Chemistry:Coniferyl aldehyde
From HandWiki
| |
| Names | |
|---|---|
| IUPAC names
(Z)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enal
(E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enal | |
| Other names
coniferaldehyde
cis-coniferyl aldehyde trans-coniferyl aldehyde | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C10H10O3 | |
| Molar mass | 178.18 g/mol |
| Density | 1.186 g/mL |
| Melting point | 80 °C (176 °F; 353 K) |
| Boiling point | 338.8 °C (641.8 °F; 612.0 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
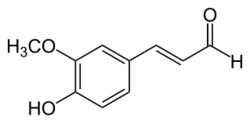
Coniferyl aldehyde is an organic compound with the formula HO(CH3O)C6H3CH=CHCHO. It is a derivative of cinnamaldehyde, featuring 4-hydroxy and 3-methoxy substituents. It is a major precursor to lignin.[1][2]
Biosynthetic role
In sweetgum (Liquidambar styraciflua), coniferyl aldehyde is a precursor to sinapaldehyde via hydroxylation mediated by coniferyl aldehyde 5-hydroxylase.[3]
Coniferyl aldehyde is reduced to coniferyl alcohol by the action of dehydrogenase enzymes.[2]
It is found in Senra incana (Hibisceae). It is a low molecular weight phenol that is susceptible to extraction from cork stoppers into wine.[4]
See also
- Phenolic compounds in wine
References
- ↑ Wout Boerjan, John Ralph, Marie Baucher "Lignin Biosynthesis" Annu. Rev. Plant Biol. 2003, vol. 54, pp. 519–46. doi:10.1146/annurev.arplant.54.031902.134938
- ↑ 2.0 2.1 Li, Laigeng; Cheng, Xiao Fei; Leshkevich, Jacqueline; Umezawa, Toshiaki; Harding, Scott A.; Chiang, Vincent L. (2001). "The Last Step of Syringyl Monolignol Biosynthesis in Angiosperms is Regulated by a Novel Gene Encoding Sinapyl Alcohol Dehydrogenase". The Plant Cell 13 (7): 1567–1586. doi:10.1105/tpc.010111. PMID 11449052.
- ↑ Osakabe, Keishi; Tsao, Cheng Chung; Li, Laigeng; Popko, Jacqueline L.; Umezawa, Toshiaki; Carraway, Daniel T.; Smeltzer, Richard H.; Joshi, Chandrashekhar P. et al. (1999). "Coniferyl aldehyde 5-hydroxylation and methylation direct syringyl lignin biosynthesis in angiosperms". Proceedings of the National Academy of Sciences 96 (16): 8955–8960. doi:10.1073/pnas.96.16.8955. PMID 10430877. Bibcode: 1999PNAS...96.8955O.
- ↑ Polyphenolic Composition of Quercus suber Cork from Different Spanish Provenances. Elvira Conde, Estrella Cadahía, María Concepción García-Vallejo and Brígida Fernández de Simón, J. Agric. Food Chem., 1998, volume 46, pp 3166–3171 doi:10.1021/jf970863k
