like species,[lower-alpha 8][100] a 2.5 meters (8.2 ft) long Dolichorhynchops in another (8.8 metres (29 ft) long) T. proriger,[66] partially digested bones and scales of a Cimolichthys in a third T. proriger,[126] partially digested vertebrae of a Clidastes in a fourth T. proriger, remains of three Platecarpus individuals in a T. nepaeolicus,[65] and Plioplatecarpus bones in a T. saskatchewanensis.[45][129] Puncture marks on fossils of ammonites,[125] the carapace of a Protostega,[130] and the gladius of an Enchoteuthis have been attributed to Tylosaurus.[124]
Pasch and May (2001) reported bite marks from a dinosaur skeleton known as the Talkeetna Mountains Hadrosaur, which was found in marine strata of the Turonian-age Matanuska Formation in Alaska. The features of these marks were found to closely match that of the teeth of T. proriger. Because the fossil's locality was of marine deposits, the study reasoned that the dinosaur must have drifted offshore as a bloat-and-float carcass that was subsequently scavenged by the mosasaur. It was unlikely that the marks were a result of predation, as that would have led to a puncture, preventing the buildup of the bloating gases that allowed the corpse to drift out to sea in the first place.[131] Garvey (2020) criticized the lack of conclusive evidence to support this hypothesis and ruled out T. proriger as a possible culprit, given that the species did not appear until the Santonian and is exclusive to the Western Interior Seaway.[48] However, close relatives did maintain a presence nearby, evidenced by fragmentary fossils of an indeterminate tylosaurine from Turonian deposits in the Russian Chukotsky District.[132]
Social behavior
The behavior of Tylosaurus towards each other may have been mostly aggressive, evidenced by fossils with injuries inflicted by another of their own kind. Such remains were frequently reported by fossil hunters during the late 19th and early 20th centuries, but few examples reside as specimens in scientific collections. Many of these fossils consist of healed bite marks and wounds that are concentrated around or near the head region, implying that there were the result of non-lethal interaction, but the motives of such contact remain speculative. In 1993, Bruce M. Rothschild and Larry D. Martin noted that some modern lizards affectionately bite their mate's head during courtship, which can sometimes result in injuries. Alternatively, they also observed that some males lizards also employ head-biting as territorial behavior against rivals in a show of dominance by grappling the head to turn over the other on its back. It is possible that Tylosaurus behaved in similar ways.[65]
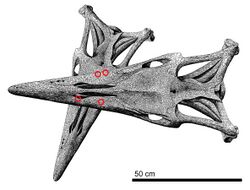
At least one fatal instance of intraspecific combat among Tylosaurus is documented in the holotype of the discussed species T. kansasensis (FHSM VP-2295), representing a 5 meters (16 ft) long animal, which possesses numerous injuries that indicate it was killed by a larger Tylosaurus. The skull roof and surrounding areas exhibit signs of trauma in the form of four massive gouges, and the dentary contains at least seven puncture wounds and gouges. These pathologies are characteristic of bite marks from a larger Tylosaurus that measured around 7 meters (23 ft) in length. The largest of the marks are about 4 centimeters (1.6 in) in length, matching the size of large mosasaur teeth, and they are positioned along two lines that converge close to 30°, matching the angle that each jaw converges towards in a mosasaur skull. In addition, FHSM VP-2295 suffered damage to its neck: the cervical vertebrae were found articulated at an unnatural angle of 40° relative to the long axis of the skull. The pattern of preservation makes it unlikely that the condition of the vertebrae was a result of disturbances by scavengers and instead indicates damage caused by a violently twisted neck during life. In a reconstructed scenario, the larger Tylosaurus would have first attacked at an angle slightly below the left side of the victim's head. This impact would cause the victim's skull to roll to the right side, allowing the aggressor to sink its teeth into the skull roof and right lower jaw, crushing the jaw and causing further breaks of nearby bones, such as the pterygoid, and the twisting of the jaw outwards, which would cause the quadrate to detach from its position and for the spinal cord to twist and sever at the skull's base, leading to a swift death.[65]
Paleopathology
Examining 12 North American Tylosaurus skeletons and one T. bernardi skeleton, Rothschild and Martin (2005) identified evidence of avascular necrosis in every individual. For aquatic animals, this condition is often a result of decompression illness, which is caused when bone-damaging nitrogen bubbles build up in inhaled air that is decompressed either by frequent deep-diving trips or by intervals of repetitive diving and short breathing. The studied mosasaurs likely gained avascular necrosis through such behaviors, and given its invariable presence in Tylosaurus it is likely that deep or repetitive diving was a general behavioral trait of the genus. The study observed that between 3-15% of vertebrae in the spinal column of North American Tylosaurus and 16% of vertebrae in T. bernardi were affected by avascular necrosis.[133] Carlsen (2017) posited that Tylosaurus gained avascular necrosis because it lacked the necessary adaptations for deep or repetitive diving, although noted that the genus had well-developed eardrums that could protect themselves from rapid changes in pressure.[134]
Unnatural fusion of some vertebrae in the tail has been reported in some Tylosaurus skeletons. A variation of these fusions may concentrate near the end of the tail to form a single mass of multiple fused vertebrae called a "club tail." Rothschild and Everhart (2015) surveyed 23 North American Tylosaurus skeletons and one T. bernardi skeleton and found that five of the North American skeletons exhibited fused tail vertebrae. The condition was not found in T. bernardi, but this does not rule out its presence due to the low sample size. Vertebral fusion occurs when the bones remodel themselves after damage from trauma or disease. However, the cause of such events can vary between individuals and/or remain hypothetical. One juvenile specimen with the club tail condition was found with a shark tooth embedded in the fusion, which confirms that at least some cases were caused by infections inflicted by predator attacks. The majority of vertebral fusion cases in Tylosaurus were caused by bone infections, but some cases may have alternatively been caused by any type of joint disease such as arthritis. However, evidence of joint disease was rare in Tylosaurus when compared to mosasaurs such as Plioplatecarpus and Clidastes.[135] Similar amassing of remodeled bone is also documented in bone fractures in other body parts. One T. kansasensis specimen possesses two fractured ribs that fully healed. Another T. proriger skull shows a fractured snout, probably caused by ramming into a hard object such as a rock. Presence of some healing indicates that the individual survived for some extended time before death, albeit under extreme pain.[114]
