Chemistry:Basketene
Basketene (IUPAC name: pentacyclo[4.4.0.02,5.03,8.04,7]dec-9-ene[1]) is an organic compound with the formula C10H10. It is a polycyclic alkene and the dehydrogenated version of basketane, which was named for its structural similarity to a basket. Due to its hydrocarbon composition and unique structure, the chemical compound is of considerable interest to those examining energy surfaces of these (CH)10 cage molecules and what possible factors influence their minima.[2] Additionally, the complex structure of this compound has intrigued researchers studying the chemistry of highly strained ring systems .[3] Basketene and its family of derivatives also have important chemical and physical properties. These molecules all tend to have a high standard enthalpy of formation, combined with their high density, leading to possible uses in explosives.[4]
Synthesis
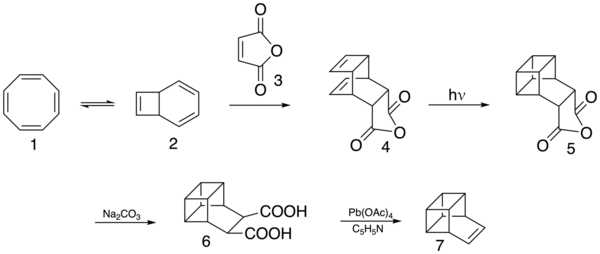
Basketene has been synthesized as follows. First cyclooctatetraene (1) undergoes thermal, electrocyclic isomerization (to 2), followed by a Diels–Alder reaction with maleic anhydride (3). [2 + 2] photochemical cycloaddition (4 to 5) closes the cage structure, which sodium carbonate hydrolyzes (to 6); lead(IV) acetate in pyridine then decarboxylates oxidatively to basketene (7).[5]
Reactions
Basketene and other cage molecules are important for discovering and testing new concepts of bonding and reactivity.[4] Oxidizing basketene with osmium tetroxide selectively cleaves the alkene to two carboxylic acid groups; ketonization then gives a cubane precursor.[6]
Basketene also undergoes a number of different rearrangements.
Rearrangements
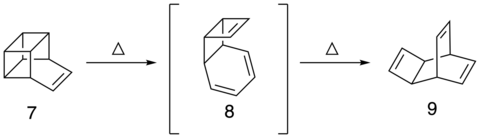
Heating Basketene alone produces Nenitzescu’s Hydrocarbon. The reaction proceeds through a reverse Diels-Alder intermediate in order to give 8 at 110 ˚C. From 8, a 3,3 shift occurs to give Nenitzescu's hydrocarbon (9).[2]
Basketene can also undergo the following thermal and photochemical rearrangement reactions. The photo-induced conrotary opening of 8 gives cis,cis,trans-cyclooctatriene (10). Furthermore, the photo-rearrangement of 10 provides the derivative 11. 11 will undergo a direct photochemical (2+2) cyclization closure to 12. :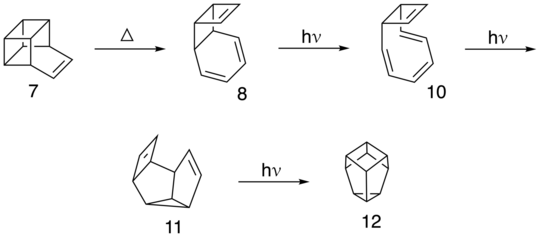
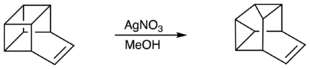
Basktene also rearranges into snoutene when in solution with Silver nitrate[1]
References
- ↑ 1.0 1.1 Verevkin, Sergei P.; Martin Kümmerlin; Ernst Hickl; Hans-Dieter Beckhaus; Christoph Rüchardt; Sergei I. Kozhushkov; Rainer Haag; Roland Boese et al. (2002). "Thermochemical and X-ray Crystallographic Investigations of Some (CH)10 Hydrocarbons: Basketene, Nenitzescu's Hydrocarbon, and Snoutene". European Journal of Organic Chemistry (Wiley VCH) 2002 (14): 2280–7. doi:10.1002/1099-0690(200207)2002:14<2280::AID-EJOC2280>3.0.CO;2-R.
- ↑ 2.0 2.1 Allred, Evan L.; Beck, Boyd R. (1973). "Concurrent thermolysis and photolysis of basketene. Formation and the interrelation of some new (CH)10 isomers". Journal of the American Chemical Society 95 (7). doi:10.1021/ja00788a065.
- ↑ Gassman, Paul G.; Yamaguchi, Ryohei (1978). "1,8-Bishomocubane". Journal of Organic Chemistry 43 (24). doi:10.1021/jo00418a028.
- ↑ 4.0 4.1 Aydinli, Betül; Çelik, Murat; M. Serdar, Gültekin; Uzun, Orhan; Balci, Metin (2003). "Controlled Synthesis of Substituted Benzobasketene Derivatives". Helvetica Chimica Acta 86. doi:10.1002/hlca.200390276.
- ↑ Smit, W. A.; Bochkov, A. F.; Caple, R. (1998). Organic Synthesis: The Science Behind the Art. The Royal Society of Chemistry. p. 184.
- ↑ Chin, Clinton G.; Cuts, Harold W.; Masamune, Satoru (1966). "Strained systems: cubane" (in en). Chemical Communications (23): 880. doi:10.1039/c19660000880. ISSN 0009-241X. https://xlink.rsc.org/?DOI=c19660000880.
 |