Chemistry:Djenkolic acid
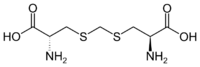
| |
| Names | |
|---|---|
| IUPAC name
(2R)-2-Amino-3-[[(2R)-2-amino-3-hydroxy-3-oxopropyl] sulfanylmethylsulfanyl]propanoic acid
| |
| Other names
Djenkolate; Jengkolic acid; S,S'-Methylenebiscysteine[1]
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C7H14N2O4S2 | |
| Molar mass | 254.33 g/mol |
| 1.02 g L−1 (at 30±0.5°C)[2] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Djenkolic acid (or sometimes jengkolic acid) is a sulfur-containing non-protein amino acid naturally found in the djenkol beans of the Southeast Asian plant Archidendron jiringa. Its chemical structure is similar to cystine but contains a methylene (single carbon) unit in between the two sulfur atoms. There is about 20 grams of djenkolic acid per kilogram of dry djenkol beans, and it has also been reported in smaller amounts in the seeds of other leguminous plants such as Leucaena esculenta (2.2 g/kg) and Pithecolobium ondulatum (2.8 g/kg).[3]
Toxicity
The toxicity of djenkolic acid in humans arises from its poor solubility under acidic conditions after consumption of the djenkol bean.[3] The amino acid precipitates into crystals which cause mechanical irritation of the renal tubules and urinary tract, resulting in symptoms such as abdominal discomfort, loin pains, severe colic, nausea, vomiting, dysuria, gross hematuria, and oliguria, occurring 2 to 6 hours after the beans were ingested.[4] Urine analysis of patients reveals erythrocytes, epithelial cells, protein, and the needle-like crystals of djenkolic acid. Urolithiasis can also happen, with djenkolic acid as the nucleus. In young children, it has also been reported to produce painful swelling of the genitalia.[5]
Treatment for this toxicity requires hydration to increase urine flow and alkalinization of urine by sodium bicarbonate. Furthermore, this poisoning can be prevented when consuming djenkol beans by boiling them beforehand, since djenkolic acid is removed from the beans.[4]
Discovery and synthesis
Djenkolic acid was first isolated by Van Veen and Hyman in 1933[6] from the urine of the natives of Java who had eaten the djenkol bean and were suffering from poisoning. They then isolated the djenkolic acid crystals by treating the djenkol beans with barium hydroxide at 30°C for a prolonged period.[2]
Du Vigneaud and Patterson managed to synthesize djenkolic acid by condensation of methylene chloride with 2 moles of L-cysteine in liquid ammonia.[2] Later on, Armstrong and du Vigneaud prepared djenkolic acid by the direct combination of 1 mole of formaldehyde with 2 moles of L-cysteine in a strongly acidic solution.[7]
References
- ↑ "Djenkolic acid". The On-line Medical Dictionary. 5 March 2000. http://cancerweb.ncl.ac.uk/cgi-bin/omd?djenkolic+acid.[|permanent dead link|dead link}}]
- ↑ 2.0 2.1 2.2 du Vigneaud V, Patterson WI (1936). "The synthesis of djenkolic acid". J. Biol. Chem. 114 (2): 533–538. doi:10.1016/S0021-9258(18)74825-4. http://www.jbc.org/cgi/reprint/114/2/533.pdf.
- ↑ 3.0 3.1 D'Mello, J. P. Felix (1991). Toxic Amino Acids. In J. P. F. D'Mello, C. M. Duffus, J. H. Duffus (Eds.) Toxic Substances in Crop Plants. Woodhead Publishing. pp. 21–48. ISBN:0-85186-863-0. Google Book Search. Retrieved on November 15, 2008.
- ↑ 4.0 4.1 Barsoum, R. S., & Sitprija, V. (2007). Tropical Nephrology. In R. W. Schrier (Ed.) Diseases of the Kidney and Urinary Tract: Clinicopathologic Foundations of Medicine. Lippincott Williams & Wilkins. p. 2037. ISBN:0-7817-9307-6. Google Book Search. Retrieved on November 15, 2008.
- ↑ J. B. Harborne, H. Baxter, G. P. Moss (Eds.) (1999) Phytochemical Dictionary: A Handbook of Bioactive Compounds from Plants. CRC Press. p. 81. ISBN:0-7817-9307-6. Google Book Search. Retrieved on November 16, 2008.
- ↑ van Veen AG, Hyman AJ (1933). "On the toxic component of the djenkol bean". Geneesk. Tijdschr. Nederl. Indie 73: 991.
- ↑ Armstrong MD, du Vigneaud V (1947). "A new synthesis of djenkolic acid". J. Biol. Chem. 168 (1): 373–377. doi:10.1016/S0021-9258(17)35126-8. PMID 20291097. http://www.jbc.org/cgi/reprint/168/1/373.pdf.
 |