Paleoecology
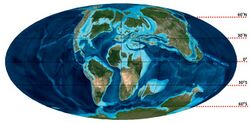
Throughout its ~20 million-year history, Tylosaurus was endemic to the North Atlantic Circle Basin, a geographic region comprising the North Atlantic and neighboring waters including the Western Interior Seaway in North America and Mediterranean Tethys in Europe above 30°N.[22] Much of the region was encompassed by a temperate ocean climate.[136][137] There are a few exceptions of occurrences in warmer low-latitude zones in scientific literature. This includes T. iembeensis from the Coniacian of Angola,[138] an isolated tooth from Maastrichtian deposits in the Democratic Republic of the Congo suggested as a possible 'Hainosaurus',[88] and a specimen near the Cretaceous–Paleogene boundary at the Ouled Abdoun Basin, Morocco.[139]
Habitat preference
Stratigraphic evidence in the WIS is inconsistent on whether Tylosaurus preferred a specific habitat. Russell (1967) suggested that Tylosaurus in the Niobrara Formation frequented open waters far offshore, potentially up to 400 km (250 mi) from the nearest coastline in the WIS.[140] This is supported by evidence in bone structure indicative of adaptation towards deeper waters. This includes osteoporosis, which naturally occurs in modern deep-diving or fast-swimming tetrapods due to bone growth in low-gravity environments both lifestyles create, and high fat deposition within bony microstructures, which together with osteoporosis would have allowed Tylosaurus to achieve neutral buoyancy in deep water.[141] However, later work found that Tylosaurus is found throughout both shallow and open water deposits of the Niobrara Formation.[141][142] On the other hand, Kiernan (2002) found that Tylosaurus comprised 57.7% of all mosasaurs found in Santonian to early Campanian deposits in Alabama that correspond to shallow continental shelves, and only 8.2% of mosasaurs in offshore deposits of similar ages. This implied that, at least in this region, Tylosaurus preferred nearshore waters.[142]
Biogeochemical analyses of fossils instead support Tylosaurus as a spatial generalist that inhabited a wide range of marine habitats with little specific preference. Analysis of rare earth element (REE) ratios, which preserve information about the habitat an animal was buried in, from T. proriger and T. sp. bones from Alabama and South Dakota by Harrell and Pérez-Huerta (2014) found a widely distributed range of ratios indicating habitation across waters with depths from 50 m (160 ft) to below >150 m (490 ft). This range was wider than all other mosasaurs examined, including Clidastes, Mosasaurus, Platecarpus, and Plioplatecarpus.[143] Carbon-13 isotope analysis (δ13C) of tooth enamel by Robbins (2010) and Polcyn et al. (2025) also support a generalized habitat distribution. Unlike REEs, δ13C is correlated with the habitat the animal fed or foraged in, with lower values associated with more offshore foraging habitats. Both studies together found a δ13C range of -8.2‰ to -13.3‰ for Campanian T. proriger-like teeth from Texas (T. sp. aff. T. proriger) and -7.6‰ to 12.6‰ (all but one within -7.6‰ to -10.6‰) for T. ivoensis from Sweden.[144][145] Isotope analyses of modern marine mammal teeth previously found that δ13C ratios between roughly -8‰ to -12.5‰ correspond to nearshore marine habitats and 11.5‰ to 15‰ to offshore marine habitats.[lower-alpha 9][146] Based on this, Polcyn et al. (2025) inferred that the T. proriger affiliates foraged in both nearshore and offshore marine habitats, while T. ivoensis foraged primarily nearshore with occasional offshore excursions. Both studies also examined a yet-undescribed Turonian specimen belonging to a 3–4 m (9.8–13.1 ft) long individual and found a δ13C ratio of -7.0‰. This corresponded to shallow waters analogous to modern kelp beds. Polcyn et al. (2025) suggested that early representatives of Tylosaurus were restricted to shallower habitats but expanded to more offshore zones over time as it evolved into larger sizes.[145]
See also
Notes
- ↑ Commenting on this problem, Everhart noted that Williston was already aware of the curvature of the mosasaur's spinal column and suggested that he would have insisted to draw it as straight in order to save space on the plate in which the reconstruction was published on.[56]
- ↑ Behind the fifth tooth in the holotype.[39]
- ↑ In one juvenile T. proriger specimen, it appears at the bottom of the vertical ramus instead.[41]
- ↑ Latter corresponds to the fifth rib in Osborn (1899).[57]
- ↑ However, LAGs can also be destroyed in life via bone remodeling. Pellegrini's original sample also included two tibia, but he found that too many LAGs were lost in both specimens to be reliable. He also found evidence that one or two LAGs were lost in the humeri specimens.[115]
- ↑ The 2018 MS thesis of Cyrus Green disputes the notion that Clidastes was an endotherm based on the skeletochronology of the genus, finding that its growth rates were too low to be endothermic and instead similar to ectotherms. The dissertation argued that the high body temperatures calculated by Harrell et al. (2016) were a result of gigantothermy. However, only four specimens were studied, and Clidastes is considered a basal mosasaur.[120]
- ↑ Identification of the mosasaur and shark vary. Scientists have identified the mosasaur as either a Platecarpus,[65] Clidastes,[126] or Latoplatecarpus.[100] The shark is either interpreted as a Cretalamna,[126] a sand shark,[127] or of uncertain identity.[100]
- ↑ Usually identified as Hainosaurus sp.;[100] Lingham-Soliar (1992) identifies the species as T. bernardi.[24]
- ↑ The study found an overlap from 11.5‰ to 12.5‰.[146]
References
- ↑ Adrienne Mayor. Fossil Legends of the First Americans. Princeton: Princeton University Press. p. 69–72, 209–211, 275–276. ISBN 978-0-691-11345-6.
- ↑ "September 10, 1804". https://lewisandclarkjournals.unl.edu/item/lc.jrn.1804-09-10.
- ↑ Ellis 2003, p. 216.
- ↑ Robert W. Meredith; James E. Martin; Paul N. Wegleitner (2007). "The largest mosasaur (Squamata: Mosasauridae) from the Missouri River area (Late Cretaceous; Pierre Shale Group) of South Dakota and its relationship to Lewis and Clark". Geological Society of America Special Papers 427: 209–214. doi:10.1130/2007.2427(15). ISBN 978-0-8137-2427-0. https://www.montclair.edu/profilepages/media/5008/user/Meredith,_Martin_2007_The_largest_mosasaur_(Squamata_Mosasauridae)_from_the_Missouri_River_area_(Late_Cretaceous_Pierre_Shale_Group)_of_South_Dakota_and_its_relationship_to_Lewis.pdf.
- ↑ 5.0 5.1 5.2 5.3 Everhart 2017, p. 206.
