Chemistry:Di-tert-butyl chromate
| |
| Names | |
|---|---|
| IUPAC name
tert-Butyl chromate
| |
| Other names
Di-tert-butyl ester of chromic acid; Bis(tert-butyl)chromate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| RTECS number |
|
| |
| |
| Properties | |
| [(CH3)3CO]2CrO2 | |
| Molar mass | 230.3 g/mol[1] |
| Appearance | red oil |
| Melting point | −2.8 °C (27.0 °F; 270.3 K)[2] |
| Miscible[2] | |
| Hazards | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 0.005 mg CrO3/m3 [skin][1] |
REL (Recommended)
|
Ca TWA 0.001 mg Cr(VI)/m3[1] |
IDLH (Immediate danger)
|
Ca [15 mg/m3 {as Cr(VI)}][1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
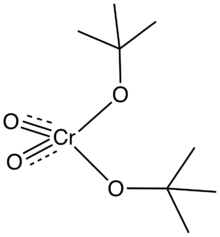
Di-tert-butyl chromate is an alkoxide with the formula CrO2(OC(CH3)3)2. It is prepared by treatment of t-butanol with chromic anhydride. It forms red crystals at temperatures below –5 °C, above which it melts to give a red oil.[3][4] The complex, which is diamagnetic, is of fundamental interest as a model for the intermediates in oxidations of alcohols by chromium(VI). This complex is stable because as a t-butyl groups lack beta-hydrogens. This complex and its analogues have tetrahedral geometry at chromium, as established by X-ray crystallography of its analogues.[5][6]
Applications
It is used as a precursor to chromium-based catalysts, such as the Phillips catalyst, which are employed for the polymerization of ethylene.[7]
Safety
Like other forms of hexavalent chromium, di-tert-butyl chromate is classified as a potential carcinogen by the United States National Institute for Occupational Safety and Health.[1][2]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 NIOSH Pocket Guide to Chemical Hazards. "#0080". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0080.html.
- ↑ 2.0 2.1 2.2 "Tert-butyl chromate". International Chemical Safety Cards. NIOSH. July 1, 2014. https://www.cdc.gov/niosh/ipcsneng/neng1533.html.
- ↑ Freeman, Fillmore (2001-04-15), "Di-tert-butyl Chromate" (in en), Encyclopedia of Reagents for Organic Synthesis, John Wiley & Sons, Ltd, doi:10.1002/047084289x.rd059m, ISBN 978-0471936237
- ↑ Richer, Jean-Claude; Hachey, Jean-Marie (1975). "Esters Chromiques Dérivés d'Alcools Tertiaires". Canadian Journal of Chemistry 53 (20): 3087–3093. doi:10.1139/v75-438.
- ↑ Amirthalingam, V.; Grant, D. F.; Senol, A. (1972). "The crystal structure and absolute configuration of cedryl chromate". Acta Crystallographica Section B: Structural Crystallography and Crystal Chemistry 28 (5): 1340–1345. doi:10.1107/S0567740872004261.
- ↑ Stavropoulos, Pericles; Bryson, Nathan; Youinou, Marie Therese; Osborn, John A. (1990). "Chromyl complexes with aryloxy and siloxy ligands". Inorganic Chemistry 29 (10): 1807–1811. doi:10.1021/ic00335a009.
- ↑ McDaniel, M. (2009). "Influence of porosity on PE molecular weight from the Phillips Cr/Silica catalyst". Journal of Catalysis 261: 34–49. doi:10.1016/j.jcat.2008.10.015.
 |

