Chemistry:2-Ethylhexanal
2-Ethylhexanal is the organic compound with the formula CH
3CH
2CH
2CH
2CH(C
2H
5)CHO. A colorless liquid, it is produced on a large scale industrially as a precursor to 2-ethylhexanoic acid and 2-ethylhexanol, both used as precursors to plasticizers. It was studied in the detergent industry since the 1930s.[1]
2-Ethylhexanal is synthesized by aldol condensation of two equivalents of butyraldehyde followed by hydrogenation of the intermediate 2-ethylhexenal.[2] The compound is chiral, but is mainly used as a racemic mixture.
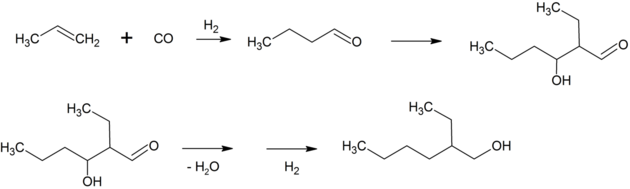
The n-butyraldehyde is made by hydroformylation of propylene, either in a self-contained plant or as the first step in a fully integrated facility. Most facilities make n-butanol and isobutanol in addition to 2-ethylhexanol. Alcohols prepared in this way are sometimes referred to as oxo alcohols. The overall process is very similar to that of the Guerbet reaction, by which it may also be produced.[3]
References
- ↑ Gangloff, W. C. (1938). "Changing trends in detergents" (in en). Oil & Soap 15 (1): 14–17. doi:10.1007/BF02549560. ISSN 2331-3420. https://aocs.onlinelibrary.wiley.com/doi/abs/10.1007/BF02549560.
- ↑ Kohlpaintner, Christian; Schulte, Markus; Falbe, Jürgen; Lappe, Peter; Weber, Jürgen (2008). "Aldehydes, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a01_321.pub2. ISBN 978-3-527-30673-2.
- ↑ Miller, Robert; Bennett, George (January 1961). "Producing 2-Ethylhexanol by the Guerbet Reaction". Industrial & Engineering Chemistry 53 (1): 33–36. doi:10.1021/ie50613a027.
 |