- ↑ Ellis 2003, p. 207.
- ↑ 7.0 7.1 Edward Drinker Cope (1869). "[Remarks on Holops brevispinus, Ornithotarsus immanis, and Macrosaurus proriger"]. Proceedings of the Academy of Natural Sciences of Philadelphia 1869: 123. ISSN 0097-3157. https://www.biodiversitylibrary.org/item/30466#page/149.
- ↑ Mike Everhart (2005). "Macrosaurus proriger". http://oceansofkansas.com/Cope1869b.html.
- ↑ 9.0 9.1 9.2 Edward Drinker Cope (1870). "Synopsis of the extinct Batrachia, Reptilia and Aves of North America". Transactions of the American Philosophical Society 14 (1): 202–205. https://www.biodiversitylibrary.org/item/123170#page/258/mode/1up.
- ↑ 10.0 10.1 Everhart 2017, p. 207.
- ↑ 11.0 11.1 Ellis 2003, p. 208.
- ↑ "Sebastes proriger, Redstripe rockfish". https://www.fishbase.se/summary/3991.
- ↑ Othniel Charles Marsh (1872). "On the structure of the skull and limbs in mosasaurid reptiles, with descriptions of new genera and species". American Journal of Science. Series 3 3 (18): 448–464. https://archive.org/details/mobot31753002152814/page/448.
- ↑ Mike Everhart (2000). "Tylosaurus proriger". http://oceansofkansas.com/page10.html.
- ↑ 15.0 15.1 Russell 1967, p. 173.
- ↑ Othniel Charles Marsh (1872). "Note on Rhinosaurus". American Journal of Science 4 (20): 147. https://books.google.com/books?id=6vcQAAAAIAAJ&pg=PA147.
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 Michael J. Everhart (2002). "New Data on Cranial Measurements and Body Length of the Mosasaur, Tylosaurus nepaeolicus (Squamata; Mosasauridae), from the Niobrara Formation of Western Kansas". Transactions of the Kansas Academy of Science 105 (1–2): 33–43. doi:10.1660/0022-8443(2002)105[0033:NDOCMA2.0.CO;2]. https://www.researchgate.net/publication/232668108.
- ↑ Joseph Leidy (1873). Contributions to the extinct vertebrate fauna of the western interior territories: Report of the United States Geological Survey of the Territories. U.S. Government Printing Office. https://www.biodiversitylibrary.org/item/125566#page/285/mode/1up.
- ↑ Edward Drinker Cope (1874). "Review of the Vertebrata of the Cretaceous period found west of the Mississippi River". Bulletin of the U.S. Geological and Geographical Survey of the Territories 1 (2): 3–48. https://www.biodiversitylibrary.org/item/99293#page/57/mode/1up.
- ↑ John C. Merriam (1894). "Über die Pythonomorphen der Kansas-Kreide" (in German). Palaeontographica 41: 1–39. https://www.biodiversitylibrary.org/item/109783#page/11.
- ↑ Russell 1967, p. 175–176.
- ↑ 22.00 22.01 22.02 22.03 22.04 22.05 22.06 22.07 22.08 22.09 22.10 22.11 22.12 22.13 22.14 22.15 Cite error: Invalid
<ref>tag; no text was provided for refs namedJimenezCaldwell - ↑ 23.0 23.1 Louis Dollo (1885). "Le Hainosaure" (in French). Revue des Questions Scientifiques 18: 285–289. ISSN 0035-2160. https://www.biodiversitylibrary.org/item/207708#page/291.
- ↑ 24.00 24.01 24.02 24.03 24.04 24.05 24.06 24.07 24.08 24.09 24.10 24.11 24.12 24.13 24.14 24.15 24.16 Lingham-Soliar, T. (1992). "The Tylosaurine Mosasaurs (Reptilia, Mosasauridae) from the Upper Cretaceous of Europe and Africa". Bulletin de l'Institut Royal des Sciences Naturelles de Belgique, Sciences de la Terre 62: 171–194. https://core.ac.uk/reader/35118494. Retrieved December 24, 2020.
- ↑ 25.0 25.1 Nicholls, E.L. (1988). "The first record of the mosasaur Hainosaurus (Reptilia: Lacertilia) from North America". Canadian Journal of Earth Sciences 25 (10): 1564–1570. doi:10.1139/e88-149. Bibcode: 1988CaJES..25.1564N.
- ↑ 26.0 26.1 26.2 26.3 Lindgren, J. (2005). "The first record of Hainosaurus (Reptilia: Mosasauridae) from Sweden". Journal of Paleontology 79 (6): 1157–1165. doi:10.1666/0022-3360(2005)079[1157:tfrohr2.0.co;2].
- ↑ 27.00 27.01 27.02 27.03 27.04 27.05 27.06 27.07 27.08 27.09 27.10 27.11 27.12 27.13 27.14 27.15 27.16 Bullard, T.S.; Caldwell, M.W. (2010). "Redescription and rediagnosis of the tylosaurine mosasaur Hainosaurus pembinensis Nicholls, 1988, as Tylosaurus pembinensis (Nicholls, 1988)". Journal of Vertebrate Paleontology 30 (2): 416–426. doi:10.1080/02724631003621870. Bibcode: 2010JVPal..30..416B.
- ↑ 28.00 28.01 28.02 28.03 28.04 28.05 28.06 28.07 28.08 28.09 28.10 28.11 28.12 28.13 28.14 28.15 Jiménez-Huidobro, P.; Caldwell, M.W. (2016). "Reassessment and reassignment of the early Maastrichtian mosasaur Hainosaurus bernardi Dollo, 1885, to Tylosaurus Marsh, 1872". Journal of Vertebrate Paleontology 36 (3). doi:10.1080/02724634.2016.1096275. Bibcode: 2016JVPal..36E6275J.
- ↑ Jahn J. Hornung; Mike Reich; Udo Frerichs (2018). "A mosasaur fauna (Squamata: Mosasauridae) from the Campanian (Upper Cretaceous) of Hannover, northern Germany". Alcheringa: An Australasian Journal of Palaeontology 42 (4): 543–559. doi:10.1080/03115518.2018.1434899. Bibcode: 2018Alch...42..543H.
- ↑ Sven Sachs; Jahn J. Hornung; Udo Scheer (2018). "Mosasaurid and plesiosaurian remains from marginal facies of the lower Campanian (Upper Cretaceous) Bottrop and Vaals formations of western Germany". Cretaceous Research 87 (2018): 358–367. doi:10.1016/j.cretres.2017.05.026. Bibcode: 2018CrRes..87..358S.
