Chemistry:2-Bromopyridine
From HandWiki
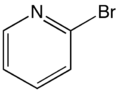
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Bromopyridine | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| C5H4NBr | |||
| Molar mass | 158.00 | ||
| Appearance | colorless liquid | ||
| Boiling point | 194.8 °C | ||
| Related compounds | |||
Related compounds
|
2-Chloropyridine 3-Bromopyridine | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tracking categories (test):
2-Bromopyridine is an isomer of bromopyridine with the formula BrC5H4N. It is a colorless liquid that is used as an intermediate in organic synthesis. It can be prepared from 2-aminopyridine via diazotization followed by bromination.[1]
Reactions
2-Bromopyridine reacts with butyllithium to give 2-lithiopyridine,[2] which is a versatile reagent.[3] Pyrithione can be prepared in a two-step synthesis from 2-bromopyridine by oxidation to the N-oxide with a suitable peracid followed by substitution using either sodium dithionite or sodium sulfide with sodium hydroxide to introduce the thiol functional group.[4]
References
- ↑ Allen, C. F. H.; Thirtle, John R. "2-Bromopyridine" Organic Syntheses 1946, volume 26, pp. 16-18. doi:10.15227/orgsyn.026.0016
- ↑ Yamamoto, Yasunori; Sugai, Juugaku; Takizawa, Miho; Miyaura, Norio (2011). "Synthesis of Lithium 2-Pyridyltriolborate and ITS Cross-Coupling Reaction with Aryl Halides". Organic Syntheses 88: 79. doi:10.15227/orgsyn.088.0079.
- ↑ Satinder V. Kessar, Paramjit Singh, Dmitry Zuev, Zhenlei Song, Ya Wu "2-Lithiopyridine" e-EROS Encyclopedia of Reagents for Organic Synthesis, 2015. doi:10.1002/047084289X.rl025.pub3
- ↑ Knight, David W.; Hartung, Jens (15 September 2006). "1-Hydroxypyridine-2(1H)-thione". Encyclopedia of Reagents for Organic Synthesis. John Wiley & Sons. doi:10.1002/047084289X.rh067.pub2. ISBN 0471936235.
 |