Chemistry:Flupyradifurone
| |
| Names | |
|---|---|
| IUPAC name
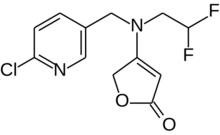
4-[(6-Chloropyridine-3-yl)methyl](2,2-difluoroethyl)amino}furan-2(5H)-one
| |
| Other names
Flupyradifurone, Sivanto
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C12H11ClF2N2O2 | |
| Molar mass | 288.68 g·mol−1 |
| Appearance | White to beige solid |
| Odor | None |
| Density | 1.43 g/mL |
| Melting point | 69 °C (156 °F; 342 K) |
| 3.2 g/L (pH 4)
3.0 g/L (pH 7) | |
| Solubility in Other | 0.0005 g/L (n-Heptane)
>250 g/L (Methanol) |
| Hazards | |
| GHS pictograms |   
|
| GHS Signal word | Warning |
| H302, H373, H410 | |
| P260, P264, P270, P273, P301+312, P314, P330, P391, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Flupyradifurone is a systemic butenolide insecticide developed by Bayer CropScience under the name Sivanto. Flupyradifurone protects crops from sap-feeding pests such as aphids and is safer for non-target organisms compared to other insecticides.[1] Sivanto was launched in 2014 since it obtained its first commercial registration in central America (Guatemala and Honduras).[2] Insecticide Resistance Action Committee (IRAC) classified Flupyradifurone as 4D subset (butenolide) and it is the first pesticide in the butenolide category.[3][4] It was approved by European Union in 2015.[5]
Characteristics
Sap-feeding insects have developed resistance to nearly all other kinds of insecticides[6] but flupyradifurone is effective at controlling aphids and whitefly, thereby maintaining yields of crops such as vegetables, fruits, cotton, and coffee.[7] It also demonstrated positive toxicological and ecotoxicological safety.[2]
According to a study by Bayer, who developed the compound, flupyradifurone has a high rate of uptake by plants and crops. The authors used phosphor imaging analysis to monitor the uptake and translocation of [pyridinylmethyl-14C]-labeled flupyradifurone by tomato plants.[2] The result revealed fast root uptake and the even distribution of labeled flupyradifurone in the entire plant. Flupyradifurone is delivered via xylem translocation in the plants. The concentration in the plants reached the highest point in 7 and 14 days. After 24 days of the application, a significant decline is observed.[2] Flupyradifurone also featured excellent speed of action. The speed of action can be observed by the prohibition of honeydew excretion. After applying the flupyradifurone to the plant by spraying, the honeydew excretion is inhibited within 2 hours and all the aphids die after 48 hours. The result demonstrated that flupyradifurone has an exceptional speed of action compared to other insecticides.[2]
Flupyradifurone presented potent efficacy in controlling aphids and whiteflies. According to bioassays research, the flupyradifurone has lower LC50 numbers of aphid and whitefly compared to imidacloprid.[2] In the field trial, flupyradifurone controlled the lettuce aphid at a favorable rate via foliar applying. It had the highest efficiency (96%) of controlling lettuce aphid after 6–10 days of the application.[2]
Mode of action
Flupyradifurone is an agonist of insect nicotinic acetylcholine receptors,[8][2] causing depolarization of nerve cells membranes.[7] Insects cannot detoxify flupyradifurone according to the research of CYP6CM1-mediated metabolism.[2] Since flupyradifurone cannot be inactivated by acetylcholinesterase, it will lead to the failure of nerve system of insects and end up with the death of insects.[7]
Synthesis
Flupyradifurone is structurally related to the natural insecticide, stemofoline which occurs in the plant species Stemona japonica.[9][2] Stemofoline has some advantageous features, such as being fast-acting and an effective antifeedant, but does not efficiently bind to insect nicotinic acetylcholine receptors.[10][11]
There are two ways to synthesis flupyradifurone. First, flupyradifurone can be synthesized from reacting tetronic acid with 2,2-difluoroethylamine to produce the intermediate, 4-[(2-fluoroethyl)amino]furane-2(5H)-one. Heating the reaction intermediate with 2-chloro-5-(chloromethyl)pyridine in THF under reflux yields flupyradifurone.[2] In the second method, tetronic acid, 4-touluenesulfonic acid, and N-[(6-chloropyridin-3-yl)methyl]-2,2-difluoroethane-1-amine in toluene are heated under reflux for 2 hours to produce flupyradifurone.[2]
Toxicological and ecotoxicological safety
Using the U.S. Environmental Protection Agency guidelines to classify carcinogenic effects, flupyradifurone is categorized as "not likely to be carcinogenic to humans." It is not irritant to humans' eyes and skin,[2][12] and the EPA is not concerned about occupational exposure.[12]
For the non-target species, flupyradifurone also presents toxicological safety at low concentration.[12][disputed (for: ref states "Aquatic Life Exposure – Flupyradifurone is very highly toxic to aquatic and benthic invertebrates." and that it is mobile from treated fields into groundwater) ] Flupyradifurone will still bind to the nicotinic acetylcholine receptors of honey bees, however, it will only affect the taste and appetitive learning performance at the highest labeled concentration.[13] The LD50 of honey bee acute contact is >100 μg/bee and acute oral is 1200 ng/bee. For bumblebee, the acute contact LD50 is >100 μg/bee.