- ↑ 31.0 31.1 Armand Thevenin (1896). "Mosasauriens de la Craie Grise de Vaux Eclusier près Peronne (Somme)". Bulletin de la Société Géologique de France 3 (24): 900–916. ISSN 0037-9409. https://www.biodiversitylibrary.org/item/100438#page/916.
- ↑ Per O. Persson (1963). "Studies on Mesozoic marine reptile faunas with particular regard to the Plesiosauria". Institutes of Mineralogy, Palaeontology, and Quaternary Geology, University of Lund 118: 1–57. ISSN 0458-4732.
- ↑ Russell 1967, p. 135–136.
- ↑ 34.00 34.01 34.02 34.03 34.04 34.05 34.06 34.07 34.08 34.09 34.10 Lindgren, J.; Siverson, M. (2002). "Tylosaurus ivoensis: a giant mosasaur from the early Campanian of Sweden". Earth and Environmental Science Transactions of the Royal Society of Edinburgh 93 (1): 73–93. doi:10.1017/s026359330000033x.
- ↑ 35.0 35.1 35.2 35.3 Caldwell, M.W.; Konishi, T.; Obata, I.; Muramoto, K. (2008). "A new species of Taniwhasaurus (Mosasauridae, Tylosaurinae) from the upper Santonian-lower Campanian (Upper Cretaceous) of Hokkaido, Japan". Journal of Vertebrate Paleontology 28 (2): 339–348. doi:10.1671/0272-4634(2008)28[339:ansotm2.0.co;2].
- ↑ Miguel Telles Antunes (1964). "Estratigrafía; Repteis" (in pt). O Neocretácico e o Cenozóico do litoral de Angola. Lisbon: Junta de Investigações do Ultramar. p. 165. OCLC 30868727.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedJacobsetal - ↑ Octávio Mateus; Michael J. Polcyn; Louis L. Jacobs; Ricardo Araújo; Anne S. Schulp; João Marinheiro; Bruno Pereira; Diana Vineyard (2012). "Cretaceous amniotes from Angola: dinosaurs, pterosaurs, mosasaurs, plesiosaurs, and turtles". V Jornadas Internacionales sobre Paleontología de Dinosaurios y su Entorno: 71–105. https://docentes.fct.unl.pt/omateus/publications/cretaceous-amniotes-angola-dinosaurs-pterosaurs-mosasaurs-plesiosaurs-and-turtl.
- ↑ 39.0 39.1 39.2 Everhart, M.J. (2005). "Tylosaurus kansasensis, a new species of tylosaurine (Squamata, Mosasauridae) from the Niobrara Chalk of western Kansas, USA". Netherlands Journal of Geosciences 84 (3): 231–240. doi:10.1017/S0016774600021016. Bibcode: 2005NJGeo..84..231E. https://www.researchgate.net/publication/27711025.
- ↑ 40.0 40.1 Everhart 2017, p. 208.
- ↑ 41.00 41.01 41.02 41.03 41.04 41.05 41.06 41.07 41.08 41.09 41.10 41.11 Jiménez-Huidobro, P.; Simões, T.R.; Caldwell, M.W. (2016). "Re-characterization of Tylosaurus nepaeolicus (Cope, 1874) and Tylosaurus kansasensis Everhart, 2005: Ontogeny or sympatry?". Cretaceous Research 65: 68–81. doi:10.1016/j.cretres.2016.04.008. Bibcode: 2016CrRes..65...68J.
- ↑ 42.0 42.1 42.2 42.3 Stewart, R.F.; Mallon, J. (2018). "Allometric growth in the skull of Tylosaurus proriger (Squamata: Mosasauridae) and its taxonomic implications". Vertebrate Anatomy Morphology Palaeontology 66: 75–90. doi:10.18435/vamp29339.
- ↑ 43.00 43.01 43.02 43.03 43.04 43.05 43.06 43.07 43.08 43.09 43.10 43.11 43.12 43.13 43.14 43.15 43.16 43.17 43.18 43.19 Zietlow, A.R. (2020). "Craniofacial ontogeny in Tylosaurinae". PeerJ 8. doi:10.7717/peerj.10145. PMID 33150074.
- ↑ Timon S. Bullard (2006). Anatomy and systematics of North American tylosaurine mosasaurs (MS). University of Alberta. doi:10.7939/r3-c3qm-y670.
- ↑ 45.0 45.1 45.2 "Omācīw". https://royalsaskmuseum.ca/rsm/visit/exhibits/travelling-exhibits/omcw.
- ↑ "Royal Saskatchewan Museum". Regina Leader-Post. October 14, 2010. https://www.pressreader.com/canada/regina-leader-post/20101014/281616711725503.
- ↑ 47.00 47.01 47.02 47.03 47.04 47.05 47.06 47.07 47.08 47.09 47.10 47.11 47.12 47.13 Jiménez-Huidobro, P.; Caldwell, M.W.; Paparella, I.; Bullard, T.S. (2018). "A new species of tylosaurine mosasaur from the upper Campanian Bearpaw Formation of Saskatchewan, Canada". Journal of Systematic Palaeontology 17 (10): 1–16. doi:10.1080/14772019.2018.1471744.
- ↑ 48.0 48.1 48.2 48.3 48.4 48.5 Garvey, S.T. (2020). A new high-latitude Tylosaurus (Squamata, Mosasauridae) from Canada with unique dentition (MS). University of Alberta. Archived from the original on 2020-07-23.
- ↑ 49.0 49.1 49.2 Edward Drinker Cope (1872). "On the geology and paleontology of the Cretaceous strata of Kansas". Annual Report of the United States Geological and Geographical Survey of the Territories 5: 318–349. https://www.biodiversitylibrary.org/item/111849#page/350.
- ↑ Jane P. Davidson (2015). "Misunderstood Marine Reptiles: Late Nineteenth-Century Artistic Reconstructions of Prehistoric Marine Life". Transactions of the Kansas Academy of Science 118 (1 & 2): 53–67. doi:10.1660/062.118.0107.
- ↑ 51.0 51.1 Jane P. Davidson; Michael J. Everhart (2014). "Fictionalized facts; "The Young Fossil Hunters" by Charles H. Sternberg". Transactions of the Kansas Academy of Science 117 (1–2): 41–54. doi:10.1660/062.117.0106. https://www.researchgate.net/publication/269579644.
- ↑ Everhart 2017, p. 226.
- ↑ Everhart 2017, p. 316–317.
