Chemistry:Cetyl palmitate
From HandWiki
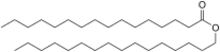
| |
| Names | |
|---|---|
| Preferred IUPAC name
Hexadecyl hexadecanoate | |
| Other names
Palmityl palmitate
Palmitic acid palmityl ester Palmitic acid hexadecyl ester Palmitic acid cetyl ester n-hexadecyl palmitate Palmatic acid n-hexadecyl ester | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C32H64O2 | |
| Molar mass | 480.862 g·mol−1 |
| Appearance | Colourless wax |
| Melting point | 54 °C (129 °F; 327 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Hexadecyl hexadecanoate, also known as cetyl palmitate, is the ester derived from hexadecanoic acid and 1-hexadecanol. This white waxy solid is the primary constituent of spermaceti, the once highly prized wax found in the skull of sperm whales.[2] Cetyl palmitate is a component of some solid lipid nanoparticles.
Stony corals, which build the coral reefs, contain large amounts of cetyl palmitate wax in their tissues, which may function in part as an antifeedant.[3]
Applications
Cetyl palmitate is used in cosmetics as a thickener and emulsifier.[4]
References
- ↑ http://www.sciencelab.com/msds.php?msdsId=9923365
- ↑ Wilhelm Riemenschneider and Hermann M. Bolt "Esters, Organic" Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a09_565.pub2
- ↑ Dobretsov, S.; Al-Wahaibi, A. S. M.; Lai, D.; Al-Sabahi, J.; Claereboudt, M.; Proksch, P.; Soussi, B., "Inhibition of Bacterial Fouling by Soft Coral Natural Products", International Biodeterioration & Biodegradation 2015, volume 98, 53-58. doi:10.1016/j.ibiod.2014.10.019
- ↑ "Cetyl Palmitate for Skin - is It Safe? | Skincare Lab". 10 July 2020. https://www.skincarelab.org/cetyl-palmitate/.
 |