References
- ↑ Jeschke, Peter; Nauen, Ralf; Beck, Michael Edmund (2013). "Nicotinic Acetylcholine Receptor Agonists: A Milestone for Modern Crop Protection" (in en). Angewandte Chemie International Edition 52 (36): 9464–9485. doi:10.1002/anie.201302550. ISSN 1521-3773. PMID 23934864.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 Nauen, Ralf; Jeschke, Peter; Velten, Robert; Beck, Michael E.; Ebbinghaus-Kintscher, Ulrich; Thielert, Wolfgang; Wölfel, Katharina; Haas, Matthias et al. (2015). "Flupyradifurone: a brief profile of a new butenolide insecticide" (in en). Pest Management Science 71 (6): 850–862. doi:10.1002/ps.3932. ISSN 1526-4998. PMID 25351824.
- ↑ "Interactive MoA Classification | Insecticide Resistance Action Committee (IRAC)". https://irac-online.org/modes-of-action/.
- ↑ "Regulations.gov". https://www.regulations.gov/document/EPA-HQ-OPP-2013-0226-0015.
- ↑ (in en) Commission Implementing Regulation (EU) 2015/2084 of 18 November 2015 approving the active substance flupyradifurone, in accordance with Regulation (EC) No 1107/2009 of the European Parliament and of the Council concerning the placing of plant protection products on the market, and amending the Annex to Commission Implementing Regulation (EU) No 540/2011 (Text with EEA relevance), 2015-11-19, http://data.europa.eu/eli/reg_impl/2015/2084/oj/eng, retrieved 2021-12-01
- ↑ Bass, Chris; Puinean, Alin M.; Zimmer, Christoph T.; Denholm, Ian; Field, Linda M.; Foster, Stephen P.; Gutbrod, Oliver; Nauen, Ralf et al. (2014-08-01). "The evolution of insecticide resistance in the peach potato aphid, Myzus persicae" (in en). Insect Biochemistry and Molecular Biology 51: 41–51. doi:10.1016/j.ibmb.2014.05.003. ISSN 0965-1748. PMID 24855024.
- ↑ 7.0 7.1 7.2 "Bayer Sivanto :: The science behind Sivanto". https://www.sivanto.bayer.com/sivanto-science.html#sivanto-compound-information.
- ↑ "Flupyradifurone: A new insecticide or just another neonicotinoid?" (in en-US). 2015-02-05. https://www.pesticideresearch.com/site/2015/02/05/flupyradifurone-a-new-insecticide-or-just-another-neonicotinoid/.
- ↑ Yamamoto, Izuru; Casida, John E., eds (1999) (in en-gb). Nicotinoid Insecticides and the Nicotinic Acetylcholine Receptor. p. 46. doi:10.1007/978-4-431-67933-2. ISBN 978-4-431-68011-6.
- ↑ Kaltenegger, Elisabeth; Brem, Brigitte; Mereiter, Kurt; Kalchhauser, Hermann; Kählig, Hanspeter; Hofer, Otmar; Vajrodaya, Srumya; Greger, Harald (August 2003). "Insecticidal pyrido[1,2-a]azepine alkaloids and related derivatives from Stemona species". Phytochemistry 63 (7): 803–816. doi:10.1016/s0031-9422(03)00332-7. ISSN 0031-9422. PMID 12877922.
- ↑ Mungkornasawakul, Pitchaya; Pyne, Stephen G.; Jatisatienr, Araya; Supyen, Damrat; Jatisatienr, Chaiwat; Lie, Wilford; Ung, Alison T.; Skelton, Brian W. et al. (2004-03-02). "Phytochemical and Larvicidal Studies on Stemona curtisii: Structure of a New Pyrido[1,2-a]azepine Stemona Alkaloid". Journal of Natural Products 67 (4): 675–677. doi:10.1021/np034066u. ISSN 0163-3864. PMID 15104502.
- ↑ 12.0 12.1 12.2 "New Active Ingredient and New Use Special Registration Reviews" (in en). Minnesota Department of Agriculture. https://www.mda.state.mn.us/chemicals/pesticides/regs/newreviews.
- ↑ Hesselbach, Hannah; Scheiner, Ricarda (2018-03-21). "Effects of the novel pesticide flupyradifurone (Sivanto) on honeybee taste and cognition" (in en). Scientific Reports 8 (1): 4954. doi:10.1038/s41598-018-23200-0. ISSN 2045-2322. PMID 29563522. Bibcode: 2018NatSR...8.4954H.
 |