- ↑ Everhart 2017, p. 216.
- ↑ 55.0 55.1 Samuel W. Williston (1898). "Mosasaurs". University Geological Survey of Kansas 4 (5): 81–221. http://www.kgs.ku.edu/Publications/Bulletins/Vol4/index.html.
- ↑ 56.0 56.1 Everhart 2017, p. 228.
- ↑ 57.0 57.1 57.2 57.3 57.4 Osborn, H.F. (1899). "A complete mosasaur skeleton, osseous and cartilaginous". Memoirs of the American Museum of Natural History 1 (4): 167–188. doi:10.1126/science.10.260.919. PMID 17837338. Bibcode: 1899Sci....10..919O. http://digitallibrary.amnh.org/handle/2246/5737.
- ↑ Mike Everhart (2008). "The Tylosaurus Collection". http://oceansofkansas.com/mus-tylo.html.
- ↑ 59.0 59.1 59.2 59.3 Lindgren, J.; Caldwell, M.W.; Konishi, T.; Chiappe, L.M. (2010). "Convergent Evolution in Aquatic Tetrapods: Insights from an Exceptional Fossil Mosasaur". PLOS ONE 5 (8). doi:10.1371/journal.pone.0011998. PMID 20711249. Bibcode: 2010PLoSO...511998L.
- ↑ 60.0 60.1 Caldwell, M.W. (2012). "A challenge to categories: "What, if anything, is a mosasaur?"". Bulletin de la Société Géologique de France 183 (1): 17–34. doi:10.2113/gssgfbull.183.1.7.
- ↑ Michael Everhart; John W. M. Jagt; Eric W. A. Mulder; Anne S. Schulp (2016). "Mosasaurs—how large did they really get?". 5th Triennial Mosasaur Meeting—A Global Perspective on Mesozoic Marine Amniotes. pp. 8–10. https://www.researchgate.net/publication/303518964.
- ↑ Everhart 2017, p. 214.
- ↑ Everhart 2017, p. 192.
- ↑ Everhart, M.J. (2005). "Rapid evolution, diversification and distribution of mosasaurs (Reptilia; Squamata) prior to the KT Boundary". Tate 2005 11th Annual Symposium in Paleontology and Geology. https://www.researchgate.net/publication/297758947.
- ↑ 65.0 65.1 65.2 65.3 65.4 Everhart, M.J. (2008). "A bitten skull of Tylosaurus kansasensis (Squamata: Mosasauridae) and a review of mosasaur-on-mosasaur pathology in the fossil record". Transactions of the Kansas Academy of Science 111 (3/4): 251–262. doi:10.1660/0022-8443-111.3.251. https://www.researchgate.net/publication/232686334.
- ↑ 66.0 66.1 Everhart, M.J. (2004). "Plesiosaurs as the food of mosasaurs; new data on the stomach contents of a Tylosaurus proriger (Squamata; Mosasauridae) from the Niobrara Formation of western Kansas". The Mosasaur 7: 41–46. https://www.researchgate.net/publication/284484258.
- ↑ Everhart 2017, p. 213.
- ↑ Everhart 2017, p. 215.
- ↑ "Largest mosasaur on display" (in en-GB). https://www.guinnessworldrecords.com/world-records/118041-largest-mosasaur-on-display.
- ↑ CBC News (2008-08-27). "Manitoba dig uncovers 80-million-year-old sea creature". CBC (Manitoba). https://www.cbc.ca/news/canada/manitoba/manitoba-dig-uncovers-80-million-year-old-sea-creature-1.711525.
- ↑ Christiansen, P.; Bonde, M. (2002). "A new species of gigantic mosasaur from the Late Cretaceous of Israel". Journal of Vertebrate Paleontology 22 (3): 629–644. doi:10.1671/0272-4634(2002)022[0629:ANSOGM2.0.CO;2].
- ↑ Russell 1967, p. 14.
- ↑ 73.0 73.1 Russell 1967, p. 16.
- ↑ Russell 1967, p. 17.
- ↑ 75.0 75.1 75.2 Russell 1967, p. 18.
- ↑ Camp, C.L. (1942). "California mosasaurs". Memoirs of the University of California 13: 1–68. https://catalog.hathitrust.org/Record/102284839.
- ↑ Schulp, A.S.; Mulder, E.W.A.; Schwenk, K. (2005). "Did mosasaurs have forked tongues?". Netherlands Journal of Geosciences 84 (3): 359–371. doi:10.1017/S0016774600021144. Bibcode: 2005NJGeo..84..359S.
- ↑ 78.0 78.1 Russell 1967, p. 25.
- ↑ Russell 1967, p. 26.
- ↑ Russell 1967, p. 59.
- ↑ 81.0 81.1 Fernández, M.S.; Talevi, M. (2015). "An halisaurine (Squamata: Mosasauridae) from the Late Cretaceous of Patagonia, with a preserved tympanic disc: Insights into the mosasaur middle ear". Comptes Rendus Palevol 14 (6–7): 483–493. doi:10.1016/j.crpv.2015.05.005. Bibcode: 2015CRPal..14..483F.
- ↑ 82.0 82.1 82.2 82.3 82.4 82.5 Palci, A.; Konishi, T.; Caldwell, M.W. (2020). "A comprehensive review of the morphological diversity of the quadrate bone in mosasauroids (Squamata: Mosasauroidea), with comments on the homology of the infrastapedial process". Journal of Vertebrate Paleontology 40 (6). doi:10.1080/02724634.2021.1879101. Bibcode: 2020JVPal..40E9101P.
- ↑ Russell 1967, p. 61.
- ↑ 84.0 84.1 Russell 1967, p. 57.
- ↑ 85.0 85.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedFlores - ↑ Russell 1967, p. 50.
- ↑ Ross, M.R. (2009). "Charting the Late Cretaceous seas: mosasaur richness and morphological diversification". Journal of Vertebrate Paleontology 29 (2): 409–416. doi:10.1671/039.029.0212. Bibcode: 2009JVPal..29..409R. https://digitalcommons.liberty.edu/bio_chem_fac_pubs/115.
- ↑ 88.0 88.1 88.2 88.3 Cite error: Invalid
<ref>tag; no text was provided for refs namedHornungandReich - ↑ 89.0 89.1 Konishi, T.; Caldwell, M. (2007). "Ecological and evolutionary implications of ontogenetic changes in the marginal dentition of Tylosaurus proriger (Squamata: Mosasauridae)". Journal of Vertebrate Paleontology 27 (3, Supplement): 101A. https://vertpaleo.org/wp-content/uploads/2021/03/JVPabstracts2007.pdf.
