Chemistry:Dithioerythritol
| |
| Names | |
|---|---|
| Preferred IUPAC name
(2R,3S)-1,4-Bis(sulfanyl)butane-2,3-diol | |
| Other names
(2R,3S)-1,4-Dimercaptobutane-2,3-diol (no longer recommended)
2,3-Dihydroxybutane-1,4-dithiol Erythro-2,3-dihydroxy-1,4-butanedithiol Erythro-1,4-dimercapto-2,3-butanediol Cleland's reagent (also used for DTT) | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C4H10O2S2 | |
| Molar mass | 154.253 g/mol |
| Melting point | 82 to 84 °C (180 to 183 °F; 355 to 357 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
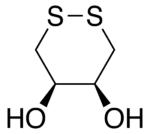
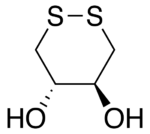
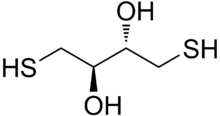
Dithioerythritol (DTE) is a sulfur containing sugar alcohol derived from the corresponding 4-carbon monosaccharide erythrose. It is an epimer of dithiothreitol (DTT). The molecular formula for DTE is C4H10O2S2.
Chemical properties
DTE is a crystalline solid soluble in water and alcohols.[1][2]
Applications
Like DTT, DTE makes an excellent reducing agent, which can be used for reduction of disulfide bonds. The reduction potential of DTE is the same as for DTT, about –0.331 mV.[2] The pKa values of the thiol groups of DTE are 9.0 and 9.9, which is higher than the corresponding values for DTT (9.3 and 9.5).[2] Since reduction of disulfide bonds requires thiolate (ionized thiol), DTE is less efficient at lower pH compared to DTT.[2]
Reduction with DTE is slower than with DTT. This is presumably because the orientation of the OH groups in its cyclic disulfide-bonded form (oxidized form) is less stable due to greater steric repulsion than their orientation in the disulfide-bonded form of DTT. In the disulfide-bonded form of DTT, these hydroxyl groups are trans to each other, whereas they are cis to each other in DTE.[3]
References
- ↑ Cleland, W.W. (April 1964). "Dithiothreitol, A New Protective Reagent for SH Groups". Biochemistry 3 (4): 480–2. doi:10.1021/bi00892a002. PMID 14192894.
- ↑ 2.0 2.1 2.2 2.3 Jocelyn, Peter C. (1987). "Chemical reduction of disulfides". Sulfur and Sulfur Amino Acids. Methods in Enzymology. 143. Academic Press. pp. 246–256. doi:10.1016/0076-6879(87)43048-6. ISBN 9780121820435.
- ↑ Loechler, Edward L.; Hollocher, Thomas C. (November 1980). "Reduction of flavins by thiols. 1. Reaction mechanism from the kinetics of the attack and breakdown steps" (in en). Journal of the American Chemical Society 102 (24): 7312–7321. doi:10.1021/ja00544a027. ISSN 0002-7863. https://pubs.acs.org/doi/abs/10.1021/ja00544a027.
External links
 |