- ↑ 90.0 90.1 90.2 90.3 Bardet, N.; Pereda Suberbiola, X.; COrral, J.C. (2006). "A Tylosaurine Mosasauridae (Squamata) from the Late Cretaceous of the Basque-Cantabrian Region". Estudios Geológicos 62 (1): 213–218. doi:10.3989/egeol.0662121. https://estudiosgeol.revistas.csic.es/index.php/estudiosgeol/article/view/21.
- ↑ Lingham-Soliar, T. (1995). "Anatomy and functional morphology of the largest marine reptile known, Mosasaurus hoffmanni (Mosasauridae, Reptilia) from the Upper Cretaceous, Upper Maastrichtian of The Netherlands". Philosophical Transactions of the Royal Society B 347 (1320): 155–180. doi:10.1098/rstb.1995.0019. Bibcode: 1995RSPTB.347..155L. https://royalsocietypublishing.org/doi/pdf/10.1098/rstb.1995.0019.
- ↑ 92.0 92.1 92.2 92.3 92.4 Russell 1967.
- ↑ Konishi, Takuya; Brinkman, Donald; Massare, Judy A.; Caldwell, Michael W. (September 2011). "New exceptional specimens of Prognathodon overtoni (Squamata, Mosasauridae) from the upper Campanian of Alberta, Canada, and the systematics and ecology of the genus" (in en). Journal of Vertebrate Paleontology 31 (5): 1026–1046. doi:10.1080/02724634.2011.601714. ISSN 0272-4634. Bibcode: 2011JVPal..31.1026K. http://www.tandfonline.com/doi/abs/10.1080/02724634.2011.601714.
- ↑ Caldwell, Michael W. (April 1996). "Ontogeny and phylogeny of the mesopodial skeleton in mosasauroid reptiles" (in en). Zoological Journal of the Linnean Society 116 (4): 407–436. doi:10.1111/j.1096-3642.1996.tb00131.x. https://academic.oup.com/zoolinnean/article-lookup/doi/10.1111/j.1096-3642.1996.tb00131.x.
- ↑ Lindgren, Johan; Kaddumi, Hani F.; Polcyn, Michael J. (2013-09-10). "Soft tissue preservation in a fossil marine lizard with a bilobed tail fin" (in en). Nature Communications 4 (1): 2423. doi:10.1038/ncomms3423. ISSN 2041-1723. PMID 24022259. Bibcode: 2013NatCo...4.2423L. https://www.nature.com/articles/ncomms3423.
- ↑ 96.0 96.1 Snow, F.H. (1878). "On the dermal covering of a mosasauroid reptile". Transactions of the Kansas Academy of Science 6: 54–58. http://oceansofkansas.com/Snow1878.html.
- ↑ 97.0 97.1 97.2 Caldwell, M.W.; Sasso, C.D. (2004). "Soft-tissue preservation in a 95 million year old marine lizard: form, function, and aquatic adaptation". Journal of Vertebrate Paleontology 24 (4): 980–985. doi:10.1671/0272-4634(2004)024[0980:spiamy2.0.co;2]. https://www.researchgate.net/publication/231345547.
- ↑ Lindgren, J.; Everhart, M.J.; Caldwell, M.W. (2011). "Three-Dimensionally Preserved Integument Reveals Hydrodynamic Adaptations in the Extinct Marine Lizard Ectenosaurus (Reptilia, Mosasauridae)". PLOS ONE 6 (11). doi:10.1371/journal.pone.0027343. PMID 22110629. Bibcode: 2011PLoSO...627343L.
- ↑ 99.0 99.1 Lindgren, J.; Sjövall, P.; Carney, R.M.; Uvdal, P.; Gren, J.A.; Dyke, G.; Schultz, B.P.; Shawkey, M. D. et al. (2014). "Skin pigmentation provides evidence of convergent melanism in extinct marine reptiles". Nature 506 (7489): 484–488. doi:10.1038/nature12899. PMID 24402224. Bibcode: 2014Natur.506..484L.
- ↑ 100.0 100.1 100.2 100.3 100.4 100.5 100.6 100.7 Konishi, T.; Newbrey, M.; Caldwell, M. (2014). "A small, exquisitely preserved specimen of Mosasaurus missouriensis (Squamata, Mosasauridae) from the upper Campanian of the Bearpaw Formation, western Canada, and the first stomach contents for the genus". Journal of Vertebrate Paleontology 34 (4): 802–819. doi:10.1080/02724634.2014.838573. Bibcode: 2014JVPal..34..802K.
- ↑ Konishi, T.; Lindgren, J.; Caldwell, M.W.; Chiappe, L. (2012). "Platecarpus tympaniticus (Squamata, Mosasauridae): osteology of an exceptionally preserved specimen and its insights into the acquisition of a streamlined body shape in mosasaurs". Journal of Vertebrate Paleontology 6 (12): 1313–1327. doi:10.1080/02724634.2012.699811. Bibcode: 2012JVPal..32.1313K.
- ↑ Bell, G.L. Jr.; Polcyn, M.J. (2005). "Dallasaurus turneri, a new primitive mosasauroid from the Middle Turonian of Texas and comments on the phylogeny of the Mosasauridae (Squamata)". Netherlands Journal of Geoscience 84 (3): 177–194. doi:10.1017/S0016774600020965. Bibcode: 2005NJGeo..84..177B. https://www.researchgate.net/publication/27711031.
- ↑ Madzia, D.; Cau, A. (2017). "Inferring 'weak spots' in phylogenetic trees: application to mosasauroid nomenclature". PeerJ 5. doi:10.7717/peerj.3782. PMID 28929018. Bibcode: 2017PeerJ...5e3782M.
- ↑ 104.0 104.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedPolcynetal2008 - ↑ 105.0 105.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedGeologicTime - ↑ 106.0 106.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedJagtetal2005 - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedKeutgenetal - ↑ Madzia, D.; Cau, A. (2020). "Estimating the evolutionary rates in mosasauroids and plesiosaurs: discussion of niche occupation in Late Cretaceous seas". PeerJ 8. doi:10.7717/peerj.8941. PMID 32322442.
- ↑ Russell 1967, p. 69.
- ↑ 110.0 110.1 Lingham-Soliar, T. (1998). "Unusual death of a Cretaceous giant". Lethaia 31 (4): 308–310. doi:10.1111/j.1502-3931.1998.tb00520.x. Bibcode: 1998Letha..31..308L. http://doc.rero.ch/record/16192/files/PAL_E3401.pdf.
- ↑ 111.0 111.1 111.2 Konishi, T.; Jiménez-Huidobro, P.; Caldwell, M.W. (2018). "The Smallest-Known Neonate Individual of Tylosaurus (Mosasauridae, Tylosaurinae) Sheds New Light on the Tylosaurine Rostrum and Heterochrony". Journal of Vertebrate Paleontology 38 (5): 1–11. doi:10.1080/02724634.2018.1510835. Bibcode: 2018JVPal..38E0835K.
- ↑ 112.0 112.1 Álvarez–Herrera, G.; Agnolin, F.; Novas, F. (2020-04-24). "A rostral neurovascular system in the mosasaur Taniwhasaurus antarcticus" (in en). The Science of Nature 107 (3): 19. doi:10.1007/s00114-020-01677-y. ISSN 1432-1904. PMID 32333118. Bibcode: 2020SciNa.107...19A.
- ↑ 113.0 113.1 Paulina-Carabajal, A.; Jimenez-Huidobro, P.; Caldwell, M.W. (2023). "New Information on the neurovascularization of the snout In mosasaurs: the case of Tylosaurus nepaeolicus (Squamata:Mosasauroidea), using CT scans". Reunión de Comunicaciones de la Asociación Paleontológica Argentina, General Roca, Río Negro, 22 al 24 de noviembre de 2023, Programa y Libro De Resúmenes. p. 168. https://www.researchgate.net/publication/377844859.
- ↑ 114.0 114.1 Everhart, M.J. (2001). "Mosasaur Pathology". http://oceansofkansas.com/mosapath.html.
- ↑ 115.0 115.1 115.2 Pellegrini, R. (2007). "Skeletochronology of the limb elements of mosasaurs (Squamata; Mosasauridae)". Transactions of the Kansas Academy of Science 110 (1/2): 83–99. doi:10.1660/0022-8443(2007)110[83:SOTLEO2.0.CO;2]. https://www.researchgate.net/publication/232691128.
- ↑ Field, D.J.; LeBlanc, A.; Gau, A.; Behlke, A.D. (2015). "Pelagic neonatal fossils support viviparity and precocial life history of Cretaceous mosasaurs". Palaeontology 58 (3): 401–407. doi:10.1111/pala.12165. Bibcode: 2015Palgy..58..401F.
- ↑ Zietlow, A.R. (2024). "Tylosaurine diversity informed by morphological variation in extant Varanus". 7th Triennial Mosasaur Meeting – A global perspective on Mesozoic marine amniotes. pp. 205–208. https://www.nhmmaastricht.nl/wp-content/uploads/2024/10/NHM_Abstract-Maastrichtian-Mosasaurus-Meet.pdf.
- ↑ 118.0 118.1 Harrell Jr., T.L.; Pérez-Huerta, A.; Suarez, C.A. (2016). "Endothermic mosasaurs? Possible thermoregulation of Late Cretaceous mosasaurs (Reptilia, Squamata) indicated by stable oxygen isotopes in fossil bioapatite in comparison with coeval marine fish and pelagic seabirds". Palaeontology 59 (3): 351–363. doi:10.1111/pala.12240. Bibcode: 2016Palgy..59..351H.
- ↑ Tattersall, G.J.; Leite, C.A.C.; Sanders, C.E.; Cadena, V.; Andrade, D.V.; Abe, A.S.; Milsom, W.K. (2016). "Seasonal reproductive endothermy in tegu lizards". Science Advances 2 (1): e1500951. doi:10.1126/sciadv.1500951. PMID 26844295. Bibcode: 2016SciA....2E0951T.
- ↑ Cyrus C. Green (2018). Osteohistology And Skeletochronology Of an Ontogenetic Series Of Clidastes (Squamata: Mosasauridae): Growth And Metabolism In Basal Mosasaurids (MS). Fort Hays State University. doi:10.58809/MQVC1909. S2CID 91615758.
- ↑ 121.0 121.1 Carpenter, J.A. (2017). Locomotion and skeletal morphology of Late Cretaceous mosasaur, Tylosaurus proriger (BS). Georgia Southern University.
- ↑ Song, Y.; Lindgren, J. (2025). "Convergence in aquatic locomotion: reconstructing mosasaurian (Squamata: Mosasauria) tail fins from osteological correlates and covariation with extant sharks". Paleobiology: 1–10. doi:10.1017/pab.2025.10080.
- ↑ Massare, J (1988). "Swimming capabilities of Mesozoic marine reptiles: Implications for method of predation". Paleobiology 14 (2): 187–205. doi:10.1017/S009483730001191X. Bibcode: 1988Pbio...14..187M.
- ↑ 124.0 124.1 Larson, N.L. (2010), Enchoteuthididae, giant squids from the Upper Cretaceous of the Western Interior, https://www.researchgate.net/publication/313977725 In: 16th Annual Tate Conference, June 4–6, 2010: pp. 66-79.
- ↑ 125.0 125.1 Kauffman, E.G. (2004). "Mosasaur Predation on Upper Cretaceous Nautiloids and Ammonites from the United States Pacific Coast". PALAIOS 19 (1): 96–100. doi:10.1669/0883-1351(2004)019<0096:MPOUCN>2.0.CO;2. Bibcode: 2004Palai..19...96K. http://doc.rero.ch/record/14992/files/PAL_E2143.pdf.
- ↑ 126.0 126.1 126.2 Cicimurri, D.J.; Everhart, M.J. (2001). "An Elasmosaur with Stomach Contents and Gastroliths from the Pierre Shale (Late Cretaceous) of Kansas". Transactions of the Kansas Academy of Science 104 (3 & 4): 129–143. doi:10.1660/0022-8443(2001)104[0129:aewsca2.0.co;2].
- ↑ "Museum of Geology, South Dakota School of Mines". 2008. http://oceansofkansas.com/SDSMT.html.
- ↑ Martin, J.E.; Bjork, P.R. (1987). Martin, J.E.; Ostrander, G.E.. eds. "Gastric residues associated with a mosasaur from the Late Cretaceous (Campanian) Pierre Shale in South Dakota.". Papers in Vertebrate Paleontology in Honor of Morton Green: Dakoterra 3: 68–72.
- ↑ Royal Sask Museum [@royalsaskmuseum] (March 6, 2020). "A map showing the position of Omācīw, the Tylosaurus skeleton, as it was found in the quarry near Lake Diefenbaker in 1995. Its bones were arranged almost as they were in life.". https://twitter.com/royalsaskmuseum/status/1235950465801543680.
- ↑ Everhart 2017, p. 147-148.
- ↑ Pasch, A.D.; May, K.C. (2001). "Taphonomy and paleoenvironment of a hadrosaur (Dinosauria) from the Matanuska Formation (Turonian) in South-Central Alaska". Mesozoic Vertebrate Life. Indiana University Press. pp. 219–236. ISBN 978-0-253-33907-2. https://archive.org/details/mesozoicvertebra0000unse/page/219/mode/1up.
- ↑ Grigoriev, D.V.; Grabovskiy, A.A. (2020). "Arctic mosasaurs (Squamata, Mosasauridae) from the Upper Cretaceous of Russia". Cretaceous Research 114 (2020). doi:10.1016/j.cretres.2020.104499. Bibcode: 2020CrRes.11404499G. https://www.researchgate.net/publication/341319590.
- ↑ Rothschild, B.M.; Martin, L.D. (2005). "Mosasaur ascending: the phytogeny of bends". Netherlands Journal of Geosciences 84 (Special Issue 3): 341–344. doi:10.1017/S0016774600021120. Bibcode: 2005NJGeo..84..341R.
- ↑ Carlsen, A.W. (2017). "Frequency of decompression illness among recent and extinct mammals and "reptiles": a review". The Science of Nature 104 (7–8). doi:10.1007/s00114-017-1477-1. PMID 28656350. Bibcode: 2017SciNa.104...56C.
- ↑ Rothschild, B.; Everhart, M.J. (2015). "Co-Ossification of Vertebrae in Mosasaurs (Squamata, Mosasauridae); Evidence of Habitat Interactions and Susceptibility to Bone Disease". Transactions of the Kansas Academy of Science 118 (3&4): 265–275. doi:10.1660/062.118.0309. https://www.researchgate.net/publication/283849396.
- ↑ Nicholls, E.L.; Russell, A.P. (1990). "Paleobiogeography of the Cretaceous Western Interior Seaway of North America: the vertebrate evidence". Palaeogeography, Palaeoclimatology, Palaeoecology 79 (1–2): 149–169. doi:10.1016/0031-0182(90)90110-S. Bibcode: 1990PPP....79..149N.
- ↑ Bardet, N. (2012). "Maastrichtian marine reptiles of the Mediterranean Tethys: a palaeobiogeographical approach". Bulletin de la Société Géologique de France 183 (6): 573–596. doi:10.2113/gssgfbull.183.6.573. Bibcode: 2012BSGF..183..573B.
- ↑ Mateus, O.; Callapez, P.M.; Polcyn, M.J.; Schulp, A.S.; Gonçalves, A.O.; Jacobs, L.L. (2019). "The Fossil Record of Biodiversity in Angola Through Time: A Paleontological Perspective". Biodiversity of Angola. pp. 53–76. doi:10.1007/978-3-030-03083-4. ISBN 978-3-030-03082-7.
- ↑ Bardet, N.; Fischer, V.; Jalil, N-E.; Khaldoune, F.; Yazami, O.K.; Pereda-Suberbiola, X.; Longrich, N. (2025). "Mosasaurids Bare the Teeth: An Extraordinary Ecological Disparity in the Phosphates of Morocco Just Prior to the K/Pg Crisis". Diversity 17 (2): 114. doi:10.3390/d17020114. Bibcode: 2025Diver..17..114B.
- ↑ Russell 1967, p. 191.
- ↑ 141.0 141.1 Sheldon, A. (1997). "Ecological Implications of Mosasaur Bone Microstructure". Ancient Marine Reptiles. Academic Press. pp. 333–354. doi:10.1016/b978-012155210-7/50018-1. ISBN 978-0-12-155210-7.
- ↑ 142.0 142.1 Kiernan, C.R. (2002). "Stratigraphic distribution and habitat segregation of mosasaurs in the Upper Cretaceous of western and central Alabama, with a historical review of Alabama mosasaur discoveries". Journal of Vertebrate Paleontology 22 (1): 91–103. doi:10.1671/0272-4634(2002)022[0091:sdahso2.0.co;2]. https://www.researchgate.net/publication/254314199.
- ↑ Harrell Jr., T.L.; Pérez-Huerta, A. (2014). "Habitat preference of mosasaurs indicated by rare earth element (REE) content of fossils from the Upper Cretaceous marine deposits of Alabama, New Jersey, and South Dakota (USA)". Netherlands Journal of Geosciences 94 (1): 145–154. doi:10.1017/njg.2014.29.
- ↑ Robbins, J.A. (2010). Investigating Holocene Climate Change on the Northern Channel Islands and Cretaceous Mosasaur Ecology using Stable Isotopes (Ph.D. thesis). Southern Methodist University. ProQuest 849723494.
- ↑ 145.0 145.1 Polcyn, M.J.; Robbins, J.A.; Schulp, A.S.; Lindgren, J.; Jacobs, L.L. (2025). "The Evolution of Mosasaurid Foraging Behavior Through the Lens of Stable Carbon Isotopes". Diversity 17 (4): 291. doi:10.3390/d17040291. Bibcode: 2025Diver..17..291P.
- ↑ 146.0 146.1 Clementz, M.T.; Koch, P.L. (2001). "Differentiating aquatic mammal habitat and foraging ecology with stable isotopes in tooth enamel". Oecologia 129 (3): 461–472. doi:10.1007/s004420100745. PMID 28547202. Bibcode: 2001Oecol.129..461C.
Bibliography
- Russell, Dale A. (1967). Systematics and morphology of American mosasaurs. 23. New Haven: Bulletin of the Peabody Museum of Natural History. OCLC 205385. https://www.biodiversitylibrary.org/item/88808#page/7/mode/1up.
- Ellis, Richard (2003). Sea Dragons: Predators of the Prehistoric Oceans. Lawrence: University Press of Kansas. ISBN 978-0-7006-1394-6. https://archive.org/details/seadragonspredat0000elli/mode/1up.
- Everhart, Michael J. (2017). Oceans of Kansas, Second Edition: A Natural History of the Western Interior Sea (Life of the Past). 2. Bloomington: Indiana University Press. ISBN 978-0-253-02632-3.
External links
- Oceans of Kansas; includes much additional information and many illustrations and photographs of tylosaurines, including restorations and skeletons.
- E.D. Cope's original description of Macrosaurus proriger from western Kansas, with the published figure (1870) and recent photographs.
Template:Mosasauridae Wikidata ☰ Q131460 entry
 